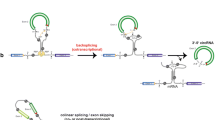
Abstract
Methylation of cytosine residues in DNA of higher eukaryotes, including humans, creates “hot spots” of C→T transitions in the genome. The predominantly methylated sequence in mammalian DNAs is CG (CpG). Among CG-containing codons, CGA codons for arginine are unique due to their ability to create stop codons TGA (UGA in mRNA) upon epigenetic-mediated mutation. As such nonsense mutations can have a strong adverse effect on the cell and organism, we have performed a study, on the example of human genes, aimed to characterise the anticipated effects of epigenetic-mediated nonsense mutations CGA→TGA in somatic cells. It is commonly accepted that premature termination codons (PTCs) lead to the biosynthesis of truncated and usually inactive proteins. In addition, transcripts with PTC can be destroyed by a nonsense-mediated mRNA decay (NMD) machinery. We have considered the cell potentialities (gene families, diploidy, and alternative splicing) to overcome the worst consequences of nonsense mutation. As a special case, in the biosynthesis of a particular group of proteins called selenoproteins, the mutation CGA→UGA would not lead to the premature translation termination and NMD but rather to the insertion of selenocysteine or cysteine instead of former arginine. The finding of SECIS (Sel insertion sequence)-like structures in a variety of mRNAs allowed us to postulate the existence of facultative selenoproteins, whose biosynthesis might be coupled with the redefinition of premature UGA stop codons arising upon mutations, as in the case of “classic” selenoproteins. Nevertheless, a detailed structural analysis of 165 transcripts has shown that roughly 80–90% of functional human mRNAs are potential substrates for NMD upon the PTC emergence. A hypothesis was put forward highlighting a role of arginine CGA codons together with glutamine CAA and CAG codons in the control of mRNA quality and life span. According to this hypothesis, the conversion of the ribonucleic codons CGA, CAA, or CAG into stop codons UGA, UAA or UAG owing to spontaneous or enzymatic cytosine deamination might serve as a trigger for the transcript destruction by NMD (C→U control). Thus, the consequences of epigenetic-mediated nonsense mutations are diverse and may largely depend on the structure of the transcript (CGA codon position, the presence and position of introns and SECIS elements, and splicing potential) of the cognate gene. However, this diversity and the presumable role of CGA codons in performing the everyday function by controlling whether genes are expressed correctly do not exclude their long-term role as limiters of the cell and organism life span. Thus, the presumable role of CGA codons in genome functioning and stability opens new perspectives to influence aging and concomitant deceases by codon editing.



Similar content being viewed by others
References
Anisimov VN (2008) Molecular and physiological mechanisms of aging (in Russian), vol 1. Nauka, St. Petersburg
Bird AP (1980) DNA methylation and the frequency of CpG in animal DNA. Nucleic Acid Res 8:1499–1504
Buijs A, van Wijnen M, van den Blink D, van Gijn M, Klein SK (2013) A ZMYM2-FGFR1 8p11 myeloproliferative neoplasm with a novel nonsense RUNX1 mutation and tumor lysis upon imatinib treatment. Cancer Genet 206:140–144
Burns MB, Temiz NA, Harris RS (2013) Evidence for APOBEC3B mutagenesis in multiple human cancers. Nat Genet 45:977–983
Cooper DN, Krawczak M (1989) Cytosine methylation and the fate of CpG dinucleotides in vertebrate genomes. Hum Genet 83:181–188
