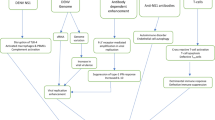
Abstract
Human rhinoviruses (RV) are a frequent cause of respiratory tract infections with substantial morbidity and mortality in some patients. Nevertheless, the genetic basis of susceptibility to RV in humans has been relatively understudied. Experimental infections of mice and in vitro infections of human cells have indicated that various pathogen recognition receptors (TLRs, RIG-I, and MDA5) regulate innate immune responses to RV. However, deficiency of MDA5 is the only one among these so far uncovered that confers RV susceptibility in humans. Other work has shown increased RV susceptibility in patients with a polymorphism in CDHR3 that encodes the cellular receptor for RV-C entry. Here, we provide a comprehensive review of the genetic determinants of human RV susceptibility in the context of what is known about RV biology.

Similar content being viewed by others
References
Alexopoulou L, Holt AC, Medzhitov R, Flavell RA (2001) Recognition of double-stranded RNA and activation of NF-kappaB by Toll-like receptor 3. Nature 413:732–738. https://doi.org/10.1038/35099560
Asgari S, Schlapbach LJ, Anchisi S, Hammer C, Bartha I, Junier T, Mottet-Osman G, Posfay-Barbe KM, Longchamp D, Stocker M, Cordey S, Kaiser L, Riedel T, Kenna T, Long D, Schibler A, Telenti A, Tapparel C, McLaren PJ, Garcin D, Fellay J (2017) Severe viral respiratory infections in children with IFIH1 loss-of-function mutations. Proc Natl Acad Sci USA 114:8342–8347. https://doi.org/10.1073/pnas.1704259114
Bartlett NW, Walton RP, Edwards MR, Aniscenko J, Caramori G, Zhu J, Glanville N, Choy KJ, Jourdan P, Burnet J, Tuthill TJ, Pedrick MS, Hurle MJ, Plumpton C, Sharp NA, Bussell JN, Swallow DM, Schwarze J, Guy B, Almond JW, Jeffery PK, Lloyd CM, Papi A, Killington RA, Rowlands DJ, Blair ED, Clarke NJ, Johnston SL (2008) Mouse models of rhinovirus-induced disease and exacerbation of allergic airway inflammation. Nat Med 14:199–204. https://doi.org/10.1038/nm1713
Basnet S, Bochkov YA, Brockman-Schneider RA, Kuipers I, Aesif SW, Jackson DJ, Lemanske RF Jr, Ober C, Palmenberg AC, Gern JE (2019a) CDHR3 Asthma-risk genotype affects susceptibility of airway epithelium to rhinovirus C infections. Am J Respir Cell Mol Biol 61:450–458. https://doi.org/10.1165/rcmb.2018-0220OC
Basnet S, Palmenberg AC, Gern JE (2019b) Rhinoviruses and their receptors. Chest 155:1018–1025. https://doi.org/10.1016/j.chest.2018.12.012
Becker TM, Durrani SR, Bochkov YA, Devries MK, Rajamanickam V, Jackson DJ (2013) Effect of exogenous interferons on rhinovirus replication and airway inflammatory responses. Ann Allergy Asthma Immunol 111:397–401. https://doi.org/10.1016/j.anai.2013.07.029
Bentley JK, Han M, Jaipalli S, Hinde JL, Lei J, Ishikawa T, Goldsmith AM, Rajput C, Hershenson MB (2019) Myristoylated rhinovirus VP4 protein activates TLR2-dependent proinflammatory gene expression. Am J Physiol Lung Cell Mol Physiol 317:L57–L70. https://doi.org/10.1152/ajplung.00365.2018
Bieback K, Lien E, Klagge IM, Avota E, Schneider-Schaulies J, Duprex WP, Wagner H, Kirschning CJ, Ter Meulen V, Schneider-Schaulies S (2002) Hemagglutinin protein of wild-type measles virus activates toll-like receptor 2 signaling. J Virol 76:8729–8736. https://doi.org/10.1128/jvi.76.17.8729-8736.2002
