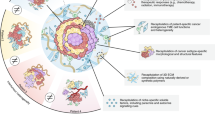
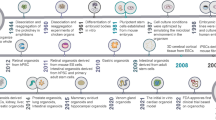
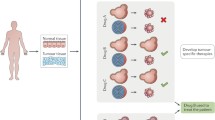
Abstract
Tumor heterogeneity is a major challenge for breast cancer researchers who have struggled to find effective treatments despite recent advances in oncology. Although the use of 2D cell culture methods in breast cancer research has been effective, it cannot model the heterogeneity of breast cancer as found within the body. The development of 3D culture of tumor cells and breast cancer organoids has provided a new approach in breast cancer research, allowing the identification of biomarkers, study of the interaction of tumor cells with the microenvironment, and for drug screening and discovery. In addition, the possibility of gene editing in organoids, especially using the CRISPR/Cas9 system, is convenient, and has allowed a more detailed study of tumor behavior in models closer to the physiological condition. The present review covers the application of organoids in breast cancer research. The recent use of gene-editing systems to provide insights into therapeutic approaches for breast cancer, is highlighted. The study of organoids and the possibility of gene manipulation may be a step towards the personalized treatment of breast cancer, which has so far remained unattainable due to the high heterogeneity of breast cancer.



Similar content being viewed by others
Availability of data and materials
Not applicable.
References
Abbott A (2003) Goodbye, flat biology. Nature 424:861
Abdel Hadi M, Alkhunaizi Z, Elsharkawy T, Jindan F, Iftikhar M (2021) Challenges in breast cancer margins: a 10 years audit
Ali HR, Rueda OM, Chin S-F, Curtis C, Dunning MJ, Aparicio SA, Caldas C (2014) Genome-driven integrated classification of breast cancer validated in over 7,500 samples. Genome Biol 15(8):1–14
Arun G, Diermeier S, Akerman M, Chang K-C, Wilkinson JE, Hearn S, Kim Y, MacLeod AR, Krainer AR, Norton L (2016) Differentiation of mammary tumors and reduction in metastasis upon Malat1 lncRNA loss. Genes Dev 30(1):34–51
Asghar W, El Assal R, Shafiee H, Pitteri S, Paulmurugan R, Demirci U (2015) Engineering cancer microenvironments for in vitro 3-D tumor models. Mater Today 18(10):539–553
Barrangou R, Fremaux C, Deveau H, Richards M, Boyaval P, Moineau S, Romero DA, Horvath P (2007) CRISPR provides acquired resistance against viruses in prokaryotes. Science 315(5819):1709–1712
Bartfeld S, Clevers H (2015) Organoids as model for infectious diseases: culture of human and murine stomach organoids and microinjection of Helicobacter pylori. J Vis Exp JoVE (105)
Ben-David U, Siranosian B, Ha G, Tang H, Oren Y, Hinohara K, Strathdee CA, Dempster J, Lyons NJ, Burns R (2018) Genetic and transcriptional evolution alters cancer cell line drug response. Nature 560(7718):325–330
Beumer J, Artegiani B, Post Y, Reimann F, Gribble F, Nguyen TN, Zeng H, Van den Born M, Van Es JH, Clevers H (2018) Enteroendocrine cells switch hormone expression along the crypt-to-villus BMP signalling gradient. Nat Cell Biol 20(8):909–916
Bissell MJ, Hall HG, Parry G (1982) How does the extracellular matrix direct gene expression? J Theor Biol 99(1):31–68. https://doi.org/10.1101/sqb.2005.70.013
Bissell M, Kenny P, Radisky D (2005) Microenvironmental regulators of tissue structure and function also regulate tumor induction and progression: the role of extracellular matrix and its degrading enzymes. Cold Spring Harbor Symp Quant Biol. https://doi.org/10.1101/sqb.2005.70.013