Cooper DN, Krawczak M (1990) The mutational spectrum of single base-pair substitutions causing human genetic disease: patterns and predictions. Hum Genet 85:55–74
Cooper DN, Youssoufian H (1988) The CpG dinucleotide and human genetic disease. Hum Genet 78:151–155
Eckhardt F, Lewin J, Cortese R, Rakyan VK, Attwood J, Burger M, Burton J, Cox TV, Davies R, Down TA, Haefliger C, Horton R, Otto T, Pettett R, Seemann S, Thompson C, West T, Rogers J, Olek A, Berlin K, Beck S (2006) DNA methylation profiling of human chromosomes 6, 20 and 22. Nat Genet 38:1378–1385
Ehrlich M, Norris KF, Wang RY-H, Kuo KC, Gehrke CW (1986) DNA cytosine methylation and heat-induced deamination. Biosci Rep 6:388–393
Frederico LA, Kunkel TA, Shaw BR (1990) A sensitive genetic assay for the detection of cytosine deamination: determination of rate constants and the activation energy. Biochemistry 29:2532–2537
Fujii A, Harada T, Iwama E, Ota K, Furuyama K, Ijichi K, Okamoto T, Okamoto I, Takayama K, Nakanishi Y (2015) Hypermethylation of the CpG dinucleotide in epidermal growth factor receptor codon 790: implications for a mutational hotspot leading to the T790M mutation in non-small-cell lung cancer. Cancer Genet 208:271–278
Khajavi M, Inoue K, Lupski JR (2006) Nonsense-mediated mRNA decay modulates clinical outcome of genetic disease. Eur J Hum Genet 14:1074–1081
Krawczak M, Ball EV, Cooper DN (1998) Neighboring-nucleotide effects on the rates of germ-line single-base-pair substitution in human genes. Am J Hum Genet 63:474–488
Kryukov GV, Castellano S, Novoselov SV, Lobanov AV, Zehtab O, Guigo R, Gladyshev VN (2003) Characterization of mammalian selenoproteomes. Science 300:1439–1443
Kurosaki T, Maquat LE (2016) Nonsense-mediated mRNA decay in humans at a glance. J Cell Sci 129:461–467
Labunskyy VM, Hatfield DL, Gladyshev VN (2014) Selenoproteins: molecular pathways and physiological roles. Physiol Rev 94:739–777
Leroy B, Anderson M, Soussi T (2014) TP53 mutations in human cancer: database reassessment and prospects for the next decade. Hum Mutat 35:672–688
Lewin B (2008) Genes IX. Jones and Bartlett Publ, Boston
Longo VD, Mitteldorf J, Skulachev VP (2005) Programmed and altruistic ageing. Nat Rev Genet 6:866–872
Magebu AN, Jones PA (1994) Ubiquitous and tenacious methylation of the CpG site in codon 248 of the p53 gene may explain its frequent appearance as a mutational hot spot in human cancer. Mol Cell Biol 14:4225–4232
Malousi A, Kouidou S (2012) DNA hypermethylation of alternatively spliced and repeat sequences in humans. Mol Genet Genom 287:631–642
Mazin AL (2009) Suicidal function of DNA methylation in age-related genome disintegration. Ageing Res Rev 8:314–327
Mazzio EA, Soliman KF (2012) Basic concepts of epigenetics: impact of environmental signals on gene expression. Epigenetics 7:119–130
Milholland B, Auton A, Suh Y, Vijg J (2015) Age-related somatic mutations in the cancer genome. Oncotarget 6:24627–24635
Mort M, Ivanov D, Cooper DN, Chuzhanova NA (2008) A meta-analysis of nonsense mutations causing human genetic disease. Hum Mutat 29:1037–1047
Petit V, Guétard D, Renard M, Keriel A, Sitbon M, Wain-Hobson S, Vartanian J-P (2009) Murine APOBEC1 is a powerful mutator of retroviral and cellular RNA in vitro and in vivo. J Mol Biol 385:65–78
Podolskiy DI, Lobanov AV, Kryukov GV, Gladyshev VN (2016) Analysis of cancer genomes reveals basic features of human aging and its role in cancer development. Nat Commun 7:12157
