Abstract
The aim of this study was to characterize the Tunisian Fasciola spp. flukes by morphometric and molecular analyses. Flukes were collected from livers of sheep slaughtered in Sejnane slaughterhouses (Bizerte gouvernorate, Northwest Tunisia) between January and March 2021.
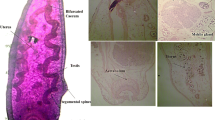
Five morphometric parameters were determined for all the liver flukes, as follows: (i) total body length (BL), (ii) distance between ventral sucker and the tail (VS-T), (iii) distance between oral sucker and ventral sucker (OS-VS), (iv) abdomen diameter (AD), (v) tail diameter (TD) and the body length to width ratio (BL/BW). Molecular identification of the fluke specimens was carried out by polymerase chain reaction, restriction fragment polymorphism (PCR-RFLP) of a 680 bp sequence of the internal transcribes spacer 1 (ITS1) gene and by amplification, sequencing, and phylogenetic analysis of a 500 bp sequence of the ITS2 gene. Morphometric measurements showed that the mean of the total body length of the adult flukes was 21.1 ± 2.7 mm with minimum and maximum lengths of 13 and 31 mm, respectively. The PCR-RFLP analysis revealed a single profile consisting of three bands of approximately 370, 100, and 60 bp. Fasciola sequences described in the present study (GenBank numbers: OQ457027 and OQ457028) showed 99.58–100% identity to Fasciola hepatica. In conclusion, the results of this study show that molecular and phylogenetic analyses confirm the presence of a single species of F. hepatica in the Sejnane region Northwest of Tunisia. However, further studies are needed to identify the occurrence of Fasciola species in other Tunisian regions.




Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Abdel-Fatah OR, Arafa WM, Wahba AA, El-Dakhly KM (2022) Economic losses, morpho-molecular identification, and identity of Fasciola species recovered from Egypt. J Parasit Dis 46:1036–1046. https://doi.org/10.1007/s12639-022-01526-x
Abebe R, Abunna F, Mekuria S, Megersa B (2010) Fasciolosis: prevalence, financial losses due to liver condemnation and evaluation of a simple sedimen- tation diagnostic technique in cattle slaughtered at Hawassa Municipal abattoir, southern Ethiopia. Ethiop Vet J 14(1):39–51. https://doi.org/10.4314/evj.v14i1.63868
Adlard RD, Barker SC, Blair D, Cribb TH (1993) Comparison of the second internal transcribed spacer (Ribosomal DNA) from populations and species of fasciolidae (Digenea). Int J Parasitol 23:423–425. https://doi.org/10.1016/0020-7519(93)90022-Q
Admassu B, Shite A, Kinfe G (2015) A review on bovine fasciolosis, vol 7, pp 139–146. https://doi.org/10.5829/idosi.ejbs.2015.7.03.9558
Akhlaghi E, Mohammadi MA, Ziaali N et al (2017) Morphometric and molecular study of Fasciola isolates from ruminants in Iran. Turkiye parazitolojii Derg 41:192–197. https://doi.org/10.5152/tpd.2017.5214
Akkari H, Gharbi M, Darghouth MA (2011) Infestation of tracer lambs by Fasciola hepatica in Tunisia : determining periods for strategic anthelmintic treatments. OIE Rev Sci Tech 30:917–929
Alasaad S, Huang CQ, Li QY et al (2007) Characterization of Fasciola samples from different host species and geographical localities in Spain by sequences of internal transcribed spacers of rDNA. Parasitol Res 101:1245–1250. https://doi.org/10.1007/s00436-007-0628-2
Altschul SF, Gish W, Miller W et al (1990) Basic local alignment search tool. J Mol Biol 215:403–410. https://doi.org/10.1016/S0022-2836(05)80360-2
Amer S, Dar Y, Ichikawa M et al (2011) Parasitology International Identi fi cation of Fasciola species isolated from Egypt based on sequence analysis of genomic (ITS1 and ITS2) and mitochondrial (NDI and COI) gene markers. Parasitol Int 60:5–12. https://doi.org/10.1016/j.parint.2010.09.003
