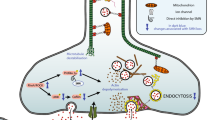
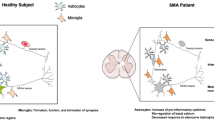
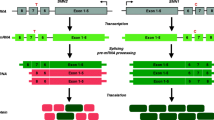
Abstract
Low levels of the survival of motor neuron (SMN) protein cause the neurodegenerative disease spinal muscular atrophy (SMA). SMA is a pediatric disease characterized by spinal motor neuron degeneration. SMA exhibits several levels of severity ranging from early antenatal fatality to only mild muscular weakness, and disease prognosis is related directly to the amount of functional SMN protein that a patient is able to express. Current therapies are being developed to increase the production of functional SMN protein; however, understanding the effect that natural stresses have on the production and function of SMN is of critical importance to ensuring that these therapies will have the greatest possible effect for patients. Research has shown that SMN, both on the mRNA and protein level, is highly affected by cellular stress. In this review we will summarize the research that highlights the roles of SMN in the disease process and the response of SMN to various environmental stresses.
Similar content being viewed by others
References
Achsel T, Barabino S, Cozzolino M, Carrì MT (2013) The intriguing case of motor neuron disease: ALS and SMA come closer. Biochem Soc Trans 41(6):1593–1597. doi:10.1042/BST20130142
Acsadi G, Lee I, Li X, Khaidakov M, Pecinova A, Parker GC, Hüttemann M (2009) Mitochondrial dysfunction in a neural cell model of spinal muscular atrophy. J Neurosci Res 87(12):2748–2756. doi:10.1002/jnr.22106
Akten B, Kye MJ, Hao LT, Wertz MH, Singh S, Nie D, Huang J et al (2011) Interaction of survival of motor neuron (SMN) and HuD proteins with mRNA Cpg15 rescues motor neuron axonal deficits. Proc Natl Acad Sci USA 108(25):10337–10342. doi:10.1073/pnas.1104928108
Al-Fageeh MB, Smales CM (2006) Control and regulation of the cellular responses to cold shock: the responses in yeast and mammalian systems. Biochem J 397(2):247–259. doi:10.1042/BJ20060166
Alías L, Bernal S, Fuentes-Prior P, Barceló MJ, Also E, Martínez-Hernández R, Rodríguez-Alvarez FJ et al (2009) Mutation update of spinal muscular atrophy in Spain: molecular characterization of 745 unrelated patients and identification of four novel mutations in the SMN1 gene. Hum Genet 125(1):29–39. doi:10.1007/s00439-008-0598-1
Balchin D, Hayer-Hartl M, Hartl FU (2016) In vivo aspects of protein folding and quality control. Science 353(6294):aac4354. doi:10.1126/science.aac4354
Bebee TW, Dominguez CE, Samadzadeh-Tarighat S, Akehurst KL, Chandler DS (2012) Hypoxia is a modifier of SMN2 splicing and disease severity in a severe SMA mouse model. Hum Mol Genet 21(19):4301–4313. doi:10.1093/hmg/dds263
Bladen CL, Thompson R, Jackson JM, Garland C, Wegel C, Ambrosini A, Pisano P et al (2014) Mapping the differences in care for 5,000 spinal muscular atrophy patients, a survey of 24 National Registries in North America, Australasia and Europe. J Neurol 261(1):152–163. doi:10.1007/s00415-013-7154-1
Boon K-L, Xiao S, McWhorter ML, Donn T, Wolf-Saxon E, Bohnsack MT, Moens CB, Beattie CE (2009) Zebrafish survival motor neuron mutants exhibit presynaptic neuromuscular junction defects. Hum Mol Genet 18(19):3615–3625. doi:10.1093/hmg/ddp310
Boyd PJ, Tu WY, Shorrock HK, Groen EJN, Carter RN, Powis RA, Thomson SR, Thomson D, Graham LC, Motyl AAL, Wishart TM, Highley JR, Morton NM, Becker T, Becker CG, Heath PR, Gillingwater TH (2017) Bioenergetic status modulates motor neuron vulnerability and pathogenesis in a zebrafish model of spinal muscular atrophy. PLoS Genet 13(4):e1006744. doi:10.1371/journal.pgen.1006744
Bühler D, Raker V, Lührmann R, Fischer U (1999) Essential role for the tudor domain of SMN in spliceosomal U snRNP assembly: implications for spinal muscular atrophy. Hum Mol Genet 8(13):2351–2357
Burghes AH (1997) When is a deletion not a deletion? When it is converted. Am J Hum Genet 61(1):9–15. doi:10.1086/513913
Burghes AHM, Beattie CE (2009) Spinal muscular atrophy: why do low levels of survival motor neuron protein make motor neurons sick? Nat Rev Neurosci 10(8):597–609. doi:10.1038/nrn2670
Burnett BG, Muñoz E, Tandon A, Kwon DY, Sumner CJ, Fischbeck KH (2009) Regulation of SMN protein stability. Mol Cell Biol 29(5):1107–1115. doi:10.1128/MCB.01262-08
Cao W, Jamison SF, Garcia-Blanco MA (1997) Both phosphorylation and dephosphorylation of ASF/SF2 are required for pre-mRNA splicing in vitro. RNA 3(12):1456–1467
Cao W, Sohail M, Liu G, Koumbadinga GA, Lobo VG, Xie J (2011) Differential effects of PKA-controlled CaMKK2 variants on neuronal differentiation. RNA Biol 8(6):1061–1072. doi:10.4161/rna.8.6.16691
Carissimi C, Saieva L, Gabanella F, Pellizzoni L (2006a) Gemin8 is required for the architecture and function of the survival motor neuron complex. J Biol Chem 281(48):37009–37016. doi:10.1074/jbc.M607505200
Carissimi C, Saieva L, Baccon J, Chiarella P, Maiolica A, Sawyer A, Rappsilber J, Pellizzoni L (2006b) Gemin8 is a novel component of the survival motor neuron complex and functions in small nuclear ribonucleoprotein assembly. J Biol Chem 281(12):8126–8134. doi:10.1074/jbc.M512243200
