Abstract
bHLH/PAS genes encode a family of basic helix-loop-helix (bHLH) transcription factors with bHLH, PAS and PAS_3 domain. bHLH/PAS genes are involved in many essential physiological and developmental processes, such as hypoxic response neural development, the circadian clock, and learning ability. Despite their important functions, the origin and evolution of this bHLH/PAS gene family has yet to be elucidated. In this study, we aim to explore the origin, evolution, gene structure conservation of this gene family and provide a model to analyze the evolution of other gene families. Our results show that genes of the bHLH/PAS family only exist in metazoans. They may have originated from the common ancestor of metazoans and expanded into vertebrates. We identified bHLH/PAS genes in more than ten species representing the main lineages and constructed the phylogenetic trees (Beyasian, ML and NJ) to classify them into three groups. The exon–intron structure analysis revealed that a relatively conserved “1001-0210” eight-exon structure exists in most groups and lineages. In addition, we found the exon fusion pattern in several groups in this conserved eight-exon structure. Further analysis indicated that bHLH/PAS protein paralogs evolved from several gene duplication events followed by functional divergence and purifying selection. We presented a phylogenetic model to describe the evolutionary history of the exon structures of bHLH/PAS genes. Taken together, our study revealed the evolutionary model, functional divergence and gene structure conservation of bHLH/PAS genes. These findings provide clues for the functional and evolutionary mechanism of bHLH/PAS genes.




Similar content being viewed by others
References
Abascal F, Zardoya R et al (2005) ProtTest: selection of best-fit models of protein evolution. Bioinformatics (Oxford, England) 21(9):2104–2105
Conant GC, Wolfe KH (2008) Turning a hobby into a job: how duplicated genes find new functions. Nat Rev Genet 9(12):938–950
Erbel-Sieler C, Dudley C et al (2004) Behavioral and regulatory abnormalities in mice deficient in the NPAS1 and NPAS3 transcription factors. Proc Natl Acad Sci USA 101(37):13648–13653
Esser C, Rannug A et al (2009) The aryl hydrocarbon receptor in immunity. Trends Immunol 30(9):447–454
Gilles-Gonzalez MA, Gonzalez G (2004) Signal transduction by heme-containing PAS-domain proteins. J Appl Physiol 96(2):774–783
Griffin EA Jr, Staknis D et al (1999) Light-independent role of CRY1 and CRY2 in the mammalian circadian clock. Science (New York, NY) 286(5440):768–771
Gu X (1999) Statistical methods for testing functional divergence after gene duplication. Mol Biol Evol 16(12):1664–1674
Gu X (2001) Maximum-likelihood approach for gene family evolution under functional divergence. Mol Biol Evol 18(4):453–464
Gu X, Vander Velden K (2002) DIVERGE: phylogeny-based analysis for functional-structural divergence of a protein family. Bioinformatics (Oxford, England) 18(3):500–501
Guindon S, Gascuel O (2003) A simple, fast, and accurate algorithm to estimate large phylogenies by maximum likelihood. Syst Biol 52(5):696–704
Hahn MW (2009) Distinguishing among evolutionary models for the maintenance of gene duplicates. J Hered 100(5):605–617
Hahn ME, Karchner SI et al (2006) Unexpected diversity of aryl hydrocarbon receptors in non-mammalian vertebrates: insights from comparative genomics. J Exp Zool A Comp Exp Biol 305(9):693–706
Hokamp K, McLysaght A et al (2003) The 2R hypothesis and the human genome sequence. J Struct Funct Genomics 3(1–4):95–110