Bochkov YA, Gern JE (2016) Rhinoviruses and their receptors: implications for allergic disease. Curr Allergy Asthma Rep 16:30. https://doi.org/10.1007/s11882-016-0608-7
Bochkov YA, Watters K, Ashraf S, Griggs TF, Devries MK, Jackson DJ, Palmenberg AC, Gern JE (2015) Cadherin-related family member 3, a childhood asthma susceptibility gene product, mediates rhinovirus C binding and replication. Proc Natl Acad Sci USA 112:5485–5490. https://doi.org/10.1073/pnas.1421178112
Boehme KW, Guerrero M, Compton T (2006) Human cytomegalovirus envelope glycoproteins B and H are necessary for TLR2 activation in permissive cells. J Immunol 177:7094–7102. https://doi.org/10.4049/jimmunol.177.10.7094
Bonnelykke K, Sleiman P, Nielsen K, Kreiner-Moller E, Mercader JM, Belgrave D, den Dekker HT, Husby A, Sevelsted A, Faura-Tellez G, Mortensen LJ, Paternoster L, Flaaten R, Molgaard A, Smart DE, Thomsen PF, Rasmussen MA, Bonas-Guarch S, Holst C, Nohr EA, Yadav R, March ME, Blicher T, Lackie PM, Jaddoe VW, Simpson A, Holloway JW, Duijts L, Custovic A, Davies DE, Torrents D, Gupta R, Hollegaard MV, Hougaard DM, Hakonarson H, Bisgaard H (2014) A genome-wide association study identifies CDHR3 as a susceptibility locus for early childhood asthma with severe exacerbations. Nat Genet 46:51–55. https://doi.org/10.1038/ng.2830
Bonnelykke K, Coleman AT, Evans MD, Thorsen J, Waage J, Vissing NH, Carlsson CJ, Stokholm J, Chawes BL, Jessen LE, Fischer TK, Bochkov YA, Ober C, Lemanske RF Jr, Jackson DJ, Gern JE, Bisgaard H (2018) Cadherin-related family member 3 genetics and rhinovirus C respiratory illnesses. Am J Respir Crit Care Med 197:589–594. https://doi.org/10.1164/rccm.201705-1021OC
Casrouge A, Zhang SY, Eidenschenk C, Jouanguy E, Puel A, Yang K, Alcais A, Picard C, Mahfoufi N, Nicolas N, Lorenzo L, Plancoulaine S, Senechal B, Geissmann F, Tabeta K, Hoebe K, Du X, Miller RL, Heron B, Mignot C, de Villemeur TB, Lebon P, Dulac O, Rozenberg F, Beutler B, Tardieu M, Abel L, Casanova JL (2006) Herpes simplex virus encephalitis in human UNC-93B deficiency. Science 314:308–312. https://doi.org/10.1126/science.1128346
Cervantes JL, Weinerman B, Basole C, Salazar JC (2012) TLR8: the forgotten relative revindicated. Cell Mol Immunol 9:434–438. https://doi.org/10.1038/cmi.2012.38
Chow KT, Gale M Jr, Loo YM (2018) RIG-I and Other RNA sensors in antiviral immunity. Annu Rev Immunol 36:667–694. https://doi.org/10.1146/annurev-immunol-042617-053309
Cooper DN, Krawczak M, Polychronakos C, Tyler-Smith C, Kehrer-Sawatzki H (2013) Where genotype is not predictive of phenotype: towards an understanding of the molecular basis of reduced penetrance in human inherited disease. Hum Genet 132:1077–1130. https://doi.org/10.1007/s00439-013-1331-2
Diebold SS, Kaisho T, Hemmi H, Akira S, Reisesousa C (2004) Innate antiviral responses by means of TLR7-mediated recognition of single-stranded RNA. Science 303:1529–1531. https://doi.org/10.1126/science.1093616
Dittmann M, Hoffmann HH, Scull MA, Gilmore RH, Bell KL, Ciancanelli M, Wilson SJ, Crotta S, Yu Y, Flatley B, Xiao JW, Casanova JL, Wack A, Bieniasz PD, Rice CM (2015) A serpin shapes the extracellular environment to prevent influenza A virus maturation. Cell 160:631–643. https://doi.org/10.1016/j.cell.2015.01.040
Everman JL, Sajuthi S, Saef B, Rios C, Stoner AM, Numata M, Hu D, Eng C, Oh S, Rodriguez-Santana J, Vladar EK, Voelker DR, Burchard EG, Seibold MA (2019) Functional genomics of CDHR3 confirms its role in HRV-C infection and childhood asthma exacerbations. J Allergy Clin Immunol 144:962–971. https://doi.org/10.1016/j.jaci.2019.01.052
