Abstract
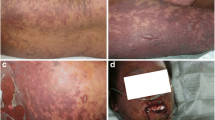
Stevens–Johnson syndrome (SJS) and toxic epidermal necrolysis (TEN) are rare severe cutaneous adverse reactions to drugs. Granulysin (GNLY) plays a key role in keratinocyte apoptosis during SJS/TEN pathophysiology. To determine if GNLY-encoding mutations might be related to the protein’s functional disturbances, contributing to SJS/TEN pathogenesis, we performed direct sequencing of GNLY’s coding region in a group of 19 Colombian SJS/TEN patients. A GNLY genetic screening was implemented in a group of 249 healthy individuals. We identified the c.11G > A heterozygous sequence variant in a TEN case, which creates a premature termination codon (PTC) (p.Trp4Ter). We show that a mutant protein is synthesised, possibly due to a PTC-readthrough mechanism. Functional assays demonstrated that the mutant protein was abnormally located in the nuclear compartment, potentially leading to a toxic effect. Our results argue in favour of GNLY non-synonymous sequence variants contributing to SJS/TEN pathophysiology, thereby constituting a promising, clinically useful molecular biomarker.


Similar content being viewed by others
Data availability statement
The data and the GNLY c.11G > A variant that support the findings of this study were submitted to a locus-specific database (https://databases.lovd.nl/shared/genes/GNLY).
References
Abe R, Yoshioka N, Murata J, Fujita Y, Shimizu H (2009) Granulysin as a marker for early diagnosis of the Stevens-Johnson syndrome. Ann Intern Med 151:514–515
Blanchet S, Rowe M, Von der Haar T, Fabret C, Demais S, Howard MJ, Namy O (2015) New insights into stop codon recognition by eRF1. Nucleic Acids Res 43:3298–3308. https://doi.org/10.1093/nar/gkv154
Brogna S, Wen J (2009) Nonsense-mediated mRNA decay (NMD) mechanisms. Nat Struct Mol Biol 16:107–113. https://doi.org/10.1038/nsmb.1550
Caruso A, Bellia C, Pivetti A, Agnello L, Bazza F, Scazzone C, Bivona G, Lo Sasso B, Ciaccio M (2014) Effects of EPHX1 and CYP3A4 polymorphisms on carbamazepine metabolism in epileptic patients. Pharmgenomics Pers Med 7:117–120. https://doi.org/10.2147/pgpm.S55548
Chung WH, Hung SI, Hong HS, Hsih MS, Yang LC, Ho HC, Wu JY, Chen YT (2004) Medical genetics: a marker for Stevens–Johnson syndrome. Nature 428:486. https://doi.org/10.1038/428486a
Chung WH, Hung SI, Yang JY, Su SC, Huang SP, Wei CY, Chin SW, Chiou CC, Chu SC, Ho HC, Yang CH, Lu CF, Wu JY, Liao YD, Chen YT (2008) Granulysin is a key mediator for disseminated keratinocyte death in Stevens–Johnson syndrome and toxic epidermal necrolysis. Nat Med 14:1343–1350. https://doi.org/10.1038/nm.1884
Ciccacci C, Di Fusco D, Marazzi MC, Zimba I, Erba F, Novelli G, Palombi L, Borgiani P, Liotta G (2013) Association between CYP2B6 polymorphisms and Nevirapine-induced SJS/TEN: a pharmacogenetics study. Eur J Clin Pharmacol 69:1909–1916. https://doi.org/10.1007/s00228-013-1549-x
Cridge AG, Crowe-McAuliffe C, Mathew SF, Tate WP (2018) Eukaryotic translational termination efficiency is influenced by the 3′ nucleotides within the ribosomal mRNA channel. Nucleic Acids Res 46:1927–1944. https://doi.org/10.1093/nar/gkx1315
Dabrowski M, Bukowy-Bieryllo Z, Zietkiewicz E (2015) Translational readthrough potential of natural termination codons in eucaryotes—the impact of RNA sequence. RNA Biol 12:950–958. https://doi.org/10.1080/15476286.2015.1068497
Dodiuk-Gad RP, Chung WH, Valeyrie-Allanore L, Shear NH (2015) Stevens–Johnson syndrome and toxic epidermal necrolysis: an update. Am J Clin Dermatol 16:475–493. https://doi.org/10.1007/s40257-015-0158-0
Duong TA, Valeyrie-Allanore L, Wolkenstein P, Chosidow O (2017) Severe cutaneous adverse reactions to drugs. Lancet 390:1996–2011. https://doi.org/10.1016/s0140-6736(16)30378-6
Esposito S, Bianco A, Russo R, Di Maro A, Isernia C, Pedone PV (2019) Therapeutic perspectives of molecules from Urtica dioica extracts for cancer treatment. Molecules. https://doi.org/10.3390/molecules24152753