Cartegni L, Krainer AR (2002) Disruption of an SF2/ASF-dependent exonic splicing enhancer in SMN2 causes spinal muscular atrophy in the absence of SMN1. Nat Genet 30(4):377–384. doi:10.1038/ng854
Cartegni L, Hastings ML, Calarco JA, de Stanchina E, Krainer AR (2006) Determinants of exon 7 splicing in the spinal muscular atrophy genes, SMN1 and SMN2. Am J Hum Genet 78(1):63–77. doi:10.1086/498853
Chan YB, Miguel-Aliaga I, Franks C, Thomas N, Trülzsch B, Sattelle DB, Davies KE, van den Heuvel M (2003) Neuromuscular defects in a Drosophila survival motor neuron gene mutant. Hum Mol Genet 12(12):1367–1376
Chang HC-H, Dimlich DN, Yokokura T, Mukherjee A, Kankel MW, Sen A, Sridhar V et al (2008) Modeling spinal muscular atrophy in Drosophila. PLoS One 3(9):e3209. doi:10.1371/journal.pone.0003209
Chen H-H, Chang J-G, Lu R-M, Peng T-Y, Tarn W-Y (2008) The RNA binding protein hnRNP Q modulates the utilization of exon 7 in the survival motor neuron 2 (SMN2) gene. Mol Cell Biol 28(22):6929–6938. doi:10.1128/MCB.01332-08
Cho S, Moon H, Loh TJ, Oh HK, Cho S, Choy HE, Song WK, Chun JS, Zheng X, Shen H (2014) hnRNP M facilitates exon 7 inclusion of SMN2 pre-mRNA in spinal muscular atrophy by targeting an enhancer on exon 7. Biochim Biophys Acta 1839(4):306–315. doi:10.1016/j.bbagrm.2014.02.006
Clermont O, Burlet P, Benit P, Chanterau D, Saugier-Veber P, Munnich A, Cusin V (2004) Molecular analysis of SMA patients without homozygous SMN1 deletions using a new strategy for identification of SMN1 subtle mutations. Hum Mutat 24(5):417–427. doi:10.1002/humu.20092
Cléry A, Jayne S, Benderska N, Dominguez C, Stamm S, Allain FH-T (2011) Molecular basis of purine-rich RNA recognition by the human SR-like protein Tra2-Β1. Nat Struct Mol Biol 18(4):443–450. doi:10.1038/nsmb.2001
Cobben JM, Lemmink HH, Snoeck I, Barth PA, van der Lee JH, de Visser M (2008) Survival in SMA type I: a prospective analysis of 34 consecutive cases. Neuromuscul Disord NMD 18(7):541–544. doi:10.1016/j.nmd.2008.05.008
Coovert DD, Le TT, McAndrew PE, Strasswimmer J, Crawford TO, Mendell JR, Coulson SE, Androphy EJ, Prior TW, Burghes AH (1997) The survival motor neuron protein in spinal muscular atrophy. Hum Mol Genet 6(8):1205–1214
Cuscó I, Jesus Barceló M, del Río E, Baiget M, Tizzano EF (2004) Detection of novel mutations in the SMN Tudor domain in type I SMA patients. Neurology 63(1):146–149
d’Errico P, Boido M, Piras A, Valsecchi V, De Amicis E, Locatelli D, Capra S, Vagni F, Vercelli A, Battaglia G (2013) Selective vulnerability of spinal and cortical motor neuron subpopulations in delta7 SMA mice. PLoS One 8(12):e82654. doi:10.1371/journal.pone.0082654
Denegri M, Chiodi I, Corioni M, Cobianchi F, Riva S, Biamonti G (2001) Stress-induced nuclear bodies are sites of accumulation of pre-mRNA processing factors. Mol Biol Cell 12(11):3502–3514
Doktor TK, Hua Y, Andersen HS, Brøner S, Liu YH, Wieckowska A, Dembic M, Bruun GH, Krainer AR, Andresen BS (2017) RNA-sequencing of a mouse-model of spinal muscular atrophy reveals tissue-wide changes in splicing of U12-dependent introns. Nucleic Acids Res 45(1):395–416. doi:10.1093/nar/gkw731
Donlin-Asp PG, Bassell GJ, Rossoll W (2016) A role for the survival of motor neuron protein in mRNP assembly and transport. Curr Opin Neurobiol 39:53–61. doi:10.1016/j.conb.2016.04.004
Eggert C, Chari A, Laggerbauer B, Fischer U (2006) Spinal muscular atrophy: the RNP connection. Trends Mol Med 12(3):113–121. doi:10.1016/j.molmed.2006.01.005
Fallini C, Zhang H, Su Y, Silani V, Singer RH, Rossoll W, Bassell GJ (2011) The survival of motor neuron (SMN) protein interacts with the mRNA-binding protein HuD and regulates localization of poly(a) mRNA in primary motor neuron axons. J Neurosci 31(10):3914–3925. doi:10.1523/JNEUROSCI.3631-10.2011
Fallini C, Bassell GJ, Rossoll W (2012) Spinal muscular atrophy: the role of SMN in axonal mRNA regulation. Brain Res 1462:81–92. doi:10.1016/j.brainres.2012.01.044
Fallini C, Rouanet JP, Donlin-Asp PG, Guo P, Zhang H, Singer RH, Rossoll W, Bassell GJ (2014) Dynamics of survival of motor neuron (SMN) protein interaction with the mRNA-binding protein IMP1 facilitates its trafficking into motor neuron axons. Dev Neurobiol 74(3):319–332. doi:10.1002/dneu.22111
Fallini C, Donlin-Asp PG, Rouanet JP, Bassell GJ, Rossoll W (2016) Deficiency of the survival of motor neuron protein impairs mRNA localization and local translation in the growth cone of motor neurons. J Neurosci 36(13):3811–3820. doi:10.1523/JNEUROSCI.2396-15.2016
Farooq F, Balabanian S, Liu X, Holcik M, MacKenzie A (2009) P38 mitogen-activated protein kinase stabilizes SMN mRNA through RNA binding protein HuR. Hum Mol Genet 18(21):4035–4045. doi:10.1093/hmg/ddp352
Finkel RS (2013) Electrophysiological and motor function scale association in a pre-symptomatic infant with spinal muscular atrophy type I. Neuromuscul Disord NMD 23(2):112–115. doi:10.1016/j.nmd.2012.09.006
Fischer U, Liu Q, Dreyfuss G (1997) The SMN–SIP1 complex has an essential role in spliceosomal snRNP biogenesis. Cell 90(6):1023–1029
