Abstract
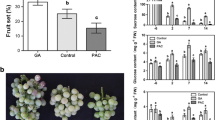
Sugar is crucial for grape berry, whether used for fresh food or wine. However, berry enlargement treatment with forchlorfenuron (N-(2-chloro4-pyridyl)-N′-phenylurea) (CPPU, a synthetic cytokinin) and gibberellin (GA) always had adverse effects on sugar accumulation in some grape varieties, especially CPPU. Therefore exploring the molecular mechanisms behind these adverse effects could provide a foundation for improving or developing technology to mitigate the effects of CPPU/GA treatments for grape growers. In the present study, invertase (INV) family, the key gene controlling sugar accumulation, was identified and characterized on the latest annotated grape genome. Their express pattern, as well as invertase activity and sugar content, were analyzed during grape berry development under CPPU and GA3 treatment to explore the potential role of INV members under berry enlargement treatment in grapes. Eighteen INV genes were identified and divided into two sub-families: 10 neutral INV genes (Vv-A/N-INV1-10) and 8 acid INV genes containing 5 CWINV (VvCWINV1-5) and 3 VIN (VvVIN1-3). At the early development stage, both CPPU and GA3 treatment decreased the hexose level in berries of ‘Pinot Noir’ grape, whereas the activity of three types inverstase (soluble acid INV, insoluble acid INV, and neutral INV) increased. Correspondingly, most of INV members were up-regulated by GA3 /CPPU application at least one sampling time point during early berry development, including VvCWINV1, 2, 3, 4, 5, VvVIN1, 2, 3 and Vv-A/N-INV1, 2, 5, 6, 7, 8, 10. At maturity, the sugar content in CPPU-treated berries is still lower than that in the control. Soluble acid INV and neutral INV, rather than insoluble acid INV, presented lower activity in CPPU-treated berries. Meanwhile, several corresponding genes, such as VvVIN2 and Vv-A/N-INV2, 8, 10 in ripening berries were obviously down-regulated by CPPU treatment. These results suggested that most of INV members could be triggered by berry enlargement treatment during early berry development, whereas VvVINs and Vv-A/N-INVs, but not VvCWINVs, could be the limiting factor resulting in decreased sugar accumulation in CPPU-treated berries at maturity. In conclusion, this study identified the INV family on the latest annotated grape genome and selected several potential members involving in the limit of CPPU on final sugar accumulation in grape berry. These results provide candidate genes for further study of the molecular regulation of CPPU and GA on sugar accumulation in grape.







Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article and its supplementary information files.
References
Acheampong AK, Zheng C, Halaly T, Giacomelli L, Takebayashi Y, Jikumaru Y, Kamiya Y, Lichter A, Or E (2017) Abnormal endogenous repression of GA signaling in a seedless table grape cultivar with high berry growth response to GA application. Front Plant Sci 8:850. https://doi.org/10.3389/fpls.2017.00850
Avigad G, (1982) Sucrose and other disaccharides. In: Loewus FA, Tanner W, eds. Encyclopedia of plant physiology, NS 13A. Plant Carbohydrates I. Berlin, Germany: Springer Verlag: 217–347
Baldet P, Hernould M, Laporte F, Mounet F, Just D, Mouras A, Chevalier C, Rothan C (2006) The expression of cell proliferation-related genes in early developing flowers is affected by a fruit load reduction in tomato plants. J Exp Bot 57:961–970. https://doi.org/10.1093/jxb/erj082
Balibrea Lara ME, Garcia MCG, Fatima T, Ehness R, Lee TK, Proels R, Tanner W, Roitsch T (2004) Extracellular invertase is an essential component of cytokinin-mediated delay of senescence. Plant Cell 16:1276–1287. https://doi.org/10.1105/tpc.018929
Bianchi M, Jensen F, Moriyama M (1991) Management of berry size and color in ‘Flame Seedless. In: Proceedings of the Coachella Valley Table Grape Seminar, Coachella, CA.
