Abstract
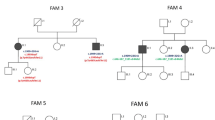
Autosomal recessive spastic ataxias are a heterogeneous group of neurodegenerative diseases usually characterized by the early onset of cerebellar and pyramidal signs. With the collaboration of the clinical European and Mediterranean SPATAX network, we identified 15 families with 34 affected members presenting with ataxia and pyramidal signs or spasticity that were not linked to the ARSACS locus on chromosome 13. In an informative consanguineous Moroccan family, we mapped a novel locus, SAX2, to chromosome 17p13. The minimal linked interval lies in a region of 6.1 cM flanked by markers D17S1845/1583 and D17S1854 (Z max = 3.21). Three of the remaining 14 families were also possibly linked to SAX2. The overall clinical picture in nine patients was cerebellar ataxia with pyramidal signs and/or spasticity. Onset occurred before the age of 15 years in two families and in adulthood in the other two. Interestingly, in the largest SAX2 family, the presenting clinical sign was dysarthria, which is not common in other forms of inherited ataxias or spastic ataxias, whereas gait difficulties appeared later. Most cases also showed fasciculations suggesting that both lower and upper motor neurons are involved in the disease process. No mutations were found in the coding exons of KIF1C, ARRB2 and ANKFY1, three genes in the candidate region.



Similar content being viewed by others
Reference
Ben Hamida M, Attia-Romdhane N, Hentati F, Triki C (1993) Early onset ataxias in Tunisia. Intrafamilial heterogeneity Adv Neurol 61:105–114
Ben Hamida M, Attia-Romdhane N, Triki CH, Oueslati S, Hentati F (1991) (Clinical and genetic analysis of 188 families with spinocerebellar degeneration. Friedreich’s disease and P. Marie’s hereditary ataxias) [French]. Rev Neurol (Paris) 147:798–808
Caldecott KW (2003) DNA single-strand break repair and spinocerebellar ataxia. Cell 112:7–10
Campuzano V, Montermini L, Molto MD, Pianese L, Cossee M, Cavalcanti F, Monros E, Rodius F, Duclos F, Monticelli A, Zara F, Canizares J, Koutnikova H, Bidichandani SI, Gellera C, Brice A, Trouillas P, De Michele G, Filla A, De Frutos R, Palau F, Patel PI, Di Donato S, Mandel J-L, Cocozza S, Koenig M, Pandolfo M (1996) Friedreich’s ataxia: autosomal recessive disease caused by an intronic GAA triplet repeat expansion. Science 271:1423–1427
Cottingham RW, Jr. Idury RM, Schaffer AA (1993) Faster sequential genetic linkage computations. Am J Hum Genet 53:252–263
Coutinho P, Barros J, Zemmouri R, Guimaraes J, Alves C, Chorao R, Lourenco E, Ribeiro P, Loureiro JL, Santos JV, Hamri A, Paternotte C, Hazan J, Silva MC, Prud’homme JF, Grid D (1999) Clinical heterogeneity of autosomal recessive spastic paraplegias: analysis of 106 patients in 46 families. Arch Neurol 56:943–949
Crosby AH, Proukakis C (2002) Is the transportation highway the right road for hereditary spastic paraplegia? Am J Hum Genet 71:1009–1016
Engert JC, Berube P, Mercier J, Dore C, Lepage P, Ge B, Bouchard JP, Mathieu J, Melancon SB, Schalling M, Lander ES, Morgan K, Hudson TJ, Richter A (2000) ARSACS, a spastic ataxia common in northeastern Quebec, is caused by mutations in a new gene encoding an 11.5-kb ORF. Nat Genet 24:120–125
Fink JK (2006) Hereditary spastic paraplegia. Curr Neurol Neurosci Rep 6:65–76
Gomez CM (2004) ARSACS goes global. Neurology 62:10–11
Gudbjartsson DF, Jonasson K, Frigge ML, Kong A (2000) Allegro, a new computer program for multipoint linkage analysis. Nature Genet 25:12–13
Harding AE (1983) Classification of the hereditary ataxias and paraplegias. Lancet 1:1151–1155
Koenig M (2003) Rare forms of autosomal recessive neurodegenerative ataxia. Semin Pediatr Neurol 10:183–192
Kong X, Matise TC (2005) MAP-O-MAT: internet-based linkage mapping. Bioinformatics 21:557–559
Moreira MC, Barbot C, Tachi N, Kozuka N, Uchida E, Gibson T, Mendonca P, Costa M, Barros J, Yanagisawa T, Watanabe M, Ikeda Y, Aoki M, Nagata T, Coutinho P, Sequeiros J, Koenig M (2001) The gene mutated in ataxia-ocular apraxia 1 encodes the new HIT/Zn-finger protein aprataxin. Nature Genet 29:189–193