Fendrick AM, Monto AS, Nightengale B, Sarnes M (2003) The economic burden of non-influenza-related viral respiratory tract infection in the United States. Arch Intern Med 163:487–494. https://doi.org/10.1001/archinte.163.4.487
Foxman EF, Storer JA, Fitzgerald ME, Wasik BR, Hou L, Zhao H, Turner PE, Pyle AM, Iwasaki A (2015) Temperature-dependent innate defense against the common cold virus limits viral replication at warm temperature in mouse airway cells. Proc Natl Acad Sci USA 112:827–832. https://doi.org/10.1073/pnas.1411030112
Gern JE, Busse WW (1999) Association of rhinovirus infections with asthma. Clin Microbiol Rev 12:9–18
Global Burden of Disease Study C (2015) Global, regional, and national incidence, prevalence, and years lived with disability for 301 acute and chronic diseases and injuries in 188 countries, 1990–2013: a systematic analysis for the Global Burden of Disease Study 2013. Lancet 386:743–800. https://doi.org/10.1016/S0140-6736(15)60692-4
Goubau D, Schlee M, Deddouche S, Pruijssers AJ, Zillinger T, Goldeck M, Schuberth C, Van der Veen AG, Fujimura T, Rehwinkel J, Iskarpatyoti JA, Barchet W, Ludwig J, Dermody TS, Hartmann G, Reis e Sousa C (2014) Antiviral immunity via RIG-I-mediated recognition of RNA bearing 5'-diphosphates. Nature 514:372–375. https://doi.org/10.1038/nature13590
Greve JM, Davis G, Meyer AM, Forte CP, Yost SC, Marlor CW, Kamarck ME, McClelland A (1989) The major human rhinovirus receptor is ICAM-1. Cell 56:839–847. https://doi.org/10.1016/0092-8674(89)90688-0
Gudbjartsson DF, Helgason H, Gudjonsson SA, Zink F, Oddson A, Gylfason A, Besenbacher S, Magnusson G, Halldorsson BV, Hjartarson E, Sigurdsson GT, Stacey SN, Frigge ML, Holm H, Saemundsdottir J, Helgadottir HT, Johannsdottir H, Sigfusson G, Thorgeirsson G, Sverrisson JT, Gretarsdottir S, Walters GB, Rafnar T, Thjodleifsson B, Bjornsson ES, Olafsson S, Thorarinsdottir H, Steingrimsdottir T, Gudmundsdottir TS, Theodors A, Jonasson JG, Sigurdsson A, Bjornsdottir G, Jonsson JJ, Thorarensen O, Ludvigsson P, Gudbjartsson H, Eyjolfsson GI, Sigurdardottir O, Olafsson I, Arnar DO, Magnusson OT, Kong A, Masson G, Thorsteinsdottir U, Helgason A, Sulem P, Stefansson K (2015) Large-scale whole-genome sequencing of the Icelandic population. Nat Genet 47:435–444. https://doi.org/10.1038/ng.3247
Guo Y, Audry M, Ciancanelli M, Alsina L, Azevedo J, Herman M, Anguiano E, Sancho-Shimizu V, Lorenzo L, Pauwels E, Philippe PB, Perez de Diego R, Cardon A, Vogt G, Picard C, Andrianirina ZZ, Rozenberg F, Lebon P, Plancoulaine S, Tardieu M, Valerie D, Jouanguy E, Chaussabel D, Geissmann F, Abel L, Casanova JL, Zhang SY (2011) Herpes simplex virus encephalitis in a patient with complete TLR3 deficiency: TLR3 is otherwise redundant in protective immunity. J Exp Med 208:2083–2098. https://doi.org/10.1084/jem.20101568
Han M, Chung Y, Young Hong J, Rajput C, Lei J, Hinde JL, Chen Q, Weng SP, Bentley JK, Hershenson MB (2016) Toll-like receptor 2-expressing macrophages are required and sufficient for rhinovirus-induced airway inflammation. J Allergy Clin Immunol 138:1619–1630. https://doi.org/10.1016/j.jaci.2016.01.037
