Abstract
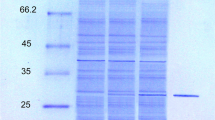
A lipase gene (lip3) was cloned from the Pseudomonas aeruginosa strain LST-03 (which tolerates organic solvents) and expressed in Escherichia coli. The cloned sequence includes an ORF consisting of 945 nucleotides, encoding a protein of 315 amino acids (Lip3 lipase, 34.8 kDa). The predicted Lip3 lipase belongs to the class of serine hydrolases; the catalytic triad consists of the residues Ser-137, Asp-258, and His-286. The gene cloned in the present study does not encode the LST-03 lipase, a previously isolated solvent-stable lipase secreted by P. aeruginosa LST-03, because the N-terminal amino acid sequence of the Lip3 lipase differs from that of the LST-03 lipase. Although the effects of pH on the activity and stability of the Lip3 lipase, and the temperature optimum of the enzyme, were similar to those of the LST-03 lipase, the relative activity of the Lip3 lipase at lower temperatures (0–35°C) was higher than that of the LST-03 lipase. In the absence of organic solvents, the half-life of the Lip3 lipase was similar to that of the LST-03 lipase. However, in the presence of most of the organic solvents tested in this study (the exceptions were ethylene glycol and glycerol), the stability of the Lip3 lipase was lower than that of the LST-03 lipase.






Similar content being viewed by others
References
Amann E, Ochs B, Abel KJ (1988) Tightly regulated tac promoter vectors useful for the expression of unfused and fused proteins in Escherichia coli. Gene 69:301–15
Arpigny JL, Feller G, Gerday C (1993) Cloning, sequence and structural features of a lipase from the Antarctic facultative psychrophile Psychrobacter immobilis B10. Biochim Biophys Acta 1171:331–333
Chihara-Siomi M, Yoshikawa K, Oshima-Hirayama N, Yamamoto K, Sogabe Y, Nakatani T, Nishioka T, Oda J (1992) Purification, molecular cloning, and expression of lipase from Pseudomonas aeruginosa. Arch Biochem Biophys 296:505–513
Duchene M, Schweizer A, Lottspeich F, Krauss G, Marget M, Vogel K, von Specht BU, Domdey H (1988) Sequence and transcriptional start site of the Pseudomonas aeruginosa outer membrane porin protein F gene. J Bacteriol 170:155–162
Feller G, Thiry M, Gerday C (1991) Nucleotide sequence of the lipase gene lip3 from the Antarctic psychotroph Moraxella TA144. Biochim Biophys Acta 1088:323–324
Fukumoto J, Iwai M, Tsujisaka Y (1964) Studies on lipase: IV. Purification and properties of a lipase secreted by Rhizopus delemer. J Gen Appl Microbiol 10:257–265
Fushinobu S, Saku T, Hidaka M, Jun SY, Nojiri H, Yamane H, Shoun H, Omori T, Wakagi T (2002) Crystal structures of a meta-cleavage product hydrolase from Pseudomonas fluorescens IP01 (CumD) complexed with cleavage products. Protein Sci 11:2184–2195
Habe H, Kasuga K, Nojiri H, Yamane H, Omori T (1996) Analysis of cumene (isopropylbenzene) degradation genes from Pseudomonas fluorescens IP01. Appl Environ Microbiol 62:4471–4477
Hanahan D (1983) Studies on transformation of Escherichia coli with plasmids. J Mol Biol 166:557-580
Kurooka S, Okamoto S, Hashimoto M (1977) A novel and simple colorimetric assay for human serum lipase. J Biochem 81:361–369
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage. Nature 227:680–685
Lee SY, Rasheed S (1990) A simple procedure for maximum yield of high-quality plasmid DNA. Biotechniques 9:676–679
Medrano FJ, Alonso J, Garcia JL, Romero A, Bode W, Gomis-Ruth FX (1998) Structure of proline iminopeptidase from Xanthomonas campestris pv. citri: A prototype for the prolyl oligopeptidase family. EMBO J 17:1–9
Murray MG, Thompson WF (1980) Rapid isolation of high molecular weight plant DNA. Nucleic Acids Res 8:4321-4325
