Abstract
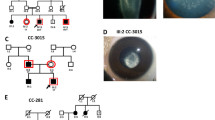
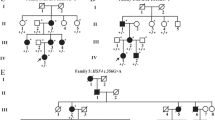
Congenital cataract an opacity of the eye lens is present at birth and results in visual impairment during early childhood. If left untreated, it can lead to permanent blindness. Its prevalence is ten times higher in developing countries like India. Thus, we aimed to investigate the underlying genetic defects in three autosomal dominant congenital cataract (ADCC) families from North India. Detailed family histories were collected, pedigrees drawn followed by slit-lamp examination and lens photography. Mutation screening was performed in the candidate genes for crystallins, connexins, and membrane proteins by Sanger sequencing. Pathogenicity of novel variant was assessed bioinformatically. In an ADCC (CC-3006) family with bilateral membranous cataract and microcornea, a novel change (c.1114C>T;p.P372S) in GJA3 has been detected. In other two ADCC families affected with subcapsular (CC-286) and shrunken membranous hypermature cataract (CC-3014), a nonsense mutation (c.463C>T;p.Q155X) in CRYβB2 and a frameshift deletion (c.590_591delAG;p.E197VfsX22) in CRYβA1/A3 respectively, are observed. These variants segregated completely with the phenotypes in respective families and were absent in their unaffected family members and unrelated controls (tested for novel variant in GJA3). Earlier p.Q155X (CRYβB2) and p.E197VfsX22 (CRYβA1/A3) are reported with entirely different phenotypes. Thus, findings in present study expand the mutation spectrum and phenotypic heterogeneity linked with GJA3, CRYβB2, and CRYβA1/A3 for congenital cataracts. Identifying underlying genetic defects is essential for disease management and appropriate genetic counseling.



Similar content being viewed by others
Data availability
Data available on reasonable request from the corresponding author.
References
Adeli K, Ogbonna G (1990) Rapid purification of human DNA from whole blood for potential application in clinical chemistry laboratories. Clin Chem 36(2):261–264
Andley UP (2007) Crystallins in the eye: function and pathology. Prog Retin Eye Res 26(1):78–98. https://doi.org/10.1016/j.preteyeres.2006.10.003
Banks EA, Toloue MM et al (2009) Connexin mutation that causes dominant congenital cataracts inhibits gap junctions, but not hemichannels, in a dominant negative manner. J Cell Sci 122(Pt 3):378–388. https://doi.org/10.1242/jcs.034124
Bateman JB, Spence MA et al (1986) Genetic linkage analysis of autosomal dominant congenital cataracts. Am J Ophthalmol 101(2):218–225. https://doi.org/10.1016/0002-9394(86)90599-4
Bateman JB, von-Bischhoffshaunsen FRB et al (2007) Gene conversion mutation in crystallin, β-B2 (CRYBB2) in a Chilean Family with autosomal dominant cataract. Ophthalmol 114(3):425–432. https://doi.org/10.1016/j.ophtha.2006.09.013
Brakenhoff RH, Aarts HJ et al (1992) The second human beta B2-crystallin gene is a pseudogene. Exp Eye Res 54(5):803–806. https://doi.org/10.1016/0014-4835(92)90036-r
Carver JA, Aquilina JA et al (1994) Alpha-crystallin: molecular chaperone and protein surfactant. Biochim Biophys Acta 1204(2):195–206. https://doi.org/10.1016/0167-4838(94)90009-4
Caufriez A, Bock D et al (2020) Peptide-based targeting of connexins and pannexins for therapeutic purposes. Expert Opin Drug Discov 15(10):1213–1222. https://doi.org/10.1080/17460441.2020.1773787
Chen C, Sun Q et al (2015) A novel Cx50 (GJA8) p.H277Y mutation associated with autosomal dominant congenital cataract identified with targeted next-generation sequencing. Graefe’s Arch Clin Exp Ophthalmol 253(6):915–924. https://doi.org/10.1007/s00417-015-3019-x
Crabbe MJ, Goode D (1995) Protein folds and functional similarity; the Greek key/immunoglobulin fold. Comput Chem 19(4):343–349
Dermietzel R, Kremer M et al (2000) Molecular and functional diversity of neural connexins in the retina. J Neurosci 20(22):8331–8343. https://doi.org/10.1523/jneurosci.20-22-08331.2000
Francis PJ, Berry V et al (2000) The genetics of childhood cataract. J Med Genet 37(7):481–488. https://doi.org/10.1136/jmg.37.7.481