Huang Y, Niu B et al (2010) CD-HIT Suite: a web server for clustering and comparing biological sequences. Bioinformatics (Oxford, England) 26(5):680–682
Hughes DA, Jastroch M et al (2009) Molecular evolution of UCP1 and the evolutionary history of mammalian non-shivering thermogenesis. BMC Evol Biol 9:4
Kewley RJ, Whitelaw ML et al (2004) The mammalian basic helix-loop-helix/PAS family of transcriptional regulators. Int J Biochem Cell Biol 36(2):189–204
Kume K, Zylka MJ et al (1999) mCRY1 and mCRY2 are essential components of the negative limb of the circadian clock feedback loop. Cell 98(2):193–205
Larkin MA, Blackshields G et al (2007) Clustal W and Clustal X version 2.0. Bioinformatics (Oxford, England) 23(21):2947–2948
Linne V, Eriksson BJ et al (2012) Single-minded and the evolution of the ventral midline in arthropods. Dev Biol 364(1):66–76
Meyer A, Van de Peer Y (2005) From 2R to 3R: evidence for a fish-specific genome duplication (FSGD). BioEssays 27(9):937–945
Milenkovic VM, Brockmann M et al (2010) Evolution and functional divergence of the anoctamin family of membrane proteins. BMC Evol Biol 10:319
Moglich A, Ayers RA et al (2009) Structure and signaling mechanism of Per-ARNT-Sim domains. Structure 17(10):1282–1294
Panopoulou G, Poustka AJ (2005) Timing and mechanism of ancient vertebrate genome duplications—the adventure of a hypothesis. Trends Genet 21(10):559–567
Pickard BS, Malloy MP et al (2005) Disruption of a brain transcription factor, NPAS3, is associated with schizophrenia and learning disability. Am J Med Genet B Neuropsychiatr Genet 136B(1):26–32
Ploski JE, Monsey MS et al (2011) The neuronal PAS domain protein 4 (Npas4) is required for new and reactivated fear memories. PLoS ONE 6(8):e23760
Preitner N, Damiola F et al (2002) The orphan nuclear receptor REV-ERBalpha controls circadian transcription within the positive limb of the mammalian circadian oscillator. Cell 110(2):251–260
Putnam NH, Srivastava M et al (2007) Sea anemone genome reveals ancestral eumetazoan gene repertoire and genomic organization. Science (New York, NY) 317(5834):86–94
Reick M, Garcia JA et al (2001) NPAS2: an analog of clock operative in the mammalian forebrain. Science (New York, NY) 293(5529):506–509
Reitzel AM, Behrendt L et al (2010) Light entrained rhythmic gene expression in the sea anemone Nematostella vectensis: the evolution of the animal circadian clock. PLoS ONE 5(9):e12805
Ronquist F, Huelsenbeck JP (2003) MrBayes 3: Bayesian phylogenetic inference under mixed models. Bioinformatics (Oxford, England) 19(12):1572–1574
Rytkonen KT, Williams TA et al (2011) Molecular evolution of the metazoan PHD-HIF oxygen-sensing system. Mol Biol Evol 28(6):1913–1926
Sailsbery JK, Dean RA (2012) Accurate discrimination of bHLH domains in plants, animals, and fungi using biologically meaningful sites. BMC Evol Biol 12:154
Stevens EA, Mezrich JD et al (2009) The aryl hydrocarbon receptor: a perspective on potential roles in the immune system. Immunology 127(3):299–311
Suyama M, Torrents D et al (2006) PAL2NAL: robust conversion of protein sequence alignments into the corresponding codon alignments. Nucleic Acids Res 34(Web Server issue):W609–W612
Tamura KDJ, Nei M, Kumar S (2007) MEGA4: Molecular Evolutionary Genetics Analysis (MEGA) software version 4.0. Mol Biol Evol 24:1596–1599
Taylor JS, Van de Peer Y et al (2001) Comparative genomics provides evidence for an ancient genome duplication event in fish. Philos Trans R Soc London 356(1414):1661–1679
