Abstract
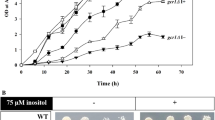
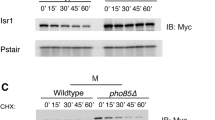
Cdc34, a ubiquitin-conjugating enzyme in Saccharomyces cerevisiae, is required for cell cycle progression. Sic1, an S-phase cyclin-dependent kinase (CDK) inhibitor, is a critical target of Cdc34-mediated ubiquitination. Other essential target protein(s) could be defined since cdc34 sic1 double mutants still arrest in G2 phase. To identify proteins which function in the Cdc34-dependent ubiquitin pathway, a series of extragenic suppressors of the cdc34-1 sic1 double mutations was isolated. One of them was found to be defective in GRR1, which is involved not only in glucose repression but also in G1 cyclin destabilization. However, neither lack of glucose repression nor stabilization of G1 cyclin caused the suppression of cdc34-1 sic1. Conversely, Grr1 overproduction in cdc34-1 sic1 cells impaired colony formation, even at the permissive temperature. A multicopy suppressor, MGO1, which rescued the growth defect associated with Grr1 overproduction was isolated, and found to be identical to SKP1. Furthermore, Grr1 bound Skp1 directly in vitro. These results strongly suggest that Grr1 functions in the ubiquitin pathway through association with Skp1.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 29 May 1997 / Accepted: 4 September 1997
Rights and permissions
About this article
Cite this article
Kishi, T., Seno, T. & Yamao, F. Grr1 functions in the ubiquitin pathway in Saccharomyces cerevisiae through association with Skp1. Mol Gen Genet 257, 143–148 (1998). https://doi.org/10.1007/s004380050633
Issue Date:
DOI: https://doi.org/10.1007/s004380050633