Ge XL, Zhang Y et al (2014) Identification of a novel GJA8 (Cx50) point mutation causes human dominant congenital cataracts. Sci Rep 4:4121. https://doi.org/10.1038/srep04121
Giepmans BN, Verlaan I et al (2001) Gap junction protein connexin-43 interacts directly with microtubules. Curr Biol 11(17):1364–1368. https://doi.org/10.1016/s0960-9822(01)00424-9
Gill D, Klose R et al (2000) Genetic heterogeneity of the Coppock-like cataract: a mutation in CRYBB2 on chromosome 22q11.2. Invest Ophthalmol Vis Sci 41(1):159–165
Goodenough DA (1992) The crystalline lens. A system networked by gap junctional intercellular communication. Semin Cell Biol 3(1):49–58. https://doi.org/10.1016/s1043-4682(10)80007-8
Grałek M, Kanigowska K et al (2007) Cataract in children–not only an ophthalmological problem. Med Wieku Rozwoj 11(2 Pt 2):227–230
Gupta R, Srivastava K et al (2006) Truncation of motifs III and IV in human lens betaA3-crystallin destabilizes the structure. Biochemistry 45(33):9964–9978. https://doi.org/10.1021/bi060499v
Haargaard B, Wohlfahrt J et al (2004) Incidence and cumulative risk of childhood cataract in a cohort of 2.6 million Danish children. Invest Ophthalmol Vis Sci 45(5):1316–1320. https://doi.org/10.1167/iovs.03-0635
Hansen L, Mikkelsen A et al (2009) Comprehensive mutational screening in a cohort of Danish families with hereditary congenital cataract. Invest Ophthalmol vis Sci 50(7):3291–3303. https://doi.org/10.1167/iovs.08-3149
Hebert C, Stains JP (2013) An intact connexin43 is required to enhance signaling and gene expression in osteoblast-like cells. J Cell Biochem 114(11):2542–2550. https://doi.org/10.1002/jcb.24603
Hejtmancik JF (2008) Congenital cataracts and their molecular genetics. Semin Cell Dev Biol 19(2):134–149. https://doi.org/10.1016/j.semcdb.2007.10.003
Huang B, He W (2010) Molecular characteristics of inherited congenital cataracts. Eur J Med Genet 53(6):347–357. https://doi.org/10.1016/j.ejmg.2010.07.001
Inana G, Piatigorsky J et al (1983) Gene and protein structure of a beta-crystallin polypeptide in murine lens: relationship of exons and structural motifs. Nature 302(5906):310–315. https://doi.org/10.1038/302310a0
Kaur S, Sukhija J et al (2022) Commentary: genetic testing in cases of pediatric cataract. Indian J Ophthalmol 70(7):2623–2624. https://doi.org/10.4103/ijo.IJO_124_22
Kumaraswamy VS, Lindley PF et al (1996) An eye lens protein-water structure: 1.2 A resolution structure of gammaB-crystallin at 150 K. Acta Crystallogr D Biol Crystallogr 52(Pt 4):611–622. https://doi.org/10.1107/S0907444995014302
Litt M, Carrero-Valenzuela R et al (1997) Autosomal dominant cerulean cataract is associated with a chain termination mutation in the human beta-crystallin gene CRYBB2. Hum Mol Genet 6(5):665–668. https://doi.org/10.1093/hmg/6.5.665
Liu BF, Liang JJ (2005) Interaction and biophysical properties of human lens Q155* betaB2-crystallin mutant. Mol vis 11:321–327
Liu YC, Wilkins M et al (2017) Cataracts. Lancet 390(10094):600–612. https://doi.org/10.1016/S0140-6736(17)30544-5
Murthy G, John N et al (2008) Status of pediatric eye care in India. Indian J Ophthalmol 56(6):481–488. https://doi.org/10.4103/0301-4738.42642
Musil LS, Beyer EC et al (1990) Expression of the gap junction protein connexin43 in embryonic chick lens: molecular cloning, ultrastructural localization, and post-translational phosphorylation. J Membr Biol 116(2):163–175. https://doi.org/10.1007/bf01868674
Paul DL, Ebihara L et al (1991) Connexin46, a novel lens gap junction protein, induces voltage-gated currents in nonjunctional plasma membrane of Xenopus oocytes. J Cell Biol 115(4):1077–1089. https://doi.org/10.1083/jcb.115.4.1077
Pfenniger A, Wohlwend A et al (2011) Mutations in connexin genes and disease. Eur J Clin Invest 41(1):103–116. https://doi.org/10.1111/j.1365-2362.2010.02378.x
Rubinos C, Villone K et al (2014) Functional effects of Cx50 mutations associated with congenital cataracts. Am J Physiol Cell Physiol 306(3):C212-220. https://doi.org/10.1152/ajpcell.00098.2013