Boj SF, Hwang C-I, Baker LA, Chio IIC, Engle DD, Corbo V, Jager M, Ponz-Sarvise M, Tiriac H, Spector MS (2015) Organoid models of human and mouse ductal pancreatic cancer. Cell 160(1–2):324–338
Breslin S, O’Driscoll L (2016) The relevance of using 3D cell cultures, in addition to 2D monolayer cultures, when evaluating breast cancer drug sensitivity and resistance. Oncotarget 7(29):45745
Bruna A, Rueda OM, Greenwood W, Batra AS, Callari M, Batra RN, Pogrebniak K, Sandoval J, Cassidy JW, Tufegdzic-Vidakovic A (2016) A biobank of breast cancer explants with preserved intra-tumor heterogeneity to screen anticancer compounds. Cell 167(1):260-274. e222
Burdall SE, Hanby AM, Lansdown MR, Speirs V (2003) Breast cancer cell lines: friend or foe? Breast Cancer Res 5(2):1–7
Byrne AT, Alferez DG, Amant F, Annibali D, Arribas J, Biankin AV, Bruna A, Budinska E, Caldas C, Chang DK (2017) Interrogating open issues in cancer medicine with patient-derived xenografts. Nat Rev Cancer 17(10):254
Camp JG, Badsha F, Florio M, Kanton S, Gerber T, Wilsch-Bräuninger M, Lewitus E, Sykes A, Hevers W, Lancaster M (2015) Human cerebral organoids recapitulate gene expression programs of fetal neocortex development. Proc Natl Acad Sci 112(51):15672–15677
Campaner E, Zannini A, Santorsola M, Bonazza D, Bottin C, Cancila V, Tripodo C, Bortul M, Zanconati F, Schoeftner S (2020) Breast cancer organoids model patient-specific response to drug treatment. Cancers 12(12):3869
Caponigro G, Sellers WR (2011) Advances in the preclinical testing of cancer therapeutic hypotheses. Nat Rev Drug Discov 10(3):179–187
Cellurale C, Girnius N, Jiang F, Cavanagh-Kyros J, Lu S, Garlick DS, Mercurio AM, Davis RJ (2012) Role of JNK in mammary gland development and breast cancer. Can Res 72(2):472–481
Chen K-Y, Srinivasan T, Lin C, Tung K-L, Gao Z, Hsu DS, Lipkin SM, Shen X (2018) Single-cell transcriptomics reveals heterogeneity and drug response of human colorectal cancer organoids. In: 2018 40th Annual international conference of the IEEE engineering in medicine and biology society (EMBC). IEEE
Chen M, Mao A, Xu M, Weng Q, Mao J, Ji J (2019) CRISPR-Cas9 for cancer therapy: opportunities and challenges. Cancer Lett 447:48–55
Cheon D-J, Orsulic S (2011) Mouse models of cancer. Annu Rev Pathol 6:95–119
Christgen M, Steinemann D, Kühnle E, Länger F, Gluz O, Harbeck N, Kreipe H (2016) Lobular breast cancer: clinical, molecular and morphological characteristics. Pathol Res Practice 212(7):583–597
Clevers H (2016) Modeling development and disease with organoids. Cell 165(7):1586–1597
Corda G, Sala G, Lattanzio R, Iezzi M, Sallese M, Fragassi G, Lamolinara A, Mirza H, Barcaroli D, Ermler S (2017) Functional and prognostic significance of the genomic amplification of frizzled 6 (FZD6) in breast cancer. J Pathol 241(3):350–361
Cyranoski D (2016) Chinese scientists to pioneer first human CRISPR trial. Nature 535(7613):476–477
Czerniecki SM, Cruz NM, Harder JL, Menon R, Annis J, Otto EA, Gulieva RE, Islas LV, Kim YK, Tran LM (2018) High-throughput screening enhances kidney organoid differentiation from human pluripotent stem cells and enables automated multidimensional phenotyping. Cell Stem Cell 22(6):929-940.e924
Dart A (2018) Organoid diversity. Nat Rev Cancer 18(7):404–405
Dekkers JF, Whittle JR, Vaillant F, Chen H-R, Dawson C, Liu K, Geurts MH, Herold MJ, Clevers H, Lindeman GJ (2020) Modeling breast cancer using CRISPR-Cas9–mediated engineering of human breast organoids. JNCI J Natl Cancer Inst 112(5):540–544
Dekkers JF, van Vliet EJ, Sachs N, Rosenbluth JM, Kopper O, Rebel HG, Wehrens EJ, Piani C, Visvader JE, Verissimo CS (2021) Long-term culture, genetic manipulation and xenotransplantation of human normal and breast cancer organoids. Nat Protoc 16(4):1936–1965