Fakoya AOJ, Omenyi P, Anthony P, Anthony F, Etti P, Otohinoyi DA, Olunu E (2018) Stevens–Johnson syndrome and toxic epidermal necrolysis; extensive review of reports of drug-induced etiologies, and possible therapeutic modalities. Open Access Maced J Med Sci 6:730–738. https://doi.org/10.3889/oamjms.2018.148
Fan WL, Shiao MS, Hui RC, Su SC, Wang CW, Chang YC, Chung WH (2017) HLA association with drug-induced adverse reactions. J Immunol Res 2017:3186328. https://doi.org/10.1155/2017/3186328
Fearon K, McClendon V, Bonetti B, Bedwell DM (1994) Premature translation termination mutations are efficiently suppressed in a highly conserved region of yeast Ste6p, a member of the ATP-binding cassette (ABC) transporter family. J Biol Chem 269:17802–17808
Feng YX, Copeland TD, Oroszlan S, Rein A, Levin JG (1990) Identification of amino acids inserted during suppression of UAA and UGA termination codons at the gag-pol junction of Moloney murine leukemia virus. Proc Natl Acad Sci USA 87:8860–8863. https://doi.org/10.1073/pnas.87.22.8860
Finn MW, Clayberger C, Krensky AM (2011) Expression and purification of 15 kDa granulysin utilizing an insect cell secretion system. Protein Expr Purif 75:70–74. https://doi.org/10.1016/j.pep.2010.07.009
Hanson DA, Kaspar AA, Poulain FR, Krensky AM (1999) Biosynthesis of granulysin, a novel cytolytic molecule. Mol Immunol 36:413–422
Harr T, French LE (2010) Toxic epidermal necrolysis and Stevens–Johnson syndrome. Orphanet J Rare Dis 5:39. https://doi.org/10.1186/1750-1172-5-39
Harris V, Jackson C, Cooper A (2016) Review of toxic epidermal necrolysis. Int J Mol Sci. https://doi.org/10.3390/ijms17122135
He XJ, Jian LY, He XL, Tang M, Wu Y, Xu YY, Sun XJ, Zhao LM (2014) Association of ABCB1, CYP3A4, EPHX1, FAS, SCN1A, MICA, and BAG6 polymorphisms with the risk of carbamazepine-induced Stevens–Johnson syndrome/toxic epidermal necrolysis in Chinese Han patients with epilepsy. Epilepsia 55:1301–1306. https://doi.org/10.1111/epi.12655
Kregiel D, Pawlikowska E, Antolak H (2018) Urtica spp.: ordinary plants with extraordinary properties. Molecules. https://doi.org/10.3390/molecules23071664
Letko E, Papaliodis DN, Papaliodis GN, Daoud YJ, Ahmed AR, Foster CS (2005) Stevens–Johnson syndrome and toxic epidermal necrolysis: a review of the literature. Ann Allergy Asthma Immunol 94:419–436. https://doi.org/10.1016/s1081-1206(10)61112-x(quiz 436-8, 456)
Lindeboom RG, Supek F, Lehner B (2016) The rules and impact of nonsense-mediated mRNA decay in human cancers. Nat Genet 48:1112–1118. https://doi.org/10.1038/ng.3664
Loughran G, Jungreis I, Tzani I, Power M, Dmitriev RI, Ivanov IP, Kellis M, Atkins JF (2018) Stop codon readthrough generates a C-terminally extended variant of the human vitamin D receptor with reduced calcitriol response. J Biol Chem 293:4434–4444. https://doi.org/10.1074/jbc.M117.818526
Man CB, Kwan P, Baum L, Yu E, Lau KM, Cheng AS, Ng MH (2007) Association between HLA-B*1502 allele and antiepileptic drug-induced cutaneous reactions in Han Chinese. Epilepsia 48:1015–1018. https://doi.org/10.1111/j.1528-1167.2007.01022.x
Maquat LE (2004) Nonsense-mediated mRNA decay: splicing, translation and mRNP dynamics. Nat Rev Mol Cell Biol 5:89–99. https://doi.org/10.1038/nrm1310
Nagy E, Maquat LE (1998) A rule for termination-codon position within intron-containing genes: when nonsense affects RNA abundance. Trends Biochem Sci 23:198–199
Namy O, Duchateau-Nguyen G, Hatin I, Hermann-Le Denmat S, Termier M, Rousset JP (2003) Identification of stop codon readthrough genes in Saccharomyces cerevisiae. Nucleic Acids Res 31:2289–2296. https://doi.org/10.1093/nar/gkg330
Nilsson M, Ryden-Aulin M (2003) Glutamine is incorporated at the nonsense codons UAG and UAA in a suppressor-free Escherichia coli strain. Biochim Biophys Acta 1627:1–6
Pacho F, Zambruno G, Calabresi V, Kiritsi D, Schneider H (2011) Efficiency of translation termination in humans is highly dependent upon nucleotides in the neighbourhood of a (premature) termination codon. J Med Genet 48:640–644. https://doi.org/10.1136/jmg.2011.089615
