Abstract
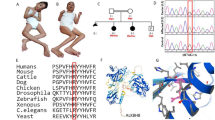
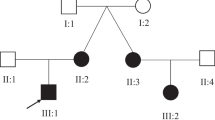
Intellectual disability is a common and highly heterogeneous disorder etiologically. In a multiplex consanguineous family, we applied autozygosity mapping and exome sequencing and identified a novel homozygous truncating mutation in PUS3 that fully segregates with the intellectual disability phenotype. Consistent with the known role of Pus3 in isomerizing uracil to pseudouridine at positions 38 and 39 in tRNA, we found a significant reduction in this post-transcriptional modification of tRNA in patient cells. Our finding adds to a growing list of intellectual disability disorders that are caused by perturbation of various tRNA modifications, which highlights the sensitivity of the brain to these highly conserved processes.


Similar content being viewed by others
References
Abbasi-Moheb L, Mertel S, Gonsior M, Nouri-Vahid L, Kahrizi K, Cirak S, Wieczorek D, Motazacker MM, Esmaeeli-Nieh S, Cremer K (2012) Mutations in NSUN2 cause autosomal-recessive intellectual disability. Am J Hum Genet 90:847–855
Alazami AM, Hijazi H, Al-Dosari MS, Shaheen R, Hashem A, Aldahmesh MA, Mohamed JY, Kentab A, Salih MA, Awaji A (2013) Mutation in ADAT3, encoding adenosine deaminase acting on transfer RNA, causes intellectual disability and strabismus. J Med Genet 50:425–430
Alazami AM, Patel N, Shamseldin HE, Anazi S, Al-Dosari MS, Alzahrani F, Hijazi H, Alshammari M, Aldahmesh MA, Salih MA (2015) Accelerating novel candidate gene discovery in neurogenetic disorders via whole-exome sequencing of prescreened multiplex consanguineous families. Cell Rep 10:148–161
Alkuraya FS (2012) Discovery of rare homozygous mutations from studies of consanguineous pedigrees. Current protocols in human genetics: 6.12. 1–6.12. 13
Alkuraya FS (2013) The application of next-generation sequencing in the autozygosity mapping of human recessive diseases. Hum Genet 132:1197–1211
Antonellis A, Green ED (2008) The role of aminoacyl-tRNA synthetases in genetic diseases. Annu Rev Genomics Hum Genet 9:87–107
Antonellis A, Ellsworth RE, Sambuughin N, Puls I, Abel A, Lee-Lin S-Q, Jordanova A, Kremensky I, Christodoulou K, Middleton LT (2003) Glycyl tRNA synthetase mutations in Charcot-Marie-Tooth disease type 2D and distal spinal muscular atrophy type V. Am J Hum Gen 72:1293–1299
Association AP (2013) Diagnostic and statistical manual of mental disorders (DSM-5®). American Psychiatric Pub
Auffinger P, Westhof E (1998) Effects of pseudouridylation on tRNA hydration and dynamics: a theoretical approach. In: Grosjean H, Benne R (eds) Modification and editing of RNA. American Society for Microbiology, Washington, DC, pp 103–112
Bekaert M, Rousset J-P (2005) An extended signal involved in eukaryotic—1 frameshifting operates through modification of the E site tRNA. Mol Cell 17:61–68
Blaby IK, Majumder M, Chatterjee K, Jana S, Grosjean H, de Crécy-Lagard V, Gupta R (2011) Pseudouridine formation in archaeal RNAs: the case of Haloferax volcanii. RNA 17:1367–1380
Brooks SS, Wall AL, Golzio C, Reid DW, Kondyles A, Willer JR, Botti C, Nicchitta CV, Katsanis N, Davis EE (2014) A novel ribosomopathy caused by dysfunction of RPL10 disrupts neurodevelopment and causes X-linked microcephaly in humans. Genetics 198:723–733