Deltcheva E, Chylinski K, Sharma CM, Gonzales K, Chao Y, Pirzada ZA, Eckert MR, Vogel J, Charpentier E (2011) CRISPR RNA maturation by trans-encoded small RNA and host factor RNase III. Nature 471(7340):602–607
DeRose YS, Gligorich KM, Wang G, Georgelas A, Bowman P, Courdy SJ, Welm AL, Welm BE (2013) Patient-derived models of human breast cancer: protocols for in vitro and in vivo applications in tumor biology and translational medicine. Curr Protoc Pharmacol 60(1):14.23.11-14.23.43
Desmedt C, Haibe-Kains B, Wirapati P, Buyse M, Larsimont D, Bontempi G, Delorenzi M, Piccart M, Sotiriou C (2008) Biological processes associated with breast cancer clinical outcome depend on the molecular subtypes. Clin Cancer Res 14(16):5158–5165
Devarasetty M, Forsythe SD, Shelkey E, Soker S (2020) In vitro modeling of the tumor microenvironment in tumor organoids. Tissue Eng Regen Med 17(6):759
Diermeier SD, Chang K-C, Freier SM, Song J, El Demerdash O, Krasnitz A, Rigo F, Bennett CF, Spector DL (2016) Mammary tumor-associated RNAs impact tumor cell proliferation, invasion, and migration. Cell Rep 17(1):261–274
Dijkstra KK, Cattaneo CM, Weeber F, Chalabi M, van de Haar J, Fanchi LF, Slagter M, van der Velden DL, Kaing S, Kelderman S (2018) Generation of tumor-reactive T cells by co-culture of peripheral blood lymphocytes and tumor organoids. Cell 174(6):1586-1598. e1512
Djomehri SI, Burman B, Gonzalez ME, Takayama S, Kleer CG (2019) A reproducible scaffold-free 3D organoid model to study neoplastic progression in breast cancer. J Cell Commun Signal 13(1):129–143
Dobrolecki LE, Airhart SD, Alferez DG, Aparicio S, Behbod F, Bentires-Alj M, Brisken C, Bult CJ, Cai S, Clarke RB (2016) Patient-derived xenograft (PDX) models in basic and translational breast cancer research. Cancer Metastasis Rev 35(4):547–573
Driehuis E, Clevers H (2017) CRISPR/Cas 9 genome editing and its applications in organoids. Am J Physiol Gastrointest Liver Physiol 312(3):257–265
Drost J, Clevers H (2018) Organoids in cancer research. Nat Rev Cancer 18(7):407–418
Drost J, Van Boxtel R, Blokzijl F, Mizutani T, Sasaki N, Sasselli V, de Ligt J, Behjati S, Grolleman JE, van Wezel T (2017) Use of CRISPR-modified human stem cell organoids to study the origin of mutational signatures in cancer. Science 358(6360):234–238
Duarte AA, Gogola E, Sachs N, Barazas M, Annunziato S, de Ruiter JR, Velds A, Blatter S, Houthuijzen JM, Van De Ven M (2018) BRCA-deficient mouse mammary tumor organoids to study cancer-drug resistance. Nat Methods 15(2):134–140
Eliopoulos N, Al-Khaldi A, Crosato M, Lachapelle K, Galipeau J (2003) A neovascularized organoid derived from retrovirally engineered bone marrow stroma leads to prolonged in vivo systemic delivery of erythropoietin in nonmyeloablated, immunocompetent mice. Gene Ther 10(6):478–489
Eliopoulos N, Francois M, Boivin M-N, Martineau D, Galipeau J (2008) Neo-organoid of marrow mesenchymal stromal cells secreting interleukin-12 for breast cancer therapy. Can Res 68(12):4810–4818
Fiorini E, Veghini L, Corbo V (2020) Modeling cell communication in cancer with organoids: making the complex simple. Front Cell Dev Biol 8:166
Fujii M, Matano M, Toshimitsu K, Takano A, Mikami Y, Nishikori S, Sugimoto S, Sato T (2018) Human intestinal organoids maintain self-renewal capacity and cellular diversity in niche-inspired culture condition. Cell Stem Cell 23(6):787-793. e786