Saez JC, Berthoud VM et al (2003) Plasma membrane channels formed by connexins: their regulation and functions. Physiol Rev 83(4):1359–1400. https://doi.org/10.1152/physrev.00007.2003
Shiels A, Bennett TM et al (2010) Cat-Map: putting cataract on the map. Mol vis 16:2007–2015
Solebo AL, Teoh L et al (2017) Epidemiology of blindness in children. Arch Dis Child 102(9):853–857. https://doi.org/10.1136/archdischild-2016-310532
Sosinsky GE, Nicholson BJ (2005) Structural organization of gap junction channels. Biochim Biophys Acta 1711(2):99–125. https://doi.org/10.1016/j.bbamem.2005.04.001
Srivastava K, Gupta R et al (2009) Truncated human betaB1-crystallin shows altered structural properties and interaction with human betaA3-crystallin. Biochemistry 48(30):7179–7189. https://doi.org/10.1021/bi900313c
Sun W, Xiao X et al (2011) Mutational screening of six genes in Chinese patients with congenital cataract and microcornea. Mol vis 17:1508–1513
Thomas BC, Minogue PJ et al (2008) Cataracts are caused by alterations of a critical N-terminal positive charge in connexin50. Invest Ophthalmol vis Sci 49(6):2549–2556. https://doi.org/10.1167/iovs.07-1658
Vanita SJR et al (1999) Genetic and segregation analysis of congenital cataract in the Indian population. Clin Genet 56(5):389–393. https://doi.org/10.1034/j.1399-0004.1999.560507.x
Vanita SV et al (2001) A unique form of autosomal dominant cataract explained by gene conversion between beta-crystallin B2 and its pseudogene. J Med Genet 38(6):392–396. https://doi.org/10.1136/jmg.38.6.392
Vidya NG, Rajkumar S et al (2018) Genetic investigation of ocular developmental genes in 52 patients with anophthalmia/microphthalmia. Ophthalmic Genet 39(3):344–352. https://doi.org/10.1080/13816810.2018.1436184
Waterhouse A, Bertoni M et al (2018) SWISS-MODEL: homology modelling of protein structures and complexes. Nucleic Acids Res 46(W1):W296–W303. https://doi.org/10.1093/nar/gky427
Werten PJ, Carver JA et al (1996) The elusive role of the N-terminal extension of beta A3- and beta A1-crystallin. Protein Eng 9(11):1021–1028. https://doi.org/10.1093/protein/9.11.1021
Wride MA (2011) Lens fibre cell differentiation and organelle loss: many paths lead to clarity. Philos Trans R Soc Lond B Biol Sci 366(1568):1219–1233. https://doi.org/10.1098/rstb.2010.0324
Yao K, Tang X et al (2005) Progressive polymorphic congenital cataract caused by a CRYBB2 mutation in a Chinese family. Mol vis 11:758–763
Yi J, Yun J et al (2011) Epidemiology and molecular genetics of congenital cataracts. Int J Ophthalmol 4(4):422–432. https://doi.org/10.3980/j.issn.2222-3959.2011.04.20
Zhang J, Zhang Y et al (2014) Congenital cataracts due to a novel 2-bp deletion in CRYBA1/A3. Mol Med Rep 10(3):1614–1618. https://doi.org/10.3892/mmr.2014.2324
Acknowledgements
The authors are grateful to all the family members for their participation in this study and their kind cooperation. The authors are thankful to the Indian Council of Medical Research (ICMR), New Delhi for providing a research grant (Grant no. F.No. 54/7/2015-BMS) sanctioned to Dr. V. Vanita. This paper is dedicated to the fond memories of our respected teacher Dr. Daljit Singh Ji.
Funding
The authors are thankful to the Indian Council of Medical Research (ICMR), New Delhi for providing a research grant (Grant no. F.No. 54/7/2015-BMS) sanctioned to Dr. V. Vanita.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethics approval and consent to participate
The written informed consent was obtained from each participant or their legal custodians (in the case of minors). The present study was approved by the Institutional Ethics Committee (IEC), Guru Nanak Dev University (GNDU), Amritsar, India consistent with the Declaration of Helsinki.
Additional information
Communicated by Shuhua Xu.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Goyal, S., Singh, R., Singh, J.R. et al. Mutation screening in autosomal dominant congenital cataract families from North India. Mol Genet Genomics 298, 1279–1288 (2023). https://doi.org/10.1007/s00438-023-02053-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00438-023-02053-x