Bykhovskaya Y, Casas K, Mengesha E, Inbal A, Fischel-Ghodsian N (2004) Missense mutation in pseudouridine synthase 1 (PUS1) causes mitochondrial myopathy and sideroblastic anemia (MLASA). Am J Hum Genet 74:1303–1308
Cantara WA, Crain PF, Rozenski J, McCloskey JA, Harris KA, Zhang X, Vendeix FA, Fabris D, Agris PF (2011) The RNA modification database, RNAMDB: 2011 update. Nucleic Acids Res 39:D195–D201. doi:10.1093/nar/gkq1028
Carbone MLA, Solinas M, Sora S, Panzeri L (1991) A gene tightly linked to CEN6 is important for growth of Saccharomyces cerevisiae. Curr Genet 19:1–8
Chang GW, Roth JR, Ames BN (1971) Histidine regulation in Salmonella typhimurium VIII. Mutations of the hisT gene. J Bacteriol 108:410–414
Charette M, Gray MW (2000) Pseudouridine in RNA: what, where, how, and why. IUBMB Life 49:341–351
Chen J, Patton JR (2000) Pseudouridine synthase 3 from mouse modifies the anticodon loop of tRNA. Biochemistry 39:12723–12730
de Ligt J, Willemsen MH, van Bon BW, Kleefstra T, Yntema HG, Kroes T, Vulto-van Silfhout AT, Koolen DA, de Vries P, Gilissen C (2012) Diagnostic exome sequencing in persons with severe intellectual disability. N Engl J Med 367:1921–1929
El‐Hattab AW, Saleh MA, Hashem A, Al‐Owain M, Asmari AA, Rabei H, Abdelraouf H, Hashem M, Alazami AM, Patel N, Shaheen R, Faqeih EA, Alkuraya FS (2016) ADAT3-related intellectual disability: further delineation of the phenotype. Am J Med Genet A. doi:10.1002/ajmg.a.37578
Fahiminiya S, Almuriekhi M, Nawaz Z, Staffa A, Lepage P, Ali R, Hashim L, Schwartzentruber J, Abu Khadija K, Zaineddin S (2014) Whole exome sequencing unravels disease-causing genes in consanguineous families in Qatar. Clin Genet 86:134–141
Fernandez-Vizarra E, Berardinelli A, Valente L, Tiranti V, Zeviani M (2007) Nonsense mutation in pseudouridylate synthase 1 (PUS1) in two brothers affected by myopathy, lactic acidosis and sideroblastic anaemia (MLASA). J Med Genet 44:173–180
Freude K, Hoffmann K, Jensen L-R, Delatycki MB, des Portes V, Moser B, Hamel B, van Bokhoven H, Moraine C, Fryns J-P (2004) Mutations in the FTSJ1 gene coding for a novel S-adenosylmethionine–binding protein cause nonsyndromic X-linked mental retardation. Am J Hum Genet 75:305–309
Gerber AP, Keller W (1999) An adenosine deaminase that generates inosine at the wobble position of tRNAs. Science 286:1146–1149
Gilissen C, Hehir-Kwa JY, Thung DT, van de Vorst M, van Bon BW, Willemsen MH, Kwint M, Janssen IM, Hoischen A, Schenck A, Leach R, Klein R, Tearle R, Bo T, Pfundt R, Yntema HG, de Vries BB, Kleefstra T, Brunner HG, Vissers LE, Veltman JA (2014) Genome sequencing identifies major causes of severe intellectual disability. Nature 511:344–347. doi:10.1038/nature13394
Gillis D, Krishnamohan A, Yaacov B, Shaag A, Jackman JE, Elpeleg O (2014) TRMT10A dysfunction is associated with abnormalities in glucose homeostasis, short stature and microcephaly. J Med Genet 51:581–586. doi:10.1136/jmedgenet-2014-102282