Hatchwell L, Collison A, Girkin J, Parsons K, Li J, Zhang J, Phipps S, Knight D, Bartlett NW, Johnston SL, Foster PS, Wark PA, Mattes J (2015) Toll-like receptor 7 governs interferon and inflammatory responses to rhinovirus and is suppressed by IL-5-induced lung eosinophilia. Thorax 70:854–861. https://doi.org/10.1136/thoraxjnl-2014-205465
Hayden FG (2004) Rhinovirus and the lower respiratory tract. Rev Med Virol 14:17–31. https://doi.org/10.1002/rmv.406
Heil F, Hemmi H, Hochrein H, Ampenberger F, Kirschning C, Akira S, Lipford G, Wagner H, Bauer S (2004) Species-specific recognition of single-stranded RNA via toll-like receptor 7 and 8. Science 303:1526–1529. https://doi.org/10.1126/science.1093620
Henrick BM, Yao XD, Taha AY, German JB, Rosenthal KL (2016) Insights into soluble toll-like receptor 2 as a downregulator of virally induced inflammation. Front Immunol 7:291. https://doi.org/10.3389/fimmu.2016.00291
Herman M, Ciancanelli M, Ou YH, Lorenzo L, Klaudel-Dreszler M, Pauwels E, Sancho-Shimizu V, Perez de Diego R, Abhyankar A, Israelsson E, Guo Y, Cardon A, Rozenberg F, Lebon P, Tardieu M, Heropolitanska-Pliszka E, Chaussabel D, White MA, Abel L, Zhang SY, Casanova JL (2012) Heterozygous TBK1 mutations impair TLR3 immunity and underlie herpes simplex encephalitis of childhood. J Exp Med 209:1567–1582. https://doi.org/10.1084/jem.20111316
Hewson CA, Jardine A, Edwards MR, Laza-Stanca V, Johnston SL (2005) Toll-like receptor 3 is induced by and mediates antiviral activity against rhinovirus infection of human bronchial epithelial cells. J Virol 79:12273–12279. https://doi.org/10.1128/JVI.79.19.12273-12279.2005
Hofer F, Gruenberger M, Kowalski H, Machat H, Huettinger M, Kuechler E, Blaas D (1994) Members of the low density lipoprotein receptor family mediate cell entry of a minor-group common cold virus. Proc Natl Acad Sci USA 91:1839–1842. https://doi.org/10.1073/pnas.91.5.1839
Hornung V, Ellegast J, Kim S, Brzozka K, Jung A, Kato H, Poeck H, Akira S, Conzelmann KK, Schlee M, Endres S, Hartmann G (2006) 5'-Triphosphate RNA is the ligand for RIG-I. Science 314:994–997. https://doi.org/10.1126/science.1132505
Hou F, Sun L, Zheng H, Skaug B, Jiang QX, Chen ZJ (2011) MAVS forms functional prion-like aggregates to activate and propagate antiviral innate immune response. Cell 146:448–461. https://doi.org/10.1016/j.cell.2011.06.041
Hu J, Nistal-Villan E, Voho A, Ganee A, Kumar M, Ding Y, Garcia-Sastre A, Wetmur JG (2010) A common polymorphism in the caspase recruitment domain of RIG-I modifies the innate immune response of human dendritic cells. J Immunol 185:424–432. https://doi.org/10.4049/jimmunol.0903291
Israel L, Wang Y, Bulek K, Della Mina E, Zhang Z, Pedergnana V, Chrabieh M, Lemmens NA, Sancho-Shimizu V, Descatoire M, Lasseau T, Israelsson E, Lorenzo L, Yun L, Belkadi A, Moran A, Weisman LE, Vandenesch F, Batteux F, Weller S, Levin M, Herberg J, Abhyankar A, Prando C, Itan Y, van Wamel WJB, Picard C, Abel L, Chaussabel D, Li X, Beutler B, Arkwright PD, Casanova JL, Puel A (2017) Human adaptive immunity rescues an inborn error of innate immunity. Cell 168(789–800):e10. https://doi.org/10.1016/j.cell.2017.01.039
Jacobs SE, Lamson DM, St George K, Walsh TJ (2013) Human rhinoviruses. Clin Microbiol Rev 26:135–162. https://doi.org/10.1128/CMR.00077-12
Jankauskaite L, Miseviciene V, Vaideliene L, Kevalas R (2018) Lower airway virology in health and disease-from invaders to symbionts. Medicina (Kaunas). https://doi.org/10.3390/medicina54050072