Moreira MC, Klur S, Watanabe M, Nemeth AH, Le Ber I, Moniz JC, Tranchant C, Aubourg P, Tazir M, Schols L, Pandolfo M, Schulz JB, Pouget J, Calvas P, Shizuka-Ikeda M, Shoji M, Tanaka M, Izatt L, Shaw CE, M’Zahem A, Dunne E, Bomont P, Benhassine T, Bouslam N, Stevanin G, Brice A, Guimaraes J, Mendonca P, Barbot C, Coutinho P, Sequeiros J, Durr A, Warter JM, Koenig M (2004) Senataxin, the ortholog of a yeast RNA helicase, is mutant in ataxia-ocular apraxia 2. Nature Genet 36:225–227
Nikali K, Suomalainen A, Saharinen J, Kuokkanen M, Spelbrink JN, Lonnqvist T, Peltonen L (2005) Infantile onset spinocerebellar ataxia is caused by recessive mutations in mitochondrial proteins Twinkle and Twinky. Hum Mol Genet 14:2981–2990
Ouahchi K, Arita M, Kayden H, Hentati F, Ben HM, Sokol R, Arai H, Inoue K, Mandel J-L, Koenig M (1995) Ataxia with isolated vitamin E deficiency is caused by mutations in the alpha-tocopherol transfer protein. Nature Genet 9:141–145
Richter A, Morgan K, Bouchard JP, Poirier J, Mercier J, Gosselin F, Melancon SB (1993) Clinical and molecular genetic studies on autosomal recessive spastic ataxia of charlevoix-saguenay (ARSACS) (review). Adv Neurol 61:97–103
Stewart GS, Maser RS, Stankovic T, Bressan DA, Kaplan MI, Jaspers NG, Raams A, Byrd PJ, Petrini JH, Taylor AM (1999) The DNA double-strand break repair gene hMRE11 is mutated in individuals with an ataxia-telangiectasia-like disorder. Cell 99:577–587
Takashima H, Boerkoel CF, John J, Saifi GM, Salih MA, Armstrong D, Mao Y, Quiocho FA, Roa BB, Nakagawa M, Stockton DW, Lupski JR (2002) Mutation of TDP1, encoding a topoisomerase I-dependent DNA damage repair enzyme, in spinocerebellar ataxia with axonal neuropathy. Nature Genet 32:267–272
Tallaksen CM, Durr A, Brice A (2001) Recent advances in hereditary spastic paraplegia. Curr Opin Neurol 14:457–463
Thiffault I, Rioux MF, Tetreault M, Jarry J, Loiselle L, Poirier J, Gros-Louis F, Mathieu J, Vanasse M, Rouleau GA, Bouchard JP, Lesage J, Brais B (2006) A new autosomal recessive spastic ataxia associated with frequent white matter changes maps to 2q33–34. Brain 129:2332–2340
Acknowledgments
We thank the members of the European and Mediterranean network dedicated to the Spastic Paraplegias and ATAXias (SPATAX) for recruiting several families. The authors wish to thank the families that participated in this study as well as Ms Nawal Benammar and Drs M. Abada-Bendib, S. Forlani, D. Grid, M. Koenig, I. Leber, J. F. Lemaire, S. Poea, M. Ruberg and P. Vondracek for their help. This study was supported by grants from the French National Institute for Health and Medical Research (INSERM), the Neurogenetic Moroccan Association (AMNG), the Verum Foundation (to A Brice), The French Agency for Research (to AD), the “Pôle de Compétences en Neurogénétique au Maroc” (PCNG) and the GIS/Rare Diseases Institute (to AD and GS). NB and HA received fellowships from the Foundation of Friedreich Ataxia (AFAF, France) and the French Association against Myopathies (AFM, France). SK was the recipient of a fellowship from the Tom-Walhig Foundation (Germany) and the Verum Foundation (Germany).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Naima Bouslam and Ahmed Bouhouche are co-first authors.
Electronic supplementary material
Below is the link to the electronic supplementary material.
439_2007_328_MOESM1_ESM.doc
Supplementary table 1: Pairwise and multipoint LOD scores calculated in all families using an autosomal recessive mode of transmission with 95% penetrance. Alpha (% of unlinked families) and HLOD (homogeneity LOD scores) were calculated with ALLEGRO assuming heterogeneity of linkage. Grey = non-excluded LOD scores (DOC 89 kb)
Rights and permissions
About this article
Cite this article
Bouslam, N., Bouhouche, A., Benomar, A. et al. A novel locus for autosomal recessive spastic ataxia on chromosome 17p. Hum Genet 121, 413–420 (2007). https://doi.org/10.1007/s00439-007-0328-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00439-007-0328-0