Guy MP, Podyma BM, Preston MA, Shaheen HH, Krivos KL, Limbach PA, Hopper AK, Phizicky EM (2012) Yeast Trm7 interacts with distinct proteins for critical modifications of the tRNAPhe anticodon loop. RNA 18:1921–1933. doi:10.1261/rna.035287.112
Guy MP, Shaw M, Weiner CL, Hobson L, Stark Z, Rose K, Kalscheuer VM, Gecz J, Phizicky EM (2015) Defects in tRNA anticodon loop 2′-O-methylation are implicated in nonsyndromic X-linked intellectual disability due to mutations in FTSJ1. Hum Mutat 36:1176–1187
Han L, Kon Y, Phizicky EM (2015) Functional importance of Psi38 and Psi39 in distinct tRNAs, amplified for tRNAGln(UUG) by unexpected temperature sensitivity of the s2U modification in yeast. RNA 21:188–201. doi:10.1261/rna.048173.114
Heiss NS, Knight SW, Vulliamy TJ, Klauck SM, Wiemann S, Mason PJ, Poustka A, Dokal I (1998) X-linked dyskeratosis congenita is caused by mutations in a highly conserved gene with putative nucleolar functions. Nat Genet 19:32–38
Hur S, Stroud RM (2007) How U38, 39, and 40 of many tRNAs become the targets for pseudouridylation by TruA. Mol Cell 26:189–203
Igoillo-Esteve M, Genin A, Lambert N, Desir J, Pirson I, Abdulkarim B, Simonis N, Drielsma A, Marselli L, Marchetti P, Vanderhaeghen P, Eizirik DL, Wuyts W, Julier C, Chakera AJ, Ellard S, Hattersley AT, Abramowicz M, Cnop M (2013) tRNA methyltransferase homolog gene TRMT10A mutation in young onset diabetes and primary microcephaly in humans. PLoS Genet 9:e1003888. doi:10.1371/journal.pgen.1003888
Jackman JE, Montange RK, Malik HS, Phizicky EM (2003) Identification of the yeast gene encoding the tRNA m1G methyltransferase responsible for modification at position 9. RNA 9:574–585
Kammen HO, Marvel CC, Hardy L, Penhoet EE (1988) Purification, structure, and properties of Escherichia coli tRNA pseudouridine synthase I. J Biol Chem 263:2255–2263
Karaca E, Harel T, Pehlivan D, Jhangiani SN, Gambin T, Akdemir ZC, Gonzaga-Jauregui C, Erdin S, Bayram Y, Campbell IM (2015) Genes that affect brain structure and function identified by rare variant analyses of mendelian neurologic disease. Neuron 88:499–513
Khan MA, Rafiq MA, Noor A, Hussain S, Flores JV, Rupp V, Vincent AK, Malli R, Ali G, Khan FS (2012) Mutation in NSUN2, which encodes an RNA methyltransferase, causes autosomal-recessive intellectual disability. Am J Hum Genet 90:856–863
Lecointe F, Simos G, Sauer A, Hurt EC, Motorin Y, Grosjean H (1998) Characterization of yeast protein Deg1 as pseudouridine synthase (Pus3) catalyzing the formation of Ψ38 and Ψ39 in tRNA anticodon loop. J Biol Chem 273:1316–1323
Lecointe F, Namy O, Hatin I, Simos G, Rousset J-P, Grosjean H (2002) Lack of pseudouridine 38/39 in the anticodon arm of yeast cytoplasmic tRNA decreases in vivo recoding efficiency. J Biol Chem 277:30445–30453
Machnicka MA, Milanowska K, Oglou OO, Purta E, Kurkowska M, Olchowik A, Januszewski W, Kalinowski S, Dunin-Horkawicz S, Rother KM (2013) MODOMICS: a database of RNA modification pathways—2013 update. Nucl Acids Res 41:D262–D267
Martinez FJ, Lee JH, Lee JE, Blanco S, Nickerson E, Gabriel S, Frye M, Al-Gazali L, Gleeson JG (2012) Whole exome sequencing identifies a splicing mutation in NSUN2 as a cause of a Dubowitz-like syndrome. J Med Genet 49:380–385. doi:10.1136/jmedgenet-2011-100686