Amer S, ElKhatam A, Zidan S et al (2016) Identity of Fasciola spp. in sheep in Egypt. Parasites and Vectors 9(1):8. https://doi.org/10.1186/s13071-016-1898-2
Amor N, Farjallah S, Said K, Ben SB (2011) First report of Fasciola hepatica in Equus caballus host species from Tunisia based on the ribosomal internal transcribed spacer regions. Turkish J Vet Anim Sci 35:319–324. https://doi.org/10.3906/vet-1007-391
Anh DN, Anh LT, Tuan LQ et al (2018) Identification of Fasciola species isolates from Nghe An Province, Vietnam, based on ITS1 sequence of ribosomal DNA using a Simple PCR-RFLP method. J Parasitol Res 2018:1–6. https://doi.org/10.1155/2018/2958026
Ayadi A, Makni F, Ben Said M (1997) État actuel de la fasciolose en Tunisie. Bull la Société Française Parasitol 15:27–32
Bargues MD, Gayo V, Sanchis J et al (2017) DNA multigene characterization of Fasciola hepatica and Lymnaea neotropica and its fascioliasis transmission capacity in Uruguay, with historical correlation, human report review and infection risk analysis. PLoS Negl Trop Dis 11(2):e0005352
Bergeon P, Laurent M (1970) Différences entre la morphologie testiculaire de Fasciola hepatica et Fasciola gigantica. Rev d’élevage médecine vétérinaire des pays Trop 23:223. https://doi.org/10.19182/remvt.7700
Dar Y, Amer S, Mercier A et al (2012) Molecular identification of Fasciola spp. (Digenea: Fasciolidae) in Egypt. Parasite 19:177–182. https://doi.org/10.1051/parasite/2012192177
Dar Y, Vignoles P, Dreyfuss G, Rondelaud D (2003) Fasciola hepatica and Fasciola gigantica: comparative morphometric studies on the redial stage of both species. Parasitol Res 91:369–373. https://doi.org/10.1007/s00436-003-0966-7
Diyana JNA, Mahiza MIN, Latiffah H et al (2020) Occurrence, morphometric, and molecular investigation of cattle and buffalo liver adult fluke in Peninsular Malaysia main abattoirs. J Parasitol Res 2020:9–14. https://doi.org/10.1155/2020/5436846
El-Rahimy HH, Mahgoub AMA, El-Gebaly NSM et al (2012) Molecular, biochemical, and morphometric characterization of Fasciola species potentially causing zoonotic disease in Egypt. Parasitol Res 111:1103–1111. https://doi.org/10.1007/s00436-012-2938-2
Eliza N, Calvani D, Ichikawa-seki M et al (2020) Which species is in the faeces at a time of global livestock movements : single nucleotide polymorphism genotyping assays for the differentiation of Fasciola spp . q. Int J Parasitol. 50(2):91–101. https://doi.org/10.1016/j.ijpara.2019.12.002
Evack JG, Schmidt RS, Boltryk SD et al (2020) Molecular confirmation of a Fasciola gigantica × Fasciola hepatica hybrid in a Chadian bovine. J Parasitol 106(2):316–322. https://doi.org/10.1645/19-66
Farjallah S, Sanna D, Amor N et al (2009) Genetic characterization of Fasciola hepatica from Tunisia and Algeria based on mitochondrial and nuclear DNA sequences. Parasitol Res 105:1617–1621. https://doi.org/10.1007/s00436-009-1601-z
Gupta A, Bhardwaj A (2013) Mitochondrial DNA- a tool for phylogenetic and biodiversity search in equines. J Biodivers Endanger Species 01:1–8. https://doi.org/10.4172/2332-2543.s1-006
Hammami H, Hamed N, Ayadi A (2005) Epidemiological studies on Fasciola hepatica in Gafsa oases (South West of Tunisia). Parasite 14:261–264
Haridwal S, Malatji MP, Mukaratirwa S (2021) Food and Waterborne Parasitology Morphological and molecular characterization of Fasciola hepatica and Fasciola gigantica phenotypes from co-endemic localities in Mpumalanga and KwaZulu-Natal provinces of South Africa. Food Waterborne Parasitol 22:e00114. https://doi.org/10.1016/j.fawpar.2021.e00114