Murray V (1989) Improved double-stranded DNA sequencing using the linear polymerase chain reaction. Nucleic Acids Res 17:8889
Nandhagopal N, Yamada A, Hatta T, Masai E, Fukuda M, Mitsui Y, Senda T (2001) Crystal structure of 2-hydroxyl-6-oxo-6-phenylhexa-2,4-dienoic acid (HPDA) hydrolase (BphD enzyme) from the Rhodococcus sp. strain RHA1 of the PCB degradation pathway. J Mol Biol 309:1139-1151
Ogino H, Ishikawa H (2001) Enzymes which are stable in the presence of organic solvents. J Biosci Bioeng 91:109–116
Ogino H, Miyamoto K, Ishikawa H (1994) Organic-solvent-tolerant bacterium which secretes organic-solvent-stable lipolytic enzyme. Appl Environ Microbiol 60:3884–3886
Ogino H, Miyamoto K, Yasuda M, Ishimi K, Ishikawa H (1999a) Growth of organic solvent-tolerant Pseudomonas aeruginosa LST-03 in the presence of various organic solvents and production of lipolytic enzyme in the presence of cyclohexane. Biochem Eng J 4:1–6
Ogino H, Watanabe F, Yamada M, Nakagawa S, Hirose T, Noguchi A, Yasuda M, Ishikawa H (1999b) Purification and characterization of organic solvent-stable protease from organic solvent-tolerant Pseudomonas aeruginosa PST-01. J Biosci Bioeng 87:61–68
Ogino H, Nakagawa S, Shinya K, Muto T, Fujimura N, Yasuda M, Ishikawa H (2000a) Purification and characterization of organic solvent-stable lipase from organic solvent-tolerant Pseudomonas aeruginosa LST-03. J Biosci Bioeng 89:451–457
Ogino H, Yokoo J, Watanabe F, Ishikawa H (2000b) Cloning and sequencing of a gene of organic solvent-stable protease secreted from Pseudomonas aeruginosa PST-01 and its expression in Escherichia coli. Biochem Eng J 5:191–200
Ogino H, Uchiho T, Yokoo J, Kobayashi R, Ichise R, Ishikawa H (2001) Role of intermolecular disulfide bonds of the organic solvent-stable PST-01 protease in the organic solvent-stability. Appl Environ Microbiol 67:942–947
Palleroni NJ (1984) Family I. Pseudomonadaceae. In: Kreig NR, Holt JG (eds) Bergey’s manual of systematic bacteriology, vol. 1. Williams and Wilkins, Baltimore, pp 141–219
Rowe-Magnus DA, Guerout AM, Ploncard P, Dychinco B, Davies J, Mazel D (2001) The evolutionary history of chromosomal super-integrons provides an ancestry for multiresistant integrons. Proc Natl Acad Sci USA. 98:652–657
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual (2nd edn). Cold Spring Harbor Laboratory Press, Cold Spring Harbor, N.Y.
Stover CK, et al (2000) Complete genome sequence of Pseudomonas aeruginosa PA01: an opportunistic pathogen. Nature 406:959–964
Vieira J, Messing J (1987) Production of single-stranded plasmid DNA. Methods Enzymol 153:3–11
Yanisch-Perron C, Vieira J, Messing J (1985) Improved M13 phage cloning vectors and host strains: Nucleotide sequences of the M13mp18 and pUC19 vectors. Gene 33:103–119
Zylstra GJ, Olsen RH, Ballou DP (1989) Cloning, expression, and regulation of the Pseudomonas cepacia protocatechuate 3,4-dioxygenase genes. J Bacteriol 171:5907–5914
Acknowledgments
This work was supported in part by a Grant-in-Aid for Scientific Research (A) (14205112) from the Japan Society for the Promotion of Science. The authors would like to express their thanks to this support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by H. Ikeda
Rights and permissions
About this article
Cite this article
Ogino, H., Hiroshima, S., Hirose, S. et al. Cloning, expression and characterization of a lipase gene (lip3) from Pseudomonas aeruginosa LST-03. Mol Genet Genomics 271, 189–196 (2004). https://doi.org/10.1007/s00438-003-0970-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00438-003-0970-8