Chen Z, Gao K, Su X, Rao P, An X (2015) Genome-Wide Identification of the Invertase Gene Family in Populus. PLoS One 10: e0138540. https://doi.org/10.1371/journal.pone.0138540
Conde C, Silva P, Fontes N, Dias ACP, Tavares RM, Sousa MJ, Gerós H (2007) Biochemical changes throughout grape berry development and fruit and wine quality. Food 1:1–22
Coombe BG (1995) Growth stages of the grapevine: adoption of a system for identifying grapevine growth stages. Aust J Grape Wine Res 1995:104–110. https://doi.org/10.1111/j.1755-0238.1995.tb00086.x
Dahro B, Wang Y, Khan M, Zhang Y, Fang T, Ming R, Li C, Liu JH (2022) Two AT-Hook proteins regulate A/NINV7 expression to modulate sucrose catabolism for cold tolerance in Poncirus trifoliata. New Phytol 235:2331–2349. https://doi.org/10.1111/nph.18304
Davies C, Robinson SP (1996) Sugar accumulation in grape berries. Cloning of two putative vacuolar invertase cDNAs and their expression in grapevine tissues. Plant Physiol 111:275–283. https://doi.org/10.1104/pp.111.1.275
Duan S, Wu Y, Zhang C, Wang L, Song S, Ma C, Zhang C, Wang S, Bondada B, Xu W (2020) Differential regulation of metabolic enzyme activities and physio-anatomical aspects in Ca2+ sufficient and starved grapevine (Vitis vinifera L.). Sci Hortic 272: 109423. https://doi.org/10.1016/j.scienta.2020.109423
Findlay N, Oliver KJ, Nil N, Coombe BG (1987) Solute accumulation by grape pericarp cells iv. perfusion of pericarp apoplast via the pedicel and evidence for xylem malfunction in ripening berries. J Exp Bot 38:668–679. https://doi.org/10.1093/jxb/38.11.1789
Fisher DB, Oparka KJ (1996) Post-phloem transport: principles and problems. J Exp Bot 47:1141–1154. https://doi.org/10.1093/jxb/47.Special_Issue.1141
Gallagher JA, Pollock CJ (1998) Isolation and characterization of a cDNA clone from Lolium temulentum L. encoding for a sucrose hydrolytic enzyme which shows alkaline/neutral invertase activity. J Exp Bot 49:789–795. https://doi.org/10.1093/jxb/49.322.789
Goodstein DM, Shu S, Howson R, Neupane R, Hayes RD, Fazo J, Mitros T, Dirks W, Hellsten U, Putnam N, Rokhsar DS (2012) Phytozome: a comparative platform for green plant genomics. Nucleic Acids Res 40:D1178-1186. https://doi.org/10.1093/nar/gkr944
Hu B, Jin JP, Guo AY, Zhang H, Luo JC, Gao G (2015) GSDS 2.0: an upgraded gene feature visualization server. Bioinformatics 31:1296–1297. https://doi.org/10.1093/bioinformatics/btu817
Ji X, Van den Ende W, Van Laere A, Cheng S, Bennett J (2005) Structure, evolution, and expression of the two invertase gene families of rice. J Mol Evol 60:615–634. https://doi.org/10.1007/s00239-004-0242-1
Juarez-Colunga S, Lopez-Gonzalez C, Morales-Elias NC, Massange-Sanchez JA, Trachsel S, Tiessen A (2018) Genome-wide analysis of the invertase gene family from maize. Plant Mol Biol 97:385–406. https://doi.org/10.1007/s11103-018-0746-5
Koch K (2004) Sucrose metabolism: regulatory mechanisms and pivotal roles in sugar sensing and plant development. Curr Opin Plant Biol 7:235–246. https://doi.org/10.1016/j.pbi.2004.03.014
Lecourieux F, Kappel C, Lecourieux D, Serrano A, Torres E, Arce-Johnson P, Delrot S (2014) An update on sugar transport and signalling in grapevine. J Exp Bot 821. https://doi.org/10.1093/jxb/ert394
Lee DW, Lee SK, Rahman MM, Kim YJ, Zhang D, Jeon JS (2019) The Role of Rice Vacuolar Invertase2 in Seed Size Control. Mol Cells 42: 711–720. https://doi.org/10.14348/molcells.2019.0109
Li YM, Forney C, Bondada B, Leng F, Xie ZS (2020) The Molecular Regulation of Carbon Sink Strength in Grapevine (Vitis vinifera L.). Front Plant Sci 11: 606918. https://doi.org/10.3389/fpls.2020.606918
Li YM, Sun XR, You JL, Li XJ, Li B, Xie ZS (2022) Transcriptional analysis of the early ripening of 'Summer Black' grape in response to abscisic acid treatment. Sci Hortic 299. https://doi.org/10.1016/j.scienta.2022.111054