Gabanella F, Butchbach MER, Saieva L, Carissimi C, Burghes AHM, Pellizzoni L (2007) Ribonucleoprotein assembly defects correlate with spinal muscular atrophy severity and preferentially affect a subset of spliceosomal snRNPs. PLoS One 2(9):e921. doi:10.1371/journal.pone.0000921
Garcia EL, Lu Z, Meers MP, Praveen K, Matera AG (2013) Developmental arrest of drosophila survival motor neuron (Smn) mutants accounts for differences in expression of minor intron-containing genes. RNA 19(11):1510–1516. doi:10.1261/rna.038919.113
Gerstein MB, Rozowsky J, Yan K-K, Wang D, Cheng C, Brown JB, Davis CA et al (2014) Comparative analysis of the transcriptome across distant species. Nature 512(7515):445–448. doi:10.1038/nature13424
Gonsalvez GB, Rajendra TK, Wen Y, Praveen K, Matera AG (2010) Sm proteins specify germ cell fate by facilitating oskar mRNA localization. Development 137(14):2341–2351. doi:10.1242/dev.042721
Gubitz AK, Feng W, Dreyfuss G (2004) The SMN complex. Exp Cell Res 296(1):51–56. doi:10.1016/j.yexcr.2004.03.022
Guil S, Long JC, Cáceres JF (2006) hnRNP A1 relocalization to the stress granules reflects a role in the stress response. Mol Cell Biol 26(15):5744–5758. doi:10.1128/MCB.00224-06
Hahnen E, Schönling J, Rudnik-Schöneborn S, Raschke H, Zerres K, Wirth B (1997) Missense mutations in exon 6 of the survival motor neuron gene in patients with spinal muscular atrophy (SMA). Hum Mol Genet 6(5):821–825
Hao LT, Duy PQ, Jontes JD, Wolman M, Granato M, Beattie CE (2013) Temporal requirement for SMN in motoneuron development. Hum Mol Genet 22(13):2612–2625. doi:10.1093/hmg/ddt110
Hao LT, Duy PQ, Jontes JD, Beattie CE (2015) Motoneuron development influences dorsal root ganglia survival and Schwann cell development in a vertebrate model of spinal muscular atrophy. Hum Mol Genet 24(2):346–360. doi:10.1093/hmg/ddu447
Harada Y, Sutomo R, Sadewa AH, Akutsu T, Takeshima Y, Wada H, Matsuo M, Nishio H (2002) Correlation between SMN2 copy number and clinical phenotype of spinal muscular atrophy: three SMN2 copies fail to rescue some patients from the disease severity. J Neurol 249(9):1211–1219. doi:10.1007/s00415-002-0811-4
Hofmann Y, Wirth B (2002) hnRNP-G promotes exon 7 inclusion of survival motor neuron (SMN) via direct interaction with Htra2-Beta1. Hum Mol Genet 11(17):2037–2049
Hofmann Y, Lorson CL, Stamm S, Androphy EJ, Wirth B (2000) Htra2-beta 1 stimulates an exonic splicing enhancer and can restore full-length SMN expression to survival motor neuron 2 (SMN2). Proc Natl Acad Sci USA 97(17):9618–9623. doi:10.1073/pnas.160181697
Howell MD, Ottesen EW, Singh NN, Anderson RL, Singh RN (2017) Gender-specific amelioration of SMA phenotype upon disruption of a deep intronic structure by an oligonucleotide. Mol Ther 25(6):1328–1341. doi:10.1016/j.ymthe.2017.03.036
Hsieh-Li HM, Chang JG, Jong YJ, Wu MH, Wang NM, Tsai CH, Li H (2000) A mouse model for spinal muscular atrophy. Nat Genet 24(1):66–70
Hua Y, Zhou J (2004) Survival motor neuron protein facilitates assembly of stress granules. FEBS Lett 572(1–3):69–74
Hua Y, Vickers TA, Okunola HL, Bennett CF, Krainer AR (2008) Antisense masking of an hnRNP A1/A2 intronic splicing silencer corrects SMN2 splicing in transgenic mice. Am J Hum Genet 82(4):834–848. doi:10.1016/j.ajhg.2008.01.014
Hubers L, Valderrama-Carvajal H, Laframboise J, Timbers J, Sanchez G, Côté J (2011) HuD interacts with survival motor neuron protein and can rescue spinal muscular atrophy-like neuronal defects. Hum Mol Genet 20(3):553–579. doi:10.1093/hmg/ddq500
Imlach WL, Beck ES, Choi BJ, Lotti F, Pellizzoni L, McCabe BD (2012) SMN is required for sensory-motor circuit function in Drosophila. Cell 151(2):427–439. doi:10.1016/j.cell.2012.09.011
Jangi M, Fleet C, Cullen P, Gupta SV, Mekhoubad S, Chiao E, Allaire N et al (2017) SMN deficiency in severe models of spinal muscular atrophy causes widespread intron retention and DNA damage. Proc Natl Acad Sci USA 114(12):E2347–E2356. doi:10.1073/pnas.1613181114
Jo OD, Martin J, Bernath A, Masri J, Lichtenstein A, Gera J (2008) Heterogeneous nuclear ribonucleoprotein A1 regulates cyclin D1 and C-Myc internal ribosome entry site function through Akt signaling. J Biol Chem 283(34):23274–23287. doi:10.1074/jbc.M801185200
Kashima T, Manley JL (2003) A negative element in SMN2 exon 7 inhibits splicing in spinal muscular atrophy. Nat Genet 34(4):460–463. doi:10.1038/ng1207
Kim GH, Kim JE, Rhie SJ, Yoon S (2015) The role of oxidative stress in neurodegenerative diseases. Exp Neurobiol 24(4):325–340. doi:10.5607/en.2015.24.4.325
Kleinschmidt AM, Patton JR, Pederson T (1989) U2 small nuclear RNP assembly in vitro. Nucleic Acids Res 17(12):4817–4828
Kotani T, Sutomo R, Sasongko TH, Sadewa AH, Gunadi, Minato T, Fujii E et al (2007) A novel mutation at the N-terminal of SMN tudor domain inhibits its interaction with target proteins. J Neurol 254(5):624–630. doi:10.1007/s00415-006-0410-x