Furuta S, Bissell MJ (2016) Pathways involved in formation of mammary organoid architecture have keys to understanding drug resistance and to discovery of druggable targets. Cold Spring Harb Symp Quant Biol. https://doi.org/10.1101/sqb.2016.81.030825
Gao H, Korn JM, Ferretti S, Monahan JE, Wang Y, Singh M, Zhang C, Schnell C, Yang G, Zhang Y (2015) High-throughput screening using patient-derived tumor xenografts to predict clinical trial drug response. Nat Med 21(11):1318–1325
Gasiunas G, Barrangou R, Horvath P, Siksnys V (2012) Cas9–crRNA ribonucleoprotein complex mediates specific DNA cleavage for adaptive immunity in bacteria. Proc Natl Acad Sci 109(39):E2579–E2586
Giordano A, Galderisi U, Marino IR (2007) From the laboratory bench to the patient’s bedside: an update on clinical trials with mesenchymal stem cells. J Cell Physiol 211(1):27–35
Grün D, Lyubimova A, Kester L, Wiebrands K, Basak O, Sasaki N, Clevers H, Van Oudenaarden A (2015) Single-cell messenger RNA sequencing reveals rare intestinal cell types. Nature 525(7568):251–255
Guo W, Keckesova Z, Donaher JL, Shibue T, Tischler V, Reinhardt F, Itzkovitz S, Noske A, Zürrer-Härdi U, Bell G (2012) Slug and Sox9 cooperatively determine the mammary stem cell state. Cell 148(5):1015–1028
Haibe-Kains B, Desmedt C, Loi S, Culhane AC, Bontempi G, Quackenbush J, Sotiriou C (2012) A three-gene model to robustly identify breast cancer molecular subtypes. J Natl Cancer Inst 104(4):311–325
Harbeck N, Penault-Llorca F, Cortes J, Gnant M, Houssami N, Poortmans P, Ruddy K, Tsang J Cardoso F (2019) Breast cancer (Primer). Nat Rev Dis Primers 66
Horvath P, Aulner N, Bickle M, Davies AM, Del Nery E, Ebner D, Montoya MC, Östling P, Pietiäinen V, Price LS (2016) Screening out irrelevant cell-based models of disease. Nat Rev Drug Discovery 15(11):751–769
Howlett A, Bissell MJ (1990) Regulation of mammary epithelial cell function: a role for stromal and basement membrane matrices. Protoplasma 159(2–3):85–95
Huch M, Boj SF, Clevers H (2013) Lgr5+ liver stem cells, hepatic organoids and regenerative medicine. Regen Med 8(4):385–387
Hwang B, Lee JH, Bang D (2018) Single-cell RNA sequencing technologies and bioinformatics pipelines. Exp Mol Med 50(8):1–14
Ishino Y, Shinagawa H, Makino K, Amemura M, Nakata A (1987) Nucleotide sequence of the iap gene, responsible for alkaline phosphatase isozyme conversion in Escherichia coli, and identification of the gene product. J Bacteriol 169(12):5429–5433
Jansen R, van Embden JD, Gaastra W, Schouls LM (2002) Identification of genes that are associated with DNA repeats in prokaryotes. Mol Microbiol 43(6):1565–1575
Jardé T, Lloyd-Lewis B, Thomas M, Kendrick H, Melchor L, Bougaret L, Watson PD, Ewan K, Smalley MJ, Dale TC (2016) Wnt and Neuregulin1/ErbB signalling extends 3D culture of hormone responsive mammary organoids. Nat Commun 7(1):1–14
Jiang C, Meng L, Yang B, Luo X (2020) Application of CRISPR/Cas9 gene editing technique in the study of cancer treatment. Clin Genet 97(1):73–88
Jinek M, Chylinski K, Fonfara I, Hauer M, Doudna J, Charpentier E (2012) A programmable dual-RNA-guided DNA endonuclease in adaptive bacterial immunity. Science 337:816–821
Jonkers J, Meuwissen R, van der Gulden H, Peterse H, van der Valk M, Berns A (2001) Synergistic tumor suppressor activity of BRCA2 and p53 in a conditional mouse model for breast cancer. Nat Genet 29(4):418–425
Kabos P, Haughian JM, Wang X, Dye WW, Finlayson C, Elias A, Horwitz KB, Sartorius CA (2011) Cytokeratin 5 positive cells represent a steroid receptor negative and therapy resistant subpopulation in luminal breast cancers. Breast Cancer Res Treat 128(1):45–55
Kamb A (2005) What’s wrong with our cancer models? Nat Rev Drug Discov 4(2):161–165