Lou Y, Gou JY, Xue HW (2007) PIP5K9, an Arabidopsis phosphatidylinositol monophosphate kinase, interacts with a cytosolic invertase to negatively regulate sugar-mediated root growth. Plant Cell 19:163–181. https://doi.org/10.1105/tpc.106.045658
Lu S, Wang J, Chitsaz F, Derbyshire MK, Geer RC, Gonzales NR, Gwadz M, Hurwitz DI, Marchler GH, Song JS, Thanki N, Yamashita RA, Yang M, Zhang D, Zheng C, Lanczycki CJ, Marchler-Bauer A (2020) CDD/SPARCLE: the conserved domain database in 2020. Nucleic Acids Res 48:D265–D268. https://doi.org/10.1093/nar/gkz991
Mothes K, Engelbrecht L (1962) On the activity of a kinetin-like root factor. Life Sci 11:852–857. https://doi.org/10.1016/0024-3205(63)90098-5
Nickell LC (1985) New plant growth regulator increases grape size. Proc Plant Growth Regulat Soc Amer 12:1–7
Nonis A, Ruperti B, Pierasco A, Canaguier A, Adam-Blondon AF, Di Gaspero G, Vizzotto G (2008) Neutral invertases in grapevine and comparative analysis with Arabidopsis, poplar and rice. Planta 229:129–142. https://doi.org/10.1007/s00425-008-0815-0
Rabot A, Portemer V, Peron T, Mortreau E, Leduc N, Hamama L, Coutos-Thevenot P, Atanassova R, Sakr S, Le Gourrierec J (2014) Interplay of sugar, light and gibberellins in expression of Rosa hybrida vacuolar invertase 1 regulation. Plant Cell Physiol 55:1734–1748. https://doi.org/10.1093/pcp/pcu106
Reid KE, Olsson N, Schlosser J, Peng F, Lund ST (2006) An optimized grapevine RNA isolation procedure and statistical determination of reference genes for real-time RT-PCR during berry development. BMC Plant Biol 6:27. https://doi.org/10.1186/1471-2229-6-27
Reynolds AG, Wardle DA, Zurowski C, Looney NE (1992) Phenylureas cppu and thidiazuron affect yield components, fruit composition, and storage potential of four seedless grape selections. J Am Soc Hortic Sci 117: 85 – 88. https://doi.org/10.21273/JASHS.117.1.85
Roman H, Girault T, Barbier F, Peron T, Brouard N, Pencik A, Novak O, Vian A, Sakr S, Lothier J, Le Gourrierec J, Leduc N (2016) Cytokinins Are Initial Targets of Light in the Control of Bud Outgrowth. Plant Physiol 172: 489–509.https://doi.org/10.1104/pp.16.00530
Rossouw D, Bosch S, Kossmann J, Botha FC, Groenewald JH (2007) Downregulation of neutral invertase activity in sugarcane cell suspension cultures leads to a reduction in respiration and growth and an increase in sucrose accumulation. Funct Plant Biol 34:490–498. https://doi.org/10.1071/FP06214
Stein O, Granot D (2019) An Overview of Sucrose Synthases in Plants. Front Plant Sci 10:95. https://doi.org/10.3389/fpls.2019.00095
Sturm A (1996) Moleculor characterization and functional analysis of sucrose-cleaving enzymes in carrot (daucus carota l). J Exp Bot 47:1187–1192. https://doi.org/10.1093/jxb/47.Special_Issue.1187
Sturm A, Chrispeels MJ (1990) cDNA cloning of carrot extracellular beta-fructosidase and its expression in response to wounding and bacterial infection. Plant Cell 2:1107–1119. https://doi.org/10.1105/tpc.2.11.1107
Sturm A, Hess D, Lee HS, Lienhard S (1999) Neutral invertase is a novel type of sucrose-cleaving enzyme. Physiol Plant 107:159–165. https://doi.org/10.1034/j.1399-3054.1999.100202.x
Swarbreck D, Wilks C, Lamesch P, Berardini T Z, Garcia-Hernandez M, Foerster H, Li D, Meyer T, Muller R, Ploetz L, Radenbaugh A, Singh S, Swing V, Tissier C, Zhang P, and Huala E. (2008) The Arabidopsis Information Resource (TAIR): gene structure and function annotation. Nucl Acids Res 36(Database issue): D1009–D1014. https://doi.org/10.1093/nar/gkm965
Tamura K, Stecher G, Peterson D, Filipski A, Kumar S (2013) MEGA6: molecular evolutionary genetics analysis version 6.0. Mol Biol Evol 30:2725–2729. https://doi.org/10.1093/molbev/mst197
Tyagi K, Maoz I, Kochanek B, Sela N, Lerno L, Ebeler SE, Lichter A (2021) Cytokinin but not gibberellin application had major impact on the phenylpropanoid pathway in grape. Hort Res 8:51. https://doi.org/10.1038/s41438-021-00488-0