Maulik PK, Mascarenhas MN, Mathers CD, Dua T, Saxena S (2011) Prevalence of intellectual disability: a meta-analysis of population-based studies. Res Dev Disabil 32:419–436
Miller DT, Adam MP, Aradhya S, Biesecker LG, Brothman AR, Carter NP, Church DM, Crolla JA, Eichler EE, Epstein CJ (2010) Consensus statement: chromosomal microarray is a first-tier clinical diagnostic test for individuals with developmental disabilities or congenital anomalies. Am J Hum Genet 86:749–764
Mochizuki Y, He J, Kulkarni S, Bessler M, Mason PJ (2004) Mouse dyskerin mutations affect accumulation of telomerase RNA and small nucleolar RNA, telomerase activity, and ribosomal RNA processing. Proc Natl Acad Sci USA 101:10756–10761
Ramser J, Winnepenninckx B, Lenski C, Errijgers V, Platzer M, Schwartz C, Meindl A, Kooy R (2004) A splice site mutation in the methyltransferase gene FTSJ1 in Xp11. 23 is associated with non-syndromic mental retardation in a large Belgian family (MRX9). J Med Genet 41:679–683
Rauch A, Wieczorek D, Graf E, Wieland T, Endele S, Schwarzmayr T, Albrecht B, Bartholdi D, Beygo J, Di Donato N (2012) Range of genetic mutations associated with severe non-syndromic sporadic intellectual disability: an exome sequencing study. Lancet 380:1674–1682
Saudi Mendeliome Group (2015) Comprehensive gene panels provide advantages over clinical exome sequencing for Mendelian diseases. Genome Biol 16:134. doi:10.1186/s13059-015-0693-2
Shaheen R, Abdel-Salam GM, Guy MP, Alomar R, Abdel-Hamid MS, Afifi HH, Ismail SI, Emam BA, Phizicky EM, Alkuraya FS (2015) Mutation in WDR4 impairs tRNA m7G46 methylation and causes a distinct form of microcephalic primordial dwarfism. Genome Biol 16:1–11
Singer CE, Smith GR, Cortese R, Ames BN (1972) Mutant tRNA His ineffective in repression and lacking two pseudouridine modifications. Nat New Biol 238:72–74
Torres AG, Batlle E, de Pouplana LR (2014) Role of tRNA modifications in human diseases. Trends Mol Med 20:306–314
Tsui H, Arps P, Connolly D, Winkler M (1991) Absence of hisT-mediated tRNA pseudouridylation results in a uracil requirement that interferes with Escherichia coli K-12 cell division. J Bacteriol 173:7395–7400
Vissers LE, de Ligt J, Gilissen C, Janssen I, Steehouwer M, de Vries P, van Lier B, Arts P, Wieskamp N, del Rosario M (2010) A de novo paradigm for mental retardation. Nat Genet 42:1109–1112
Yao P, Fox PL (2013) Aminoacyl-tRNA synthetases in medicine and disease. EMBO Mol Med 5:332–343
Acknowledgments
We thank the family for their enthusiastic participation. We also thank the Sequencing and Genotyping Facilities at KFSHRC for their technical help. This work was supported in part by KACST Grant 13-BIO1113-20 (FSA) and National Institutes of Health Grant GM052347 (EMP).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
None.
Additional information
R. Shaheen, L. Han, E. Faqeih authors have contributed equally.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Shaheen, R., Han, L., Faqeih, E. et al. A homozygous truncating mutation in PUS3 expands the role of tRNA modification in normal cognition. Hum Genet 135, 707–713 (2016). https://doi.org/10.1007/s00439-016-1665-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00439-016-1665-7