Kwiatkowski TJ Jr, Bosco DA, Leclerc AL, Tamrazian E, Vanderburg CR, Russ C, Davis A, Gilchrist J, Kasarskis EJ, Munsat T, Valdmanis P, Rouleau GA, Hosler BA, Cortelli P, de Jong PJ, Yoshinaga Y, Haines JL, Pericak-Vance MA, Yan J, Ticozzi N, Siddique T, McKenna-Yasek D, Sapp PC, Horvitz HR, Landers JE, Brown RH Jr (2009) Mutations in the FUS/TLS gene on chromosome 16 cause familial amyotrophic lateral sclerosis. Science 323(5918):1205–1208. doi:10.1126/science.1166066
Labunskyy VM, Hatfield DL, Gladyshev VN (2014) Selenoproteins: molecular pathways and physiological roles. Physiol Rev 94(3):739–777. doi:10.1152/physrev.00039.2013
Lai MC, Lin RI, Tarn WY (2001) Transportin-SR2 mediates nuclear import of phosphorylated SR proteins. Proc Natl Acad Sci USA 98(18):10154–10159. doi:10.1073/pnas.181354098
Le TT, Pham LT, Butchbach MER, Zhang HL, Monani UR, Coovert DD, Gavrilina TO, Xing L, Bassell GJ, Burghes AHM (2005) SMNDelta7, the major product of the centromeric survival motor neuron (SMN2) gene, extends survival in mice with spinal muscular atrophy and associates with full-length SMN. Hum Mol Genet 14(6):845–857. doi:10.1093/hmg/ddi078
Le TT, McGovern VL, Alwine IE, Wang X, Massoni-Laporte A, Rich MM, Burghes AHM (2011) Temporal requirement for high SMN expression in SMA mice. Hum Mol Genet 20(18):3578–3591. doi:10.1093/hmg/ddr275
Lefebvre S, Bürglen L, Reboullet S, Clermont O, Burlet P, Viollet L, Benichou B, Cruaud C, Millasseau P, Zeviani M (1995) Identification and characterization of a spinal muscular atrophy-determining gene. Cell 80(1):155–165
Lefebvre S, Burlet P, Liu Q, Bertrandy S, Clermont O, Munnich A, Dreyfuss G, Melki J (1997) Correlation between severity and SMN protein level in spinal muscular atrophy. Nat Genet 16(3):265–269. doi:10.1038/ng0797-265
Liu Q, Fischer U, Wang F, Dreyfuss G (1997) The spinal muscular atrophy disease gene product, SMN, and its associated protein SIP1 are in a complex with spliceosomal snRNP proteins. Cell 90(6):1013–1021
Liu-Yesucevitz L, Bilgutay A, Zhang YJ, Vanderweyde T, Citro A, Mehta T, Zaarur N, McKee A, Bowser R, Sherman M, Petrucelli L, Wolozin B (2010) Tar DNA binding protein-43 (TDP-43) associates with stress granules: analysis of cultured cells and pathological brain tissue. PLoS One 5(10):e13250. doi:10.1371/journal.pone.0013250
Lorson CL, Strasswimmer J, Yao JM, Baleja JD, Hahnen E, Wirth B, Le T, Burghes AH, Androphy EJ (1998) SMN oligomerization defect correlates with spinal muscular atrophy severity. Nat Genet 19(1):63–66. doi:10.1038/ng0598-63
Lorson CL, Hahnen E, Androphy EJ, Wirth B (1999) A single nucleotide in the SMN gene regulates splicing and is responsible for spinal muscular atrophy. Proc Natl Acad Sci USA 96(11):6307–6311
Lotti F, Imlach WL, Saieva L, Beck ES, Hao LT, Li DK, Jiao W et al (2012) An SMN-dependent U12 splicing event essential for motor circuit function. Cell 151(2):440–454. doi:10.1016/j.cell.2012.09.012
Lukong KE, Larocque D, Tyner AL, Richard S (2005) Tyrosine phosphorylation of Sam68 by breast tumor kinase regulates intranuclear localization and cell cycle progression. J Biol Chem 280(46):38639–38647. doi:10.1074/jbc.M505802200
Lutz CM, Kariya S, Patruni S, Osborne MA, Liu D, Henderson CE, Li DK et al (2011) Postsymptomatic restoration of SMN rescues the disease phenotype in a mouse model of severe spinal muscular atrophy. J Clin Investig 121(8):3029–3041. doi:10.1172/JCI57291
Mahboubi H, Stochaj U (2017) Cytoplasmic stress granules: dynamic modulators of cell signaling and disease. Biochim Biophys Acta 1863(4):884–895. doi:10.1016/j.bbadis.2016.12.022
Matter N, Herrlich P, König H (2002) Signal-dependent regulation of splicing via phosphorylation of Sam68. Nature 420(6916):691–695. doi:10.1038/nature01153
McAndrew PE, Parsons DW, Simard LR, Rochette C, Ray PN, Mendell JR, Prior TW, Burghes AH (1997) Identification of proximal spinal muscular atrophy carriers and patients by analysis of SMNT and SMNC gene copy number. Am J Hum Genet 60(6):1411–1422. doi:10.1086/515465
McWhorter ML, Monani UR, Burghes AHM, Beattie CE (2003) Knockdown of the survival motor neuron (Smn) protein in zebrafish causes defects in motor axon outgrowth and pathfinding. J Cell Biol 162(5):919–931. doi:10.1083/jcb.200303168
Meister G, Fischer U (2002) Assisted RNP assembly: SMN and PRMT5 complexes cooperate in the formation of spliceosomal UsnRNPs. EMBO J 21(21):5853–5863. doi:10.1093/emboj/cdf585
Meister G, Bühler D, Laggerbauer B, Zobawa M, Lottspeich F, Fischer U (2000) Characterization of a nuclear 20S complex containing the survival of motor neurons (SMN) protein and a specific subset of spliceosomal Sm proteins. Hum Mol Genet 9(13):1977–1986
Meister G, Bühler D, Pillai R, Lottspeich F, Fischer U (2001) A multiprotein complex mediates the ATP-dependent assembly of spliceosomal U snRNPs. Nat Cell Biol 3(11):945–949. doi:10.1038/ncb1101-945
Miller N, Shi H, Zelikovich AS, Ma YC (2016) Motor neuron mitochondrial dysfunction in spinal muscular atrophy. Hum Mol Genet 25(16):3395–3406. doi:10.1093/hmg/ddw262
Misteli T, Cáceres JF, Clement JQ, Krainer AR, Wilkinson MF, Spector DL (1998) Serine phosphorylation of SR proteins is required for their recruitment to sites of transcription in vivo. J Cell Biol 143(2):297–307