Hasanpour H, Falak R, Naddaf SR et al (2020) Molecular characterization of fasciola spp. From some parts of Iran. Iran J Public Health 49:157–166. https://doi.org/10.18502/ijph.v49i1.3062
Ichikawa M, Itagaki T (2012) Molecular analysis of aspermic Fasciola flukes from Korea on the basis of the nuclear ITS1 region and mitochondrial DNA markers and comparison with Japanese aspermic Fasciola flukes. J Vet Med Sci 74:899–904. https://doi.org/10.1292/jvms.11-0523
Ichikawa M, Itagaki T (2010) Discrimination of the ITS1 types of Fasciola spp. based on a PCR-RFLP method. Parasitol Res 106:757–761. https://doi.org/10.1007/s00436-010-1724-2
Itagaki T, Hayashi K, Ohari Y (2022) The causative agents of fascioliasis in animals and humans: parthenogenetic Fasciola in Asia and other regions. Infect Genet Evol 99:105248. https://doi.org/10.1016/j.meegid.2022.105248
Itagaki T, Ichinomiya M, Fukuda K et al (2011) Hybridization experiments indicate incomplete reproductive isolating mechanism between Fasciola hepatica and Fasciola gigantica. Parasitology 138:1278–1284. https://doi.org/10.1017/S0031182011000965
Itagaki T, Kikawa M, Sakaguchi K et al (2005a) Genetic characterization of parthenogenic Fasciola sp. in Japan on the basis of the sequences of ribosomal and mitochondrial DNA. Parasitology 131:679–685. https://doi.org/10.1017/S0031182005008292
Itagaki T, Kikawa M, Terasaki K et al (2005b) Molecular characterization of parthenogenic Fasciola sp. in Korea on the basis of DNA sequences of ribosomal ITS1 and mitochondrial NDI gene. J Vet Med Sci 67:1115–1118. https://doi.org/10.1292/jvms.67.1115
Kumar S, Stecher G, Li M et al (2018) MEGA X: Molecular evolutionary genetics analysis across computing platforms. Mol Biol Evol 35:1547–1549. https://doi.org/10.1093/molbev/msy096
Laatamna AE, Tashiro M, Zokbi Z et al (2021) Molecular characterization and phylogenetic analysis of Fasciola hepatica from high- plateau and steppe areas in Algeria. Parasitol Int 80:102234. https://doi.org/10.1016/j.parint.2020.102234
Malatji MP, Pfukenyi DM, Mukaratirwa S (2019) Fasciola species and their vertebrate and snail intermediate hosts in East and Southern Africa : a review. J Helminthol 94:1–11. https://doi.org/10.1017/S0022149X19000531
Marcilla A, Bargues MD, Mas-Coma S (2002) A PCR-RFLP assay for the distinction between Fasciola hepatica and Fasciola gigantica. Mol Cell Probes 16:327–333. https://doi.org/10.1006/mcpr.2002.0429
Mas-Coma S, Bargues MD (1997) Human liver flukes: a review. Res Rev Parasitol Parasitol 57:145–218
Mas-Coma S, Bargues MD, Valero MA (2005) Fascioliasis and other plant-borne trematode zoonoses. Int J Parasitol 35:1255–1278. https://doi.org/10.1016/j.ijpara.2005.07.010
Mas-Coma S, Valero MA, Bargues MD (2009) Chapter 2 Fasciola, lymnaeids and human fascioliasis, with a global overview on disease transmission, epidemiology, evolutionary genetics, molecular epidemiology and control. Adv Parasitol 69:41–146. https://doi.org/10.1016/S0065-308X(09)69002-3
Nangru A, Maharana B, Vohra S, Kumar B (2022) Molecular identification of Theileria species in naturally infected sheep using nested PCR–RFLP. Parasitol Res 121(5):1487–1497. https://doi.org/10.1007/s00436-022-07489-5
National Institute of Meteorology, Tunisia 2021. https://www.meteo.tn/fr/institut-national-de-la-meteorologie. Consulted on March 2023
Nguyen TBN, Van DN, Nguyen TKL et al (2018) Distribution status of hybrid types in large liver flukes , Fasciola species (Digenea :Fasciolidae), from ruminants and humans in Vietnam. Korean J Parasitol 56:453–461. https://doi.org/10.3347/kjp.2018.56.5.453
Omar MA, Elmajdoub LO, Ali AO et al (2021) Original paper Genetic characterization and phylogenetic analysis of Fasciola species based on ITS2 gene sequence , with first molecular evidence of intermediate Fasciola from water buffaloes in Aswan. Egypt. Ann Parasitol 67(1):55–65. https://doi.org/10.17420/ap6701.312