Kapałczyńska M, Kolenda T, Przybyła W, Zajączkowska M, Teresiak A, Filas V, Ibbs M, Bliźniak R, Łuczewski Ł, Lamperska K (2018) 2D and 3D cell cultures–a comparison of different types of cancer cell cultures. Archives Med Sci AMS 14(4):910
Karthaus WR, Iaquinta PJ, Drost J, Gracanin A, Van Boxtel R, Wongvipat J, Dowling CM, Gao D, Begthel H, Sachs N (2014) Identification of multipotent luminal progenitor cells in human prostate organoid cultures. Cell 159(1):163–175
Kenny PA, Lee GY, Myers CA, Neve RM, Semeiks JR, Spellman PT, Lorenz K, Lee EH, Barcellos-Hoff MH, Petersen OW (2007) The morphologies of breast cancer cell lines in three-dimensional assays correlate with their profiles of gene expression. Mol Oncol 1(1):84–96
Kim HN, Habbit NL, Su CY, Choi N, Ahn EH, Lipke EA, Kim DH (2019) Microphysiological systems as enabling tools for modeling complexity in the tumor microenvironment and accelerating cancer drug development. Adv Func Mater 29(22):1807553
Korhonen T, Kuukasjärvi T, Huhtala H, Alarmo E-L, Holli K, Kallioniemi A, Pylkkänen L (2013) The impact of lobular and ductal breast cancer histology on the metastatic behavior and long term survival of breast cancer patients. Breast 22(6):1119–1124
Lancaster MA, Knoblich JA (2014) Organogenesis in a dish: modeling development and disease using organoid technologies. Science. https://doi.org/10.1126/science.1247125
Lee G, Kenny P, Lee EH, Bissell MJ (2007) Three-dimensional culture models of normal and malignant breast epithelial cells. Nat Methods 4:359–365
Li L, Knutsdottir H, Hui K, Weiss MJ, He J, Philosophe B, Cameron AM, Wolfgang CL, Pawlik TM, Ghiaur G (2019) Human primary liver cancer organoids reveal intratumor and interpatient drug response heterogeneity. JCI Insight. https://doi.org/10.1172/jci.insight.121490
Li X, Larsson P, Ljuslinder I, Öhlund D, Myte R, Löfgren-Burström A, Zingmark C, Ling A, Edin S, Palmqvist R (2020) Ex vivo organoid cultures reveal the importance of the tumor microenvironment for maintenance of colorectal cancer stem cells. Cancers 12(4):923
Linnemann JR, Miura H, Meixner LK, Irmler M, Kloos UJ, Hirschi B, Bartsch HS, Sass S, Beckers J, Theis FJ (2015) Quantification of regenerative potential in primary human mammary epithelial cells. Development 142(18):3239–3251
Liu X, Holstege H, van der Gulden H, Treur-Mulder M, Zevenhoven J, Velds A, Kerkhoven RM, van Vliet MH, Wessels LF, Peterse JL (2007) Somatic loss of BRCA1 and p53 in mice induces mammary tumors with features of human BRCA1-mutated basal-like breast cancer. Proc Natl Acad Sci 104(29):12111–12116
Liu C, Qin T, Huang Y, Li Y, Chen G, Sun C (2020) Drug screening model meets cancer organoid technology. Transl Oncol 13(11):100840
Nagle PW, Plukker JTM, Muijs CT, van Luijk P, Coppes RP (2018) Patient-derived tumor organoids for prediction of cancer treatment response. Semin Cancer Biol 53:258–264. https://doi.org/10.1016/j.semcancer.2018.06.005
Nakata A, Amemura M, Makino K (1989) Unusual nucleotide arrangement with repeated sequences in the Escherichia coli K-12 chromosome. J Bacteriol 171(6):3553–3556
Nolan E, Vaillant F, Branstetter D, Pal B, Giner G, Whitehead L, Lok S, Mann G, Rohrbach K, Huang L (2016) Cuningham Foundation Consortium for Research into Familial Breast Cancer (kConFab) RANK ligand as a potential target for breast cancer prevention in BRCA1-mutation carriers. Nat Med 22:933–939
Osborne CK, Hobbs K, Trent JM (1987) Biological differences among MCF-7 human breast cancer cell lines from different laboratories. Breast Cancer Res Treat 9(2):111–121