Monani UR (1999) A single nucleotide difference that alters splicing patterns distinguishes the SMA gene SMN1 from the copy gene SMN2. Hum Mol Genet 8(7):1177–1183. doi:10.1093/hmg/8.7.1177
Monani UR, Sendtner M, Coovert DD, Parsons DW, Andreassi C, Le TT, Jablonka S et al (2000) The human centromeric survival motor neuron gene (SMN2) rescues embryonic lethality in Smn(−/−) mice and results in a mouse with spinal muscular atrophy. Hum Mol Genet 9(3):333–339
Monani UR, Pastore MT, Gavrilina TO, Jablonka S, Le TT, Andreassi C, DiCocco JM et al (2003) A transgene carrying an A2G missense mutation in the SMN gene modulates phenotypic severity in mice with severe (type I) spinal muscular atrophy. J Cell Biol 160(1):41–52. doi:10.1083/jcb.200208079
Murray LM, Beauvais A, Gibeault S, Courtney NL, Kothary R (2015) Transcriptional profiling of differentially vulnerable motor neurons at pre-symptomatic stage in the Smn (2b/−) mouse model of spinal muscular atrophy. Acta Neuropathol Commun 3(1):55. doi:10.1186/s40478-015-0231-1
Nash LA, Burns JK, Chardon JW, Kothary R, Parks RJ (2016) Spinal muscular atrophy: more than a disease of motor neurons? Curr Mol Med 16(9):779–792. doi:10.2174/1566524016666161128113338
Neuenkirchen N, Englbrecht C, Ohmer J, Ziegenhals T, Chari A, Fischer U (2015) Reconstitution of the human U snRNP assembly machinery reveals stepwise Sm protein organization. EMBO J 34(14):1925–1941. doi:10.15252/embj.201490350
Neumann M, Sampathu DM, Kwong LK, Truax AC, Micsenyi MC, Chou TT, Bruce J, Schuck T, Grossman M, Clark CM, McCluskey LF, Miller BL, Masliah E, Mackenzie IR, Feldman H, Feiden W, Kretzschmar HA, Trojanowski JQ, Lee VM (2006) Ubiquitinated TDP-43 in frontotemporal lobar degeneration and amyotrophic lateral sclerosis. Science 314(5796):130–133
Niedzielska E, Smaga I, Gawlik M, Moniczewski A, Stankowicz P, Pera J, Filip M (2016) Oxidative stress in neurodegenerative diseases. Mol Neurobiol 53(6):4094–4125. doi:10.1007/s12035-015-9337-5
Nilsen TW, Graveley BR (2010) Expansion of the eukaryotic proteome by alternative splicing. Nature 463(7280):457–463. doi:10.1038/nature08909
Nobutoki T, Ihara T (2015) Early disruption of neurovascular units and microcirculatory dysfunction in the spinal cord in spinal muscular atrophy type I. Med Hypotheses 85(6):842–845. doi:10.1016/j.mehy.2015.09.028
Obata T, Brown GE, Yaffe MB (2000) MAP kinase pathways activated by stress: the P38 MAPK pathway. Crit Care Med 28(4 Suppl):N67–N77
Ogawa C, Usui K, Aoki M, Ito F, Itoh M, Kai C, Kanamori-Katayama M, Hayashizaki Y, Suzuki H (2007) Gemin2 plays an important role in stabilizing the survival of motor neuron complex. J Biol Chem 282(15):11122–11134. doi:10.1074/jbc.M609297200
Ogawa C, Usui K, Ito F, Itoh M, Hayashizaki Y, Suzuki H (2009) Role of survival motor neuron complex components in small nuclear ribonucleoprotein assembly. J Biol Chem 284(21):14609–14617. doi:10.1074/jbc.M809031200
Otter S, Grimmler M, Neuenkirchen N, Chari A, Sickmann A, Fischer U (2007) A comprehensive interaction map of the human survival of motor neuron (SMN) complex. J Biol Chem 282(8):5825–5833. doi:10.1074/jbc.M608528200
Pan Q, Shai O, Lee LJ, Frey BJ, Blencowe BJ (2008) Deep surveying of alternative splicing complexity in the human transcriptome by high-throughput sequencing. Nat Genet 40(12):1413–1415. doi:10.1038/ng.259
Panas MD, Ivanov P, Anderson P (2016) Mechanistic insights into mammalian stress granule dynamics. J Cell Biol 215(3):313–323
Papp LV, Holmgren A, Khanna KK (2010) Selenium and selenoproteins in health and disease. Antioxid Redox Signal 12(7):793–795. doi:10.1089/ars.2009.2973
Parker SJ, Meyerowitz J, James JL, Liddell JR, Crouch PJ, Kanninen KM, White AR (2012) Endogenous TDP-43 localized to stress granules can subsequently form protein aggregates. Neurochem Int 60(4):415–424. doi:10.1016/j.neuint.2012.01.019
Paronetto MP, Zalfa F, Botti F, Geremia R, Bagni C, Sette C (2006) The nuclear RNA-binding protein Sam68 translocates to the cytoplasm and associates with the polysomes in mouse spermatocytes. Mol Biol Cell 17(1):14–24. doi:10.1091/mbc.E05-06-0548
Parsons DW, McAndrew PE, Iannaccone ST, Mendell JR, Burghes AH, Prior TW (1998) Intragenic telSMN mutations: frequency, distribution, evidence of a founder effect, and modification of the spinal muscular atrophy phenotype by cenSMN copy number. Am J Hum Genet 63(6):1712–1723. doi:10.1086/302160
Passini MA, Bu J, Richards AM, Kinnecom C, Sardi SP, Stanek LM, Hua Y et al (2011) Antisense oligonucleotides delivered to the mouse CNS ameliorate symptoms of severe spinal muscular atrophy. Sci Transl Med 3(72):72ra18. doi:10.1126/scitranslmed.3001777
Patitucci TN, Ebert AD (2016) SMN deficiency does not induce oxidative stress in SMA iPSC-derived astrocytes or motor neurons. Hum Mol Genet 25(3):514–523. doi:10.1093/hmg/ddv489
Pedrotti S, Bielli P, Paronetto MP, Ciccosanti F, Fimia GM, Stamm S, Manley JL, Sette C (2010) The splicing regulator Sam68 binds to a novel exonic splicing silencer and functions in SMN2 alternative splicing in spinal muscular atrophy. EMBO J 29(7):1235–1247. doi:10.1038/emboj.2010.19
Pellizzoni L (2007) Chaperoning ribonucleoprotein biogenesis in health and disease. EMBO Rep 8(4):340–345. doi:10.1038/sj.embor.7400941