Periago MV, Valero MA, El Sayed M et al (2008) First phenotypic description of Fasciola hepatica/Fasciola gigantica intermediate forms from the human endemic area of the Nile Delta. Egypt. Infect Genet Evol 8:51–58. https://doi.org/10.1016/j.meegid.2007.10.001
Periago MV, Valero MA, Panova M, Mas-Coma S (2006) Phenotypic comparison of allopatric populations of Fasciola hepatica and Fasciola gigantica from European and African bovines using a computer image analysis system (CIAS). Parasitol Res 99:368–378. https://doi.org/10.1007/s00436-006-0174-3
Robinson MW, John P (2009) Zoonotic helminth infections with particular emphasis on fasciolosis and other trematodiases. Philos Trans R Soc Lond., B, Biol Sci 364:2763–2776. https://doi.org/10.1098/rstb.2009.0089
Sabourin E, Alda P, Vázquez A et al (2018) Impact of human activities on fasciolosis transmission. Trends Parasitol 34(10):891–903. https://doi.org/10.1016/j.pt.2018.08.004
Sahba GH, Arfaa F, Farahmandian I, Jalali H (1972) Animal fascioliasis in Khuzestan, southwestern Iran. J Parasitol 58:712–716. https://doi.org/10.2307/3278298
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4:406–425. https://doi.org/10.1093/oxfordjournals.molbev.a040454
Schwantes JB, Quevedo P, D’Avila MF et al (2020) Fasciola hepatica in Brazil: genetic diversity provides insights into its origin and geographic dispersion. J Helminthol 94:1–7. https://doi.org/10.1017/S0022149X19000774
Shykat CA, Islam S, Ahmed F et al (2022) Current status of Fasciolosis of goat in Sylhet, Bangladesh: an integrated morphomolecular study. J Parasitol Res 2022:1–6. https://doi.org/10.1155/2022/6159388
Sumruayphol S, Siribat P, Dujardin JP et al (2020) Fasciola gigantica, F. Hepatica and Fasciola intermediate forms: geometric morphometrics and an artificial neural network to help morphological identification. PeerJ 8:e8597. https://doi.org/10.7717/peerj.8597
Valero MA, Darce NA, Panova M, Mas-Coma S (2001) Relationships between host species and morphometric patterns in Fasciola hepatica adults and eggs from the northern Bolivian Altiplano hyperendemic region. Vet Parasitol 102:85–100. https://doi.org/10.1016/S0304-4017(01)00499-X
Wang Y, Tian RM, Gao ZM et al (2014) Optimal eukaryotic 18S and universal 16S/18S ribosomal RNA primers and their application in a study of symbiosis. PLoS One 9:e90053. https://doi.org/10.1371/journal.pone.0090053
Yakhchali M, Malekzadeh-Viayeh R, Mani-Baran A, Mardani K (2015) Morphological and molecular discrimination of fasciola species isolated from domestic ruminants of urmia city, iran. Iran J Parasitol 10:46–55
Acknowledgements
The authors would like to thank the staff of the slaughterhouse of the Sejnane region (Tunisia) for their support for sampling. The authors would also like to thank the PREPARE4VBD project (European Union’s Horizon 2020 Research and Innovation Program under grant agreement no. 101000365) for supporting molecular analysis. Thanks also to the Mediterranean and Middle East Universities Network Agreement (MUNA) for collaboration in networking.
Funding
This work was supported by the Laboratoire d’Épidémiologie des Infections Enzootiques des Herbivores en Tunisie: Application à la Lutte (Ministère de l’Enseignement Supérieur et de la Recherche Scientifique, Tunisia) (LR16AGR01), the CGIAR Research Program on Livestock (CRP Livestock), and the Unit of Parasitology and Parasitic Diseases, Department of Veterinary Medicine and Animal Production, University of Naples Federico II.
Author information
Authors and Affiliations
Contributions