Parker JS, Mullins M, Cheang MC, Leung S, Voduc D, Vickery T, Davies S, Fauron C, He X, Hu Z (2009) Supervised risk predictor of breast cancer based on intrinsic subtypes. J Clin Oncol 27(8):1160
Pauli C, Hopkins BD, Prandi D, Shaw R, Fedrizzi T, Sboner A, Sailer V, Augello M, Puca L, Rosati R (2017) Personalized in vitro and in vivo cancer models to guide precision medicine. Cancer Discov 7(5):462–477
Peng WC, Logan CY, Fish M, Anbarchian T, Aguisanda F, Álvarez-Varela A, Wu P, Jin Y, Zhu J, Li B (2018) Inflammatory cytokine TNFα promotes the long-term expansion of primary hepatocytes in 3D culture. Cell 175(6):1607-1619. e1615
Qu Y, Han B, Gao B, Bose S, Gong Y, Wawrowsky K, Giuliano AE, Sareen D, Cui X (2017) Differentiation of human induced pluripotent stem cells to mammary-like organoids. Stem Cell Rep 8(2):205–215
Roerink SF, Sasaki N, Lee-Six H, Young MD, Alexandrov LB, Behjati S, Mitchell TJ, Grossmann S, Lightfoot H, Egan DA (2018) Intra-tumour diversification in colorectal cancer at the single-cell level. Nature 556(7702):457–462
Roper J, Yilmaz ÖH (2019) Breakthrough moments: genome editing and organoids. Cell Stem Cell 24(6):841–842
Rosenbluth JM, Schackmann RC, Gray GK, Selfors LM, Li CM-C, Boedicker M, Kuiken HJ, Richardson A, Brock J, Garber J (2020) Organoid cultures from normal and cancer-prone human breast tissues preserve complex epithelial lineages. Nat Commun 11(1):1–14
Rottenberg S, Borst P (2012) Drug resistance in the mouse cancer clinic. Drug Resist Updates 15(1–2):81–89
Sachs N, de Ligt J, Kopper O, Gogola E, Bounova G, Weeber F, Balgobind AV, Wind K, Gracanin A, Begthel H (2018) A living biobank of breast cancer organoids captures disease heterogeneity. Cell 172(1–2):373–386310
Sasmita A, Wong Y (2018) Organoids as reliable breast cancer study models: an update. Int J Oncol Res 1:008
Sato T, Clevers H (2015) SnapShot: growing organoids from stem cells. Cell 161(7):1700-1700. e1701
Sato T, Vries RG, Snippert HJ, Van De Wetering M, Barker N, Stange DE, Van Es JH, Abo A, Kujala P, Peters PJ (2009) Single Lgr5 stem cells build crypt-villus structures in vitro without a mesenchymal niche. Nature 459(7244):262–265
Sato T, Stange DE, Ferrante M, Vries RG, Van Es JH, Van Den Brink S, Van Houdt WJ, Pronk A, Van Gorp J, Siersema PD (2011) Long-term expansion of epithelial organoids from human colon, adenoma, adenocarcinoma, and Barrett’s epithelium. Gastroenterology 141(5):1762–1772
Schmidt M, Thomssen C, Untch M (2016) Intrinsic subtypes of primary breast cancer-gene expression analysis. Oncol Res Treat 39(3):102–110
Schwank G, Koo B-K, Sasselli V, Dekkers JF, Heo I, Demircan T, Sasaki N, Boymans S, Cuppen E, van der Ent CK (2013) Functional repair of CFTR by CRISPR/Cas9 in intestinal stem cell organoids of cystic fibrosis patients. Cell Stem Cell 13(6):653–658
Simian M, Bissell MJ (2017) Organoids: a historical perspective of thinking in three dimensions. J Cell Biol 216(1):31–40
Skardal A, Shupe T, Atala A (2016) Organoid-on-a-chip and body-on-a-chip systems for drug screening and disease modeling. Drug Discov Today 21(9):1399–1411
Sloan SA, Darmanis S, Huber N, Khan TA, Birey F, Caneda C, Reimer R, Quake SR, Barres BA, Paşca SP (2017) Human astrocyte maturation captured in 3D cerebral cortical spheroids derived from pluripotent stem cells. Neuron 95(4):779-790. e776
Sopik V (2021) International variation in breast cancer incidence and mortality in young women. Breast Cancer Res Treat 186(2):497–507
Soysal SD, Tzankov A, Muenst SE (2015) Role of the tumor microenvironment in breast cancer. Pathobiology 82(3–4):142–152