Pellizzoni L, Charroux B, Dreyfuss G (1999) SMN mutants of spinal muscular atrophy patients are defective in binding to snRNP proteins. Proc Natl Acad Sci USA 96(20):11167–11172
Pellizzoni L, Yong J, Dreyfuss G (2002) Essential role for the SMN complex in the specificity of snRNP assembly. Science 298(5599):1775–1779. doi:10.1126/science.1074962
Perera ND, Sheean RK, Crouch PJ, White AR, Horne MK, Turner BJ (2016) Enhancing survival motor neuron expression extends lifespan and attenuates neurodegeneration in mutant TDP-43 mice. Hum Mol Genet 25(18):4080–4093. doi:10.1093/hmg/ddw247
Praveen K, Wen Y, Matera AG (2012) A Drosophila model of spinal muscular atrophy uncouples snRNP biogenesis functions of survival motor neuron from locomotion and viability defects. Cell Rep 1(6):624–631. doi:10.1016/j.celrep.2012.05.014
Prior TW (2007) Spinal muscular atrophy diagnostics. J Child Neurol 22(8):952–956. doi:10.1177/0883073807305668
Prior TW, Krainer AR, Hua Y, Swoboda KJ, Snyder PC, Bridgeman SJ, Burghes AHM, Kissel JT (2009) A positive modifier of spinal muscular atrophy in the SMN2 gene. Am J Hum Genet 85(3):408–413. doi:10.1016/j.ajhg.2009.08.002
Qiu H, Lee S, Shang Y, Wang WY, Au KF, Kamiya S, Barmada SJ, Finkbeiner S, Lui H, Carlton CE, Tang AA, Oldham MC, Wang H, Shorter J, Filiano AJ, Roberson ED, Tourtellotte WG, Chen B, Tsai LH, Huang EJ (2014) ALS-associated mutation FUS-R521C causes DNA damage and RNA splicing defects. J Clin Investig 124(3):981–999. doi:10.1172/JCI72723
Rajendra TK, Gonsalvez GB, Walker MP, Shpargel KB, Salz HK, Matera AG (2007) A Drosophila melanogaster model of spinal muscular atrophy reveals a function for SMN in striated muscle. J Cell Biol 176(6):831–841. doi:10.1083/jcb.200610053
Raker VA, Plessel G, Lührmann R (1996) The snRNP core assembly pathway: identification of stable core protein heteromeric complexes and an snRNP subcore particle in vitro. EMBO J 15(9):2256–2269
Raker VA, Hartmuth K, Kastner B, Lührmann R (1999) Spliceosomal U snRNP core assembly: Sm proteins assemble onto an Sm site RNA nonanucleotide in a specific and thermodynamically stable manner. Mol Cell Biol 19(10):6554–6565
Ratti A, Buratti E (2016) Physiological functions and pathobiology of TDP-43 and FUS/TLS proteins. J Neurochem 138(Suppl 1):95–111. doi:10.1111/jnc.13625
Reber S, Stettler J, Filosa G, Colombo M, Jutzi D, Lenzken SC, Schweingruber C, Bruggmann R, Bachi A, Barabino SM, Mühlemann O, Ruepp MD (2016) Minor intron splicing is regulated by FUS and affected by ALS-associated FUS mutants. EMBO J 35(14):1504–1521. doi:10.15252/embj.201593791
Richert JR, Antel JP, Canary JJ, Maxted WC, Groothuis D (1986) Adult onset spinal muscular atrophy with atrophic testes: report of two cases. J Neurol Neurosurg Psychiatry 49(5):606–608
Riessland M, Ackermann B, Förster A, Jakubik M, Hauke J, Garbes L, FritzscheI Mende Y, Blumcke I, Hahnen E, Wirth B (2010) SAHA ameliorates the SMA phenotype in two mouse models for spinal muscular atrophy. Hum Mol Genet 19(8):1492–1506. doi:10.1093/hmg/ddq023
Robbins KL, Glascock JJ, Osman EY, Miller MR, Lorson CL (2014) Defining the therapeutic window in a severe animal model of spinal muscular atrophy. Hum Mol Genet 23(17):4559–4568. doi:10.1093/hmg/ddu169
Rochette CF, Surh LC, Ray PN, McAndrew PE, Prior TW, Burghes AH, Vanasse M, Simard LR (1997) Molecular diagnosis of non-deletion SMA patients using quantitative PCR of SMN exon 7. Neurogenetics 1(2):141–147
Rossoll W, Jablonka S, Andreassi C, Kröning A-K, Karle K, Monani UR, Sendtner M (2003) Smn, the spinal muscular atrophy-determining gene product, modulates axon growth and localization of beta-actin mRNA in growth cones of motoneurons. J Cell Biol 163(4):801–812. doi:10.1083/jcb.200304128
Sahashi K, Hua Y, Ling KKY, Hung G, Rigo F, Horev G, Katsuno M et al (2012) TSUNAMI: an antisense method to phenocopy splicing-associated diseases in animals. Genes Dev 26(16):1874–1884. doi:10.1101/gad.197418.112
Saltzman AL, Pan Q, Blencowe BJ (2011) Regulation of alternative splicing by the core spliceosomal machinery. Genes Dev 25(4):373–384. doi:10.1101/gad.2004811
Sanchez G, Dury AY, Murray LM, Biondi O, Tadesse H, El Fatimy R, Kothary R, Charbonnier F, Khandjian EW, Côté J (2013) A novel function for the survival motoneuron protein as a translational regulator. Hum Mol Genet 22(4):668–684. doi:10.1093/hmg/dds474
Saxena S, Caroni P (2011) Selective neuronal vulnerability in neurodegenerative diseases: from stressor thresholds to degeneration. Neuron 71(1):35–48. doi:10.1016/j.neuron.2011.06.031
Schrank B, Götz R, Gunnersen JM, Ure JM, Toyka KV, Smith AG, Sendtner M (1997) Inactivation of the survival motor neuron gene, a candidate gene for human spinal muscular atrophy, leads to massive cell death in early mouse embryos. Proc Natl Acad Sci USA 94(18):9920–9925
Scotti MM, Swanson MS (2016) RNA mis-splicing in disease. Nat Rev Genet 17(1):19–32. doi:10.1038/nrg.2015.3
Seng CO, Magee C, Young PJ, Lorson CL, Allen JP (2015) The SMN structure reveals its crucial role in snRNP assembly. Hum Mol Genet 24(8):2138–2146. doi:10.1093/hmg/ddu734