Johnston SL, Pattemore PK, Sanderson G, Smith S, Lampe F, Josephs L, Symington P, O'Toole S, Myint SH, Tyrrell DA et al (1995) Community study of role of viral infections in exacerbations of asthma in 9–11 year old children. BMJ 310:1225–1229. https://doi.org/10.1136/bmj.310.6989.1225
Jorgensen SE, Christiansen M, Ryo LB, Gad HH, Gjedsted J, Staeheli P, Mikkelsen JG, Storgaard M, Hartmann R, Mogensen TH (2018) Defective RNA sensing by RIG-I in severe influenza virus infection. Clin Exp Immunol 192:366–376. https://doi.org/10.1111/cei.13120
Kato H, Takeuchi O, Sato S, Yoneyama M, Yamamoto M, Matsui K, Uematsu S, Jung A, Kawai T, Ishii KJ, Yamaguchi O, Otsu K, Tsujimura T, Koh CS, Reisesousa C, Matsuura Y, Fujita T, Akira S (2006) Differential roles of MDA5 and RIG-I helicases in the recognition of RNA viruses. Nature 441:101–105. https://doi.org/10.1038/nature04734
Kato H, Takeuchi O, Mikamo-Satoh E, Hirai R, Kawai T, Matsushita K, Hiiragi A, Dermody TS, Fujita T, Akira S (2008) Length-dependent recognition of double-stranded ribonucleic acids by retinoic acid-inducible gene-I and melanoma differentiation-associated gene 5. J Exp Med 205:1601–1610. https://doi.org/10.1084/jem.20080091
Kell AM, Gale M Jr (2015) RIG-I in RNA virus recognition. Virology 479–480:110–121. https://doi.org/10.1016/j.virol.2015.02.017
Lamborn IT, Jing H, Zhang Y, Drutman SB, Abbott JK, Munir S, Bade S, Murdock HM, Santos CP, Brock LG, Masutani E, Fordjour EY, McElwee JJ, Hughes JD, Nichols DP, Belkadi A, Oler AJ, Happel CS, Matthews HF, Abel L, Collins PL, Subbarao K, Gelfand EW, Ciancanelli MJ, Casanova JL, Su HC (2017) Recurrent rhinovirus infections in a child with inherited MDA5 deficiency. J Exp Med 214:1949–1972. https://doi.org/10.1084/jem.20161759
Lim HK, Huang SXL, Chen J, Kerner G, Gilliaux O, Bastard P, Dobbs K, Hernandez N, Goudin N, Hasek ML, Garcia Reino EJ, Lafaille FG, Lorenzo L, Luthra P, Kochetkov T, Bigio B, Boucherit S, Rozenberg F, Vedrinne C, Keller MD, Itan Y, Garcia-Sastre A, Celard M, Orange JS, Ciancanelli MJ, Meyts I, Zhang Q, Abel L, Notarangelo LD, Snoeck HW, Casanova JL, Zhang SY (2019) Severe influenza pneumonitis in children with inherited TLR3 deficiency. J Exp Med. https://doi.org/10.1084/jem.20181621
Loisel DA, Du G, Ahluwalia TS, Tisler CJ, Evans MD, Myers RA, Gangnon RE, Kreiner-Moller E, Bonnelykke K, Bisgaard H, Jackson DJ, Lemanske RF Jr, Nicolae DL, Gern JE, Ober C (2016) Genetic associations with viral respiratory illnesses and asthma control in children. Clin Exp Allergy 46:112–124. https://doi.org/10.1111/cea.12642
Loo YM, Gale M Jr (2011) Immune signaling by RIG-I-like receptors. Immunity 34:680–692. https://doi.org/10.1016/j.immuni.2011.05.003
Loo YM, Fornek J, Crochet N, Bajwa G, Perwitasari O, Martinez-Sobrido L, Akira S, Gill MA, Garcia-Sastre A, Katze MG, Gale M Jr (2008) Distinct RIG-I and MDA5 signaling by RNA viruses in innate immunity. J Virol 82:335–345. https://doi.org/10.1128/JVI.01080-07
Lund JM, Alexopoulou L, Sato A, Karow M, Adams NC, Gale NW, Iwasaki A, Flavell RA (2004) Recognition of single-stranded RNA viruses by Toll-like receptor 7. Proc Natl Acad Sci USA 101:5598–5603. https://doi.org/10.1073/pnas.0400937101
Maglione PJ, Simchoni N, Cunningham-Rundles C (2015) Toll-like receptor signaling in primary immune deficiencies. Ann N Y Acad Sci 1356:1–21. https://doi.org/10.1111/nyas.12763