Pavlos R, Mallal S, Phillips E (2012) HLA and pharmacogenetics of drug hypersensitivity. Pharmacogenomics 13:1285–1306. https://doi.org/10.2217/pgs.12.108
Rodriguez-Martin S, Martin-Merino E, Lerma V, Rodriguez-Miguel A, Gonzalez O, Gonzalez-Herrada C, Ramirez E, Bellon T, de Abajo FJ (2019) Incidence of Stevens–Johnson syndrome/toxic epidermal necrolysis among new users of different individual drugs in a European population: a case-population study. Eur J Clin Pharmacol 75:237–246. https://doi.org/10.1007/s00228-018-2569-3
Roujeau JC (1997) Stevens–Johnson syndrome and toxic epidermal necrolysis are severity variants of the same disease which differs from erythema multiforme. J Dermatol 24:726–729
Schwartz RA, McDonough PH, Lee BW (2013) Toxic epidermal necrolysis: Part I Introduction, history, classification, clinical features, systemic manifestations, etiology, and immunopathogenesis. J Am Acad Dermatol 69:173.e1–13. https://doi.org/10.1016/j.jaad.2013.05.003(quiz 185–6)
Silva AL, Ribeiro P, Inacio A, Liebhaber SA, Romao L (2008) Proximity of the poly(A)-binding protein to a premature termination codon inhibits mammalian nonsense-mediated mRNA decay. RNA 14:563–576. https://doi.org/10.1261/rna.815108
Takamori Y, Ogawa K, Nagata K, Takano S, Nakamura M (2005) Granulysin induces cell death with nuclear accumulation. J Med Dent Sci 52:1–7
Tassaneeyakul W, Tiamkao S, Jantararoungtong T, Chen P, Lin SY, Chen WH, Konyoung P, Khunarkornsiri U, Auvichayapat N, Pavakul K, Kulkantrakorn K, Choonhakarn C, Phonhiamhan S, Piyatrakul N, Aungaree T, Pongpakdee S, Yodnopaglaw P (2010) Association between HLA-B*1502 and carbamazepine-induced severe cutaneous adverse drug reactions in a Thai population. Epilepsia 51:926–930. https://doi.org/10.1111/j.1528-1167.2010.02533.x
Ueta M, Sawai H, Sotozono C, Hitomi Y, Kaniwa N, Kim MK, Seo KY, Yoon KC, Joo CK, Kannabiran C, Wakamatsu TH, Sangwan V, Rathi V, Basu S, Ozeki T, Mushiroda T, Sugiyama E, Maekawa K, Nakamura R, Aihara M, Matsunaga K, Sekine A, Gomes JA, Hamuro J, Saito Y, Kubo M, Kinoshita S, Tokunaga K (2015) IKZF1, a new susceptibility gene for cold medicine-related Stevens-Johnson syndrome/toxic epidermal necrolysis with severe mucosal involvement. J Allergy Clin Immunol 135:1538–45.e17. https://doi.org/10.1016/j.jaci.2014.12.1916
Williams I, Richardson J, Starkey A, Stansfield I (2004) Genome-wide prediction of stop codon readthrough during translation in the yeast Saccharomyces cerevisiae. Nucleic Acids Res 32:6605–6616. https://doi.org/10.1093/nar/gkh1004
Yang Y, Li F, Du J, Shen Y, Lin J, Zhu X, Luo X, Liang J, Xu J (2017) Variable levels of apoptotic signal-associated cytokines in the disease course of patients with Stevens–Johnson syndrome and toxic epidermal necrolysis. Australas J Dermatol 58:e61–e67. https://doi.org/10.1111/ajd.12462
Yip VL, Alfirevic A, Pirmohamed M (2015) Genetics of immune-mediated adverse drug reactions: a comprehensive and clinical review. Clin Rev Allergy Immunol 48:165–175. https://doi.org/10.1007/s12016-014-8418-y
Zhang J, Maquat LE (1997) Evidence that translation reinitiation abrogates nonsense-mediated mRNA decay in mammalian cells. EMBO J 16:826–833. https://doi.org/10.1093/emboj/16.4.826
Acknowledgements
This study and Laissue´s Lab is supported by the Universidad del Rosario (Grant: CS/ABN062/GENIUROS 019).
Author information
Authors and Affiliations
Contributions
LC, CS, HM, AR, and AD performed the clinical study. DF, DS, OL, YS, and DB performed the in vitro assays. PL directed the study and wrote the paper. All authors revised and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that conflict of interest does not exist.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Fonseca, D.J., Caro, L.A., Sierra-Díaz, D.C. et al. Mutant GNLY is linked to Stevens–Johnson syndrome and toxic epidermal necrolysis. Hum Genet 138, 1267–1274 (2019). https://doi.org/10.1007/s00439-019-02066-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00439-019-02066-w