Seo J, Singh NN, Ottesen EW, Sivanesan S, Shishimorova M, Singh RN (2016) Oxidative stress triggers body-wide skipping of multiple exons of the spinal muscular atrophy gene. PLoS One 11(4):e0154390. doi:10.1371/journal.pone.0154390
Shababi M, Lorson CL, Rudnik-Schöneborn SS (2014) Spinal muscular atrophy: a motor neuron disorder or a multi-organ disease? J Anat 224(1):15–28. doi:10.1111/joa.12083
Sharma A, Lambrechts A, Hao LT, Le TT, Sewry CA, Ampe C, Burghes AHM, Morris GE (2005) A role for complexes of survival of motor neurons (SMN) protein with gemins and profilin in neurite-like cytoplasmic extensions of cultured nerve cells. Exp Cell Res 309(1):185–197. doi:10.1016/j.yexcr.2005.05.014
Shpargel KB, Matera AG (2005) Gemin proteins are required for efficient assembly of Sm-class ribonucleoproteins. Proc Natl Acad Sci USA 102(48):17372–17377. doi:10.1073/pnas.0508947102
Singh NK, Singh NN, Androphy EJ, Singh RN (2006a) Splicing of a critical exon of human survival motor neuron is regulated by a unique silencer element located in the last intron. Mol Cell Biol 26(4):1333–1346. doi:10.1128/MCB.26.4.1333-1346.2006
Singh NK, Singh NN, Androphy EJ, Singh RN (2006b) Splicing of a critical exon of human survival motor neuron is regulated by a unique silencer element located in the last intron. Mol Cell Biol 26(4):1333–1346. doi:10.1128/MCB.26.4.1333-1346.2006
Singh NN, Lee BM, Singh RN (2015) Splicing regulation in spinal muscular atrophy by an RNA structure formed by long-distance interactions. Ann NY Acad Sci 1341:176–187. doi:10.1111/nyas.12727
Singh RN, Howell MD, Ottesen EW, Singh NN (2017a) Diverse role of survival motor neuron protein. Biochim Biophys Acta 1860(3):299–315. doi:10.1016/j.bbagrm.2016.12.008
Singh NN, Howell MD, Singh RN (2017b) Transcriptional and splicing regulation of spinal muscular atrophy genes. In: Sumner CJ, Paushkin S, Ko C-P (eds) Spinal muscular atrophy. Academic, Cambridge, Massachusetts, pp 75–97
Somers E, Stencel Z, Wishart TM, Gillingwater TH, Parson SH (2012) Density, calibre and ramification of muscle capillaries are altered in a mouse model of severe spinal muscular atrophy. Neuromuscul Disord NMD 22(5):435–442. doi:10.1016/j.nmd.2011.10.021
Somers E, Lees RD, Hoban K, Sleigh JN, Zhou H, Muntoni F, Talbot K, Gillingwater TH, Parson SH (2016) Vascular defects and spinal cord hypoxia in spinal muscular atrophy. Ann Neurol 79(2):217–230. doi:10.1002/ana.24549
Speert DP (2006) Bacterial infections of the lung in normal and immunodeficient patients. Novartis Found Symp 279:42–51 (disussion 51–5–216–9)
Stamm S (2008) Regulation of alternative splicing by reversible protein phosphorylation. J Biol Chem 283(3):1223–1227. doi:10.1074/jbc.R700034200
Stoilov P, Daoud R, Nayler O, Stamm S (2004) Human Tra2-Beta1 autoregulates its protein concentration by influencing alternative splicing of its pre-mRNA. Hum Mol Genet 13(5):509–524. doi:10.1093/hmg/ddh051
Sumpter V, Kahrs A, Fischer U, Kornstädt U, Lührmann R (1992) In vitro reconstitution of U1 and U2 snRNPs from isolated proteins and snRNA. Mol Biol Rep 16(4):229–240
Sun Y, Grimmler M, Schwarzer V, Schoenen F, Fischer U, Wirth B (2005) Molecular and functional analysis of intragenic SMN1 mutations in patients with spinal muscular atrophy. Hum Mutat 25(1):64–71. doi:10.1002/humu.20111
Sun S, Ling SC, Qiu J, Albuquerque CP, Zhou Y, Tokunaga S, Li H, Qiu H, Bui A, Yeo GW, Huang EJ, Eggan K, Zhou H, Fu XD, Lagier-Tourenne C, Cleveland DW (2015) ALS-causative mutations in FUS/TLS confer gain and loss of function by altered association with SMN and U1-snRNP. Nat Commun 6:6171. doi:10.1038/ncomms7171
Talbot K, Ponting CP, Theodosiou AM, Rodrigues NR, Surtees R, Mountford R, Davies KE (1997) Missense mutation clustering in the survival motor neuron gene: a role for a conserved tyrosine and glycine rich region of the protein in RNA metabolism? Hum Mol Genet 6(3):497–500
Temsamani J, Rhoadhouse M, Pederson T (1991) The U2 small nuclear ribonucleoprotein particle associates with nuclear factors in a pre-mRNA independent reaction. J Biol Chem 266(30):20356–20362
Tsai L-K, Chen C-L, Tsai Y-C, Ting C-H, Chien Y-H, Lee N-C, Hwu W-L (2016) Hypothermia improves disease manifestations in SMA mice via SMN augmentation. Hum Mol Genet 25(4):631–641. doi:10.1093/hmg/ddv500
Tsalikis J, Tattoli I, Ling A, Sorbara MT, Croitoru DO, Philpott DJ, Girardin SE (2015) Intracellular bacterial pathogens trigger the formation of U small nuclear RNA bodies (U bodies) through metabolic stress induction. J Biol Chem 290(34):20904–20918. doi:10.1074/jbc.M115.659466
Turner BJ, Parkinson NJ, Davies KE, Talbot K (2009) Survival motor neuron deficiency enhances progression in an amyotrophic lateral sclerosis mouse model. Neurobiol Dis 34(3):511–517. doi:10.1016/j.nbd.2009.03.005
Turunen JJ, Niemelä EH, Verma B, Frilander MJ (2013) The significant other: splicing by the minor spliceosome. Wiley Interdiscip Rev RNA 4(1):61–76. doi:10.1002/wrna.1141