Upadhyay A, Maske S, Jogaiah S, Kadoo NY, Gupta VS (2018) GA(3) application in grapes (Vitis vinifera L.) modulates different sets of genes at cluster emergence, full bloom, and berry stage as revealed by RNA sequence-based transcriptome analysis. Funct Integr Genomic 18:439–455. https://doi.org/10.1007/s10142-018-0605-0
Wan H, Wu L, Yang Y, Zhou G, Ruan YL (2018) Evolution of sucrose metabolism: the dichotomy of invertases and beyond. Trends Plant Sci 23:163–177. https://doi.org/10.1016/j.tplants.2017.11.001
Wang W, Khalil-Ur-Rehman M, Feng J, Tao J (2017) RNA-seq based transcriptomic analysis of CPPU treated grape berries and emission of volatile compounds. J Plant Physiol 218:155–166. https://doi.org/10.1016/j.jplph.2017.08.004
White AC, Rogers A, Rees M, Osborne CP (2016) How can we make plants grow faster? A source-sink perspective on growth rate. J Exp Bot 67:31–45. https://doi.org/10.1093/jxb/erv447
Xie ZS, Li B, Forney CF, Xu WP, Wang SP (2009) Changes in sugar content and relative enzyme activity in grape berry in response to root restriction. Sci Hortic 123(1):39–45. https://doi.org/10.1016/j.scienta.2009.07.017
Yang Z, Wang T, Wang H, Huang X, Qin Y, Hu G (2013) Patterns of enzyme activities and gene expressions in sucrose metabolism in relation to sugar accumulation and composition in the aril of Litchi chinensis Sonn. J Plant Physiol 170:731–740. https://doi.org/10.1016/j.jplph.2012.12.021
Ye JL, Bu YN, He MT, Wu YF, Yang XT, Zhang LL, Song XY (2022) Genome-wide analysis of invertase gene family in wheat (Triticum aestivum L.) indicates involvement of TaCWINVs in pollen development. Plant Growth Regul 98:189–189. https://doi.org/10.1007/s10725-022-00848-3
Zhang CX, Whiting M (2011) Pre-harvest foliar application of Prohexadione-Ca and gibberellins modify canopy source-sink relations and improve quality and shelf-life of “Bing” sweet cherry. Plant Growth Regul 65:145–156. https://doi.org/10.1007/s10725-011-9584-z
Zhang C, Tanabe K, Tamura F, Matsumoto K, Yoshida A (2005) 13C-photosynthate accumulation in Japanese pear fruit during the period of rapid fruit growth is limited by the sink strength of fruit rather than by the transport capacity of the pedicel. J Exp Bot 56:2713–2719. https://doi.org/10.1093/jxb/eri264
Zhang XY, Wang XL, Wang XF, Xia GH, Pan QH, Fan RC, Wu FQ, Yu XC, Zhang DP (2006) A shift of phloem unloading from symplasmic to apoplasmic pathway is involved in developmental onset of ripening in grape berry. Plant Physi 142:220–232. https://doi.org/10.1104/pp.106.081430
Zhang C, Tanabe K, Tamura F, Itai A, Yoshida M (2007) Roles of gibberellins in increasing sink demand in japanese pear fruit during rapid fruit growth. Plant Growth Regul 52(2):161–172. https://doi.org/10.1007/s10725-007-9187-x
Zhang Y, Zhen L, Tan X, Li L, Wang X (2014) The involvement of hexokinase in the coordinated regulation of glucose and gibberellin on cell wall invertase and sucrose synthesis in grape berry. Mol Biol Rep 41:7899–7910. https://doi.org/10.1007/s11033-014-3683-7
Zhu S, Liang Y, An X, Kong F, Gao D, Yin H (2017) Changes in sugar content and related enzyme activities in table grape (Vitis vinifera L.) in response to foliar selenium fertilizer. J Sci Food Agric 97:4094–4102. https://doi.org/10.1002/jsfa.8276
Acknowledgements
This work was supported by the National Natural Science Foundation of China(Grants No. 32102348 and 31872050).
Author information
Authors and Affiliations
Contributions
CLD and CLC performed all experiments and wrote the manuscript. YL involved in physiological index measurement. YML and ZSX revised the manuscript. All authors have read and approved the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Communicated by Bing Yang.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Du, CL., Cai, CL., Lu, Y. et al. Identification and expression analysis of invertase family genes during grape (Vitis vinifera L.) berry development under CPPU and GA treatment. Mol Genet Genomics 298, 777–789 (2023). https://doi.org/10.1007/s00438-023-02015-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00438-023-02015-3