Srivastava V, Huycke TR, Phong KT, Gartner ZJ (2020) Organoid models for mammary gland dynamics and breast cancer. Curr Opin Cell Biol 66:51–58
Stagg J, Lejeune L, Paquin A, Galipeau J (2004) Marrow stromal cells for interleukin-2 delivery in cancer immunotherapy. Hum Gene Ther 15(6):597–608
Tuveson D, Clevers H (2019) Cancer modeling meets human organoid technology. Science 364(6444):952–955
van de Wetering M, Francies HE, Francis JM, Bounova G, Iorio F, Pronk A, van Houdt W, van Gorp J, Taylor-Weiner A, Kester L (2015) Prospective derivation of a living organoid biobank of colorectal cancer patients. Cell 161(4):933–945
Vlachogiannis G, Hedayat S, Vatsiou A, Jamin Y, Fernández-Mateos J, Khan K, Lampis A, Eason K, Huntingford I, Burke R (2018) Patient-derived organoids model treatment response of metastatic gastrointestinal cancers. Science 359(6378):920–926
Walsh AJ, Cook RS, Sanders ME, Aurisicchio L, Ciliberto G, Arteaga CL, Skala MC (2014) Quantitative optical imaging of primary tumor organoid metabolism predicts drug response in breast cancer. Can Res 74(18):5184–5194
Wang M, Yao LC, Cheng M, Cai D, Martinek J, Pan CX, Shi W, Ma AH, de Vere White RW, Airhart S (2018) Humanized mice in studying efficacy and mechanisms of PD-1-targeted cancer immunotherapy. FASEB J 32(3):1537–1549
Weigelt B, Ghajar CM, Bissell MJ (2014) The need for complex 3D culture models to unravel novel pathways and identify accurate biomarkers in breast cancer. Adv Drug Deliv Rev 69:42–51
Xiang Y, Tanaka Y, Patterson B, Kang Y-J, Govindaiah G, Roselaar N, Cakir B, Kim K-Y, Lombroso AP, Hwang S-M (2017) Fusion of regionally specified hPSC-derived organoids models human brain development and interneuron migration. Cell Stem Cell 21(3):383-398. e387
Yang L, Liu B, Chen H, Gao R, Huang K, Guo Q, Li F, Chen W, He J (2020) Progress in the application of organoids to breast cancer research. J Cell Mol Med 24(10):5420–5427
Yin Y, Liu P-Y, Shi Y, Li P (2021) Single-cell sequencing and organoids: a powerful combination for modelling organ development and diseases. Rev Physiol Biochem Pharmacol 179(179):189–210
Yu J, Huang W (2020) The progress and clinical application of breast cancer organoids. Int J Stem Cells 13(3):295
Yung-mae MY, Donoho GP, Iversen PW, Zhang Y, Van Horn RD, Forest A, Novosiadly RD, Webster YW, Ebert P, Bray S (2017) Mouse PDX trial suggests synergy of concurrent inhibition of RAF and EGFR in colorectal cancer with BRAF or KRAS mutations. Clin Cancer Res 23(18):5547–5560
Zepp JA, Zacharias WJ, Frank DB, Cavanaugh CA, Zhou S, Morley MP, Morrisey EE (2017) Distinct mesenchymal lineages and niches promote epithelial self-renewal and myofibrogenesis in the lung. Cell 170(6):1134-1148. e1110
Zhang Z, Christin JR, Wang C, Ge K, Oktay MH, Guo W (2016) Mammary-stem-cell-based somatic mouse models reveal breast cancer drivers causing cell fate dysregulation. Cell Rep 16(12):3146–3156
Acknowledgements
This work was funded by the National Institute of Allergy and Infectious Diseases (Grant no. R01AI050875).
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
NE and SS designed the review paper and wrote the manuscript, ANE, AM, MG, and SA contributed to writing the manuscript, MRH reviewed and revised the final version of manuscript and supervised the study.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ebrahimi, N., Nasr Esfahani, A., Samizade, S. et al. The potential application of organoids in breast cancer research and treatment. Hum Genet 141, 193–208 (2022). https://doi.org/10.1007/s00439-021-02390-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00439-021-02390-0