Ponomarenko EA, Poverennaya EV, Ilgisonis EV, Pyatnitskiy MA, Kopylov AT, Zgoda VG, Lisitsa AV, Archakov AI (2016) The size of the human proteome: the width and depth. Int J Anal Chem 2016:ID 7436849
Popp MW, Maquat LE (2013) Organizing principles of mammalian nonsense-mediated mRNA decay. Annu Rev Genet 47:139–165
Rabinowicz PD, Palmer LE, May BP, Hemann MT, Lowe SW, McCombie WR, Martienssen RA (2003) Genes and transposons are differentially methylated in plants, but not in mammals. Genome Res 13:2658–2664
Romanov GA, Vanyushin BF (1980) DNA methylation in eukaryotes. I. Methylated sequences and DNA methylases (in Russian). Nauch Dokl Vyssh Shkoly Biol Nauki 11:5–20
Romanov GA, Vanyushin BF (1981) Methylation of reiterated sequences in mammalian DNAs. Effects of the tissue type, age, malignancy and hormonal induction. Biochim Biophys Acta 653:204–218
Romanov GA, Sukhoverov VS, Vanyushin BF (2015) Epigenetic mutagenesis as program of age-related protein dysfunction and aging. Russ J Dev Biol 46:78–87
Schormann N, Ricciardi R, Chattopadhyay D (2014) Uracil-DNA glycosylases—structural and functional perspectives on an essential family of DNA repair enzymes. Prot Sci 23:1667–1685
Seyedali A, Berry MJ (2014) Nonsense-mediated decay factors are involved in the regulation of selenoprotein mRNA levels during selenium deficiency. RNA 20:1248–1256
Sohail A, Hayes CS, Divvela P, Setlow P, Bhagwat AS (2002) Protection of DNA by alpha/beta-type small, acid-soluble proteins from Bacillus subtilis spores against cytosine deamination. Biochemistry 41:11325–11330
Tobe R, Naranjo-Suarez S, Everley RA, Carlson BA, Turanov AA, Tsuji PA, Yoo M-H, Gygi SP, Gladyshev VN, Hatfield DL (2013) High error rates in selenocysteine insertion in mammalian cells treated with the antibiotics, doxycycline, chloramphenicol or geneticin. J Biol Chem 288:14709–14715
Toma KG, Rebbapragada I, Durand S, Lykke-Andersen J (2015) Identification of elements in human long 3′UTRs that inhibit nonsense-mediated decay. RNA 21:1–11
Tornaletti S, Pfeifer GP (1995) Complete and tissue-independent methylation of CpG sites in the p53 gene: implications for mutations in human cancers. Oncogene 10:1493–1499
Trindade LS, Aigaki T, Peixoto AA, Balduino A, Mânica da Cruz IB, Heddle JG (2013) A novel classification system for evolutionary aging theories. Front Genet 4:Art 25
Vanyushin BF (2005) Enzymatic DNA methylation is an epigenetic control for genetic functions of the cell. Biochemistry (Mosc) 70:488–499
Wolters S, Schumacher B (2013) Genome maintenance and transcription integrity in aging and disease. Front Genet 4:Art 19
Ying H, Huttley G (2011) Exploiting CpG hypermutability to identify phenotypically significant variation within human protein-coding genes. Genome Biol Evol 3:938–949
Yoshida K, Sanada M, Kato M, Kawahata R, Matsubara A, Takita J, Shih LY, Mori H, Koeffler HP, Ogawa S (2011) A nonsense mutation of IDH1 in myelodysplastic syndromes and related disorders. Leukemia 25:184–186
Acknowledgements
This work was partially supported by Molecular and Cell Biology Program of the Presidium of the Russian Academy of Sciences.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by S. Hohmann.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Romanov, G.A., Sukhoverov, V.S. Arginine CGA codons as a source of nonsense mutations: a possible role in multivariant gene expression, control of mRNA quality, and aging. Mol Genet Genomics 292, 1013–1026 (2017). https://doi.org/10.1007/s00438-017-1328-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00438-017-1328-y