Makela SM, Osterlund P, Westenius V, Latvala S, Diamond MS, Gale M Jr, Julkunen I (2015) RIG-I signaling is essential for influenza B virus-induced rapid interferon gene expression. J Virol 89:12014–12025. https://doi.org/10.1128/JVI.01576-15
Myatt TA, Johnston SL, Zuo Z, Wand M, Kebadze T, Rudnick S, Milton DK (2004) Detection of airborne rhinovirus and its relation to outdoor air supply in office environments. Am J Respir Crit Care Med 169:1187–1190. https://doi.org/10.1164/rccm.200306-760OC
Nurani G, Lindqvist B, Casasnovas JM (2003) Receptor priming of major group human rhinoviruses for uncoating and entry at mild low-pH environments. J Virol 77:11985–11991. https://doi.org/10.1128/jvi.77.22.11985-11991.2003
Palmenberg AC (2017) Rhinovirus C, asthma, and cell surface expression of virus receptor CDHR3. J Virol. https://doi.org/10.1128/JVI.00072-17
Palmenberg AC, Spiro D, Kuzmickas R, Wang S, Djikeng A, Rathe JA, Fraser-Liggett CM, Liggett SB (2009) Sequencing and analyses of all known human rhinovirus genomes reveal structure and evolution. Science 324:55–59. https://doi.org/10.1126/science.1165557
Papadopoulos NG, Bates PJ, Bardin PG, Papi A, Leir SH, Fraenkel DJ, Meyer J, Lackie PM, Sanderson G, Holgate ST, Johnston SL (2000) Rhinoviruses infect the lower airways. J Infect Dis 181:1875–1884. https://doi.org/10.1086/315513
Perez de Diego R, Sancho-Shimizu V, Lorenzo L, Puel A, Plancoulaine S, Picard C, Herman M, Cardon A, Durandy A, Bustamante J, Vallabhapurapu S, Bravo J, Warnatz K, Chaix Y, Cascarrigny F, Lebon P, Rozenberg F, Karin M, Tardieu M, Al-Muhsen S, Jouanguy E, Zhang SY, Abel L, Casanova JL (2010) Human TRAF3 adaptor molecule deficiency leads to impaired Toll-like receptor 3 response and susceptibility to herpes simplex encephalitis. Immunity 33:400–411. https://doi.org/10.1016/j.immuni.2010.08.014
Picard C, Puel A, Bonnet M, Ku CL, Bustamante J, Yang K, Soudais C, Dupuis S, Feinberg J, Fieschi C, Elbim C, Hitchcock R, Lammas D, Davies G, Al-Ghonaium A, Al-Rayes H, Al-Jumaah S, Al-Hajjar S, Al-Mohsen IZ, Frayha HH, Rucker R, Hawn TR, Aderem A, Tufenkeji H, Haraguchi S, Day NK, Good RA, Gougerot-Pocidalo MA, Ozinsky A, Casanova JL (2003) Pyogenic bacterial infections in humans with IRAK-4 deficiency. Science 299:2076–2079. https://doi.org/10.1126/science.1081902
Picard C, von Bernuth H, Ghandil P, Chrabieh M, Levy O, Arkwright PD, McDonald D, Geha RS, Takada H, Krause JC, Creech CB, Ku CL, Ehl S, Marodi L, Al-Muhsen S, Al-Hajjar S, Al-Ghonaium A, Day-Good NK, Holland SM, Gallin JI, Chapel H, Speert DP, Rodriguez-Gallego C, Colino E, Garty BZ, Roifman C, Hara T, Yoshikawa H, Nonoyama S, Domachowske J, Issekutz AC, Tang M, Smart J, Zitnik SE, Hoarau C, Kumararatne DS, Thrasher AJ, Davies EG, Bethune C, Sirvent N, de Ricaud D, Camcioglu Y, Vasconcelos J, Guedes M, Vitor AB, Rodrigo C, Almazan F, Mendez M, Arostegui JI, Alsina L, Fortuny C, Reichenbach J, Verbsky JW, Bossuyt X, Doffinger R, Abel L, Puel A, Casanova JL (2010) Clinical features and outcome of patients with IRAK-4 and MyD88 deficiency. Medicine (Baltimore) 89:403–425. https://doi.org/10.1097/MD.0b013e3181fd8ec3
Picard C, Casanova JL, Puel A (2011) Infectious diseases in patients with IRAK-4, MyD88, NEMO, or IkappaBalpha deficiency. Clin Microbiol Rev 24:490–497. https://doi.org/10.1128/CMR.00001-11
