Abstract.
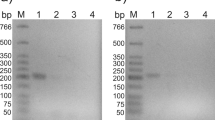
A 5.2-kb NotI DNA fragment isolated from a genomic library of Acremonium chrysogenum by hybridization with a probe internal to the Penicillium chrysogenum lys2 gene, was able to complement an α-aminoadipate reductase-deficient mutant of P. chrysogenum (lysine auxotroph L–G–). Enzyme assays showed that the α-aminoadipate reductase activity was restored in all the transformants tested. The lys2-encoded enzyme catalyzed both the activation and reduction of α-aminoadipic acid to its semialdehyde, as shown by reaction of the product with p-dimethylaminobenzaldehyde. The reaction required NADPH, and was not observed in the presence of NADH. Sequence analysis revealed that the gene encodes a protein with relatively high similarity to members of the superfamily of acyladenylate-forming enzymes. The Lys2 protein contained all nine motifs that are conserved in the adenylating domain of this enzyme family, a peptidyl carrier domain, and a reduction domain. In addition, a new NADP-binding motif located at the N-terminus of the reduction domain that may form a Rossmann-like βαβ-fold has been identified and found to be shared by all known Lys2 proteins. The lys2 gene was mapped to chromosome I (2.2 Mb, the smallest chromosome) of A. chrysogenum C10 (the chromosome that contains the "late" cephalosporin cluster) and is transcribed as a monocistronic 4.5-kb mRNA although at relatively low levels compared with the β-actin gene.
Similar content being viewed by others
Author information
Authors and Affiliations
Corresponding author
Additional information
Electronic Publication
Rights and permissions
About this article
Cite this article
Hijarrubia, M., Aparicio, J., Casqueiro, J. et al. Characterization of the lys2 gene of Acremonium chrysogenum encoding a functional α-aminoadipate activating and reducing enzyme. Mol Gen Genet 264, 755–762 (2001). https://doi.org/10.1007/s004380000364
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s004380000364