Teng H, Cai W et al (2010) Evolutionary mode and functional divergence of vertebrate NMDA receptor subunit 2 genes. PLoS ONE 5(10):e13342
Vandepoele K, De Vos W et al (2004) Major events in the genome evolution of vertebrates: paranome age and size differ considerably between ray-finned fishes and land vertebrates. Proc Natl Acad Sci USA 101(6):1638–1643
Yan J, Cai Z (2010) Molecular evolution and functional divergence of the cytochrome P450 3 (CYP3) Family in Actinopterygii (ray-finned fish). PLoS ONE 5(12):e14276
Yang Z (2007) PAML 4: phylogenetic analysis by maximum likelihood. Mol Biol Evol 24(8):1586–1591
Zhang X, Wang M et al (2010) Molecular selection and functional divergence of HIF-alpha proteins in vertebrates. Genetica 138(11–12):1241–1250
Zhou M, Yan J et al (2012) Comparative and evolutionary analysis of the HES/HEY gene family reveal exon/intron loss and teleost specific duplication events. PLoS ONE 7(7):e40649
Acknowledgments
We would like to thank Dr Siluo Huang (Huazhong University of Science and Technology) for advice on this study. This work was supported by the following funds to A.Y.G: National Natural Science Foundation of China (NSFC) (31171271, 31270885), Young Teachers’ Fund for Doctor Stations, Ministry of Education of China (20110142120042), the Scientific Research Fund for the Returned Overseas Chinese Scholars, Ministry of Education of China, and fund from the State Key Laboratory of Freshwater Ecology and Biotechnology (2012FB02).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by S. Xu.
Electronic supplementary material
Below is the link to the electronic supplementary material.
438_2013_786_MOESM2_ESM.pdf
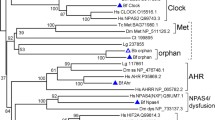
Expanded subtrees of Figure 1. (A) AHR, (B) NPAS1/3, (C) ARNT1/2, (D) CLOCK/NPAS2, (E) ARNTL1/2, (F) SIM1/2, (G)HIF-1-3α (PDF 192 kb)
438_2013_786_MOESM3_ESM.pdf
Phylogenetic trees by some other methods. (A) Phylogenetic ML tree of bHLH/PAS genes (JTT+I+G+F), (B) Phylogenetic NJ tree of bHLH/PAS genes (JTT) (PDF 348 kb)
438_2013_786_MOESM4_ESM.pdf
The exon structures of representative bHLH/PAS genes. This graph indicated the exon-intron structures of bHLH/PAS genes in human, frog, zebrafish, amphioxus, coina, sea urchin, fruit fly, nematode and sea anemone. Red box: bHLH domain; green box: PAS domain; purple box: PAS_3 domain; black box: untranslated regions (UTRs); white box: other exon regions; black line: intron. Numbers 0, 1, and 2 indicate the exon phases. The lengths of the exons are scaled based on the length of sequences except some long UTRs were shorted by “//”. The introns were shortened. Abbreviations of species names are in the following. Hs: Homo sapiens, Xt: Xenopus tropicalis, Dr: Danio rerio, Bf: Branchiostoma floridae, Ci: Ciona intestinalis, Sp: Strongylocentrotus purpuratus, Dm: Drosophila melanogaster, Ce: Caenorhabditis elegans, Nv: Nematostella vectensis. (A) AHR, SIM1/2, HIF-1-3α, NPAS1/3, (B) NPAS2/CLOCK, ARNT1/2, ARNTL1/2 (PDF 318 kb)
438_2013_786_MOESM6_ESM.pdf
The conserved exon-intron structure in 14 human bHLH/PAS genes. A conserved exon-intron structure with “1001-0210” in evolution was labeled by translucent yellow boxes in 14 human and 1 sea anemone bHLH/PAS genes (PDF 163 kb)
Rights and permissions
About this article
Cite this article
Yan, J., Ma, Z., Xu, X. et al. Evolution, functional divergence and conserved exon–intron structure of bHLH/PAS gene family. Mol Genet Genomics 289, 25–36 (2014). https://doi.org/10.1007/s00438-013-0786-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00438-013-0786-0