Pichlmair A, Schulz O, Tan CP, Naslund TI, Liljestrom P, Weber F, Reisesousa C (2006) RIG-I-mediated antiviral responses to single-stranded RNA bearing 5'-phosphates. Science 314:997–1001. https://doi.org/10.1126/science.1132998
Pothlichet J, Burtey A, Kubarenko AV, Caignard G, Solhonne B, Tangy F, Ben-Ali M, Quintana-Murci L, Heinzmann A, Chiche JD, Vidalain PO, Weber AN, Chignard M, Si-Tahar M (2009) Study of human RIG-I polymorphisms identifies two variants with an opposite impact on the antiviral immune response. PLoS ONE 4:e7582. https://doi.org/10.1371/journal.pone.0007582
Saba TG, Chung Y, Hong JY, Sajjan US, Bentley JK, Hershenson MB (2014) Rhinovirus-induced macrophage cytokine expression does not require endocytosis or replication. Am J Respir Cell Mol Biol 50:974–984. https://doi.org/10.1165/rcmb.2013-0354OC
Sajjan US, Jia Y, Newcomb DC, Bentley JK, Lukacs NW, LiPuma JJ, Hershenson MB (2006) H. influenzae potentiates airway epithelial cell responses to rhinovirus by increasing ICAM-1 and TLR3 expression. FASEB J 20:2121–2123. https://doi.org/10.1096/fj.06-5806fje
Sancho-Shimizu V, Perez de Diego R, Lorenzo L, Halwani R, Alangari A, Israelsson E, Fabrega S, Cardon A, Maluenda J, Tatematsu M, Mahvelati F, Herman M, Ciancanelli M, Guo Y, AlSum Z, Alkhamis N, Al-Makadma AS, Ghadiri A, Boucherit S, Plancoulaine S, Picard C, Rozenberg F, Tardieu M, Lebon P, Jouanguy E, Rezaei N, Seya T, Matsumoto M, Chaussabel D, Puel A, Zhang SY, Abel L, Al-Muhsen S, Casanova JL (2011) Herpes simplex encephalitis in children with autosomal recessive and dominant TRIF deficiency. J Clin Invest 121:4889–4902. https://doi.org/10.1172/JCI59259
Scully EJ, Basnet S, Wrangham RW, Muller MN, Otali E, Hyeroba D, Grindle KA, Pappas TE, Thompson ME, Machanda Z, Watters KE, Palmenberg AC, Gern JE, Goldberg TL (2018) Lethal respiratory disease associated with human rhinovirus C in Wild Chimpanzees, Uganda, 2013. Emerg Infect Dis 24:267–274. https://doi.org/10.3201/eid2402.170778
Shih SR, Chen SJ, Hakimelahi GH, Liu HJ, Tseng CT, Shia KS (2004) Selective human enterovirus and rhinovirus inhibitors: An overview of capsid-binding and protease-inhibiting molecules. Med Res Rev 24:449–474. https://doi.org/10.1002/med.10067
Silkoff PE, Flavin S, Gordon R, Loza MJ, Sterk PJ, Lutter R, Diamant Z, Turner RB, Lipworth BJ, Proud D, Singh D, Eich A, Backer V, Gern JE, Herzmann C, Halperin SA, Mensinga TT, Del Vecchio AM, Branigan P, San Mateo L, Baribaud F, Barnathan ES, Johnston SL (2018) Toll-like receptor 3 blockade in rhinovirus-induced experimental asthma exacerbations: a randomized controlled study. J Allergy Clin Immunol 141:1220–1230. https://doi.org/10.1016/j.jaci.2017.06.027
Slater L, Bartlett NW, Haas JJ, Zhu J, Message SD, Walton RP, Sykes A, Dahdaleh S, Clarke DL, Belvisi MG, Kon OM, Fujita T, Jeffery PK, Johnston SL, Edwards MR (2010) Co-ordinated role of TLR3, RIG-I and MDA5 in the innate response to rhinovirus in bronchial epithelium. PLoS Pathog 6:e1001178. https://doi.org/10.1371/journal.ppat.1001178
Springate CS, McMeekin RR, Ruehle CJ (1989) Fire deaths in aircraft without the crashworthy fuel system. Aviat Space Environ Med 60:B35–B38
Staunton DE, Merluzzi VJ, Rothlein R, Barton R, Marlin SD, Springer TA (1989) A cell adhesion molecule, ICAM-1, is the major surface receptor for rhinoviruses. Cell 56:849–853. https://doi.org/10.1016/0092-8674(89)90689-2