van der Houven van Oordt W, Diaz-Meco MT, Lozano J, Krainer AR, Moscat J, Cáceres JF (2000) The MKK(3/6)-P38-signaling cascade alters the subcellular distribution of hnRNP A1 and modulates alternative splicing regulation. J Cell Biol 149(2):307–316
Vance C, Rogelj B, Hortobágyi T, De Vos KJ, Nishimura AL, Sreedharan J, Hu X, Smith B, Ruddy D, Wright P, Ganesalingam J, Williams KL, Tripathi V, Al-Saraj S, Al-Chalabi A, Leigh PN, Blair IP, Nicholson G, de Belleroche J, Gallo JM, Miller CC, Shaw CE (2009) Mutations in FUS, an RNA processing protein, cause familial amyotrophic lateral sclerosis type 6. Science 323(5918):1208–1211. doi:10.1126/science.1165942
Veldink JH, Kalmijn S, Van der Hout AH, Lemmink HH, Groeneveld GJ, Lummen C, Scheffer H, Wokke JH, Van den Berg LH (2005) SMN genotypes producing less SMN protein increase susceptibility to and severity of sporadic ALS. Neurology 65(6):820–825
Vezain M, Saugier-Veber P, Goina E, Touraine R, Manel V, Toutain A, Fehrenbach S et al (2010) A rare SMN2 variant in a previously unrecognized composite splicing regulatory element induces exon 7 inclusion and reduces the clinical severity of spinal muscular atrophy. Hum Mutat 31(1):E1110–E1125. doi:10.1002/humu.21173
Wan L, Battle DJ, Yong J, Gubitz AK, Kolb SJ, Wang J, Dreyfuss G (2005) The survival of motor neurons protein determines the capacity for snRNP assembly: biochemical deficiency in spinal muscular atrophy. Mol Cell Biol 25(13):5543–5551. doi:10.1128/MCB.25.13.5543-5551.2005
Wan L, Ottinger E, Cho S, Dreyfuss G (2008) Inactivation of the SMN complex by oxidative stress. Mol Cell 31(2):244–254. doi:10.1016/j.molcel.2008.06.004
Wang CH, Papendick BD, Bruinsma P, Day JK (1998) Identification of a novel missense mutation of the SMN(T) gene in two siblings with spinal muscular atrophy. Neurogenetics 1(4):273–276
Wang ET, Sandberg R, Luo S, Khrebtukova I, Zhang L, Mayr C, Kingsmore SF, Schroth GP, Burge CB (2008) Alternative isoform regulation in human tissue transcriptomes. Nature 456(7221):470–476. doi:10.1038/nature07509
Wang ZB, Zhang X, Li XJ (2013) Recapitulation of spinal motor neuron-specific disease phenotypes in a human cell model of spinal muscular atrophy. Cell Res 23(3):378–393. doi:10.1038/cr.2012.166
Winkler C, Eggert C, Gradl D, Meister G, Giegerich M, Wedlich D, Laggerbauer B, Fischer U (2005) Reduced U snRNP assembly causes motor axon degeneration in an animal model for spinal muscular atrophy. Genes Dev 19(19):2320–2330. doi:10.1101/gad.342005
Workman E, Saieva L, Carrel TL, Crawford TO, Liu D, Lutz C, Beattie CE, Pellizzoni L, Burghes AHM (2009) A SMN missense mutation complements SMN2 restoring snRNPs and rescuing SMA mice. Hum Mol Genet 18(12):2215–2229. doi:10.1093/hmg/ddp157
Xu CC, Denton KR, Wang ZB, Zhang X, Li XJ (2016) Abnormal mitochondrial transport and morphology as early pathological changes in human models of spinal muscular atrophy. Dis Model Mech 9(1):39–49. doi:10.1242/dmm.021766
Yamazaki T, Chen S, Yu Y, Yan B, Haertlein TC, Carrasco MA, Tapia JC, Zhai B, Das R, Lalancette-Hebert M, Sharma A, Chandran S, Sullivan G, Nishimura AL, Shaw CE, Gygi SP, Shneider NA, Maniatis T, Reed R (2012) FUS-SMN protein interactions link the motor neuron diseases ALS and SMA. Cell Rep 2(4):799–806. doi:10.1016/j.celrep.2012.08.025
Yasuda K, Clatterbuck-Soper SF, Jackrel ME, Shorter J, Mili S (2017) FUS inclusions disrupt RNA localization by sequestering kinesin-1 and inhibiting microtubule detyrosination. J Cell Biol 216(4):1015–1034. doi:10.1083/jcb.201608022
Young PJ, Didonato CJ, Hu D, Kothary R, Androphy EJ, Lorson CL (2002) SRp30c-dependent stimulation of survival motor neuron (SMN) exon 7 inclusion is facilitated by a direct interaction with hTra2 beta 1. Hum Mol Genet 11(5):577–587
Zhang H, Xing L, Rossoll W, Wichterle H, Singer RH, Bassell GJ (2006) Multiprotein complexes of the survival of motor neuron protein SMN with Gemins traffic to neuronal processes and growth cones of motor neurons. J Neurosci 26(33):8622–8632. doi:10.1523/JNEUROSCI.3967-05.2006
Zhang Z, Lotti F, Dittmar K, Younis I, Wan L, Kasim M, Dreyfuss G (2008) SMN deficiency causes tissue-specific perturbations in the repertoire of snRNAs and widespread defects in splicing. Cell 133(4):585–600. doi:10.1016/j.cell.2008.03.031
Zou T, Yang X, Pan D, Huang J, Sahin M, Zhou J (2011) SMN deficiency reduces cellular ability to form stress granules, sensitizing cells to stress. Cell Mol Neurobiol 31(4):541–550. doi:10.1007/s10571-011-9647-8
Acknowledgements
Work in the authors’ lab has been funded by National Institute of Neurological Diseases and Stroke (R21-NS084187 to DSC and F31-NS079032 to CED). The authors would like to thank Ashlee Smith for topic discussions, critical reading of the manuscript draft and help with reference management during the writing process.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Rights and permissions
About this article
Cite this article
Dominguez, C.E., Cunningham, D. & Chandler, D.S. SMN regulation in SMA and in response to stress: new paradigms and therapeutic possibilities. Hum Genet 136, 1173–1191 (2017). https://doi.org/10.1007/s00439-017-1835-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00439-017-1835-2