Triantafilou K, Vakakis E, Richer EA, Evans GL, Villiers JP, Triantafilou M (2011) Human rhinovirus recognition in non-immune cells is mediated by Toll-like receptors and MDA-5, which trigger a synergetic pro-inflammatory immune response. Virulence 2:22–29
Unger BL, Faris AN, Ganesan S, Comstock AT, Hershenson MB, Sajjan US (2012) Rhinovirus attenuates non-typeable Hemophilus influenzae-stimulated IL-8 responses via TLR2-dependent degradation of IRAK-1. PLoS Pathog 8:e1002969. https://doi.org/10.1371/journal.ppat.1002969
von Bernuth H, Picard C, Jin Z, Pankla R, Xiao H, Ku CL, Chrabieh M, Mustapha IB, Ghandil P, Camcioglu Y, Vasconcelos J, Sirvent N, Guedes M, Vitor AB, Herrero-Mata MJ, Arostegui JI, Rodrigo C, Alsina L, Ruiz-Ortiz E, Juan M, Fortuny C, Yague J, Anton J, Pascal M, Chang HH, Janniere L, Rose Y, Garty BZ, Chapel H, Issekutz A, Marodi L, Rodriguez-Gallego C, Banchereau J, Abel L, Li X, Chaussabel D, Puel A, Casanova JL (2008) Pyogenic bacterial infections in humans with MyD88 deficiency. Science 321:691–696
Walter JM, Wunderink RG (2017) Severe respiratory viral infections: new evidence and changing paradigms. Infect Dis Clin North Am 31:455–474. https://doi.org/10.1016/j.idc.2017.05.004
Wang Q, Nagarkar DR, Bowman ER, Schneider D, Gosangi B, Lei J, Zhao Y, McHenry CL, Burgens RV, Miller DJ, Sajjan U, Hershenson MB (2009) Role of double-stranded RNA pattern recognition receptors in rhinovirus-induced airway epithelial cell responses. J Immunol 183:6989–6997. https://doi.org/10.4049/jimmunol.0901386
Wang Q, Miller DJ, Bowman ER, Nagarkar DR, Schneider D, Zhao Y, Linn MJ, Goldsmith AM, Bentley JK, Sajjan US, Hershenson MB (2011) MDA5 and TLR3 initiate pro-inflammatory signaling pathways leading to rhinovirus-induced airways inflammation and hyperresponsiveness. PLoS Pathog 7:e1002070. https://doi.org/10.1371/journal.ppat.1002070
Wu B, Hur S (2015) How RIG-I like receptors activate MAVS. Curr Opin Virol 12:91–98. https://doi.org/10.1016/j.coviro.2015.04.004
Zaki M, Thoenes M, Kawalia A, Nurnberg P, Kaiser R, Heller R, Bolz HJ (2017) Recurrent and prolonged infections in a child with a homozygous IFIH1 nonsense mutation. Front Genet 8:130. https://doi.org/10.3389/fgene.2017.00130
Zhang SY, Jouanguy E, Ugolini S, Smahi A, Elain G, Romero P, Segal D, Sancho-Shimizu V, Lorenzo L, Puel A, Picard C, Chapgier A, Plancoulaine S, Titeux M, Cognet C, von Bernuth H, Ku CL, Casrouge A, Zhang XX, Barreiro L, Leonard J, Hamilton C, Lebon P, Heron B, Vallee L, Quintana-Murci L, Hovnanian A, Rozenberg F, Vivier E, Geissmann F, Tardieu M, Abel L, Casanova JL (2007) TLR3 deficiency in patients with herpes simplex encephalitis. Science 317:1522–1527
Acknowledgements
This work was supported by the Intramural Research Program of the National Institute of Allergy and Infectious Diseases, National Institutes of Health (Grant no. ZIAAI001059). On behalf of all authors, the corresponding author states that there is no conflict of interest. We thank Drs. Huie Jing and Yu Zhang for helpful comments.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lamborn, I.T., Su, H.C. Genetic determinants of host immunity against human rhinovirus infections. Hum Genet 139, 949–959 (2020). https://doi.org/10.1007/s00439-020-02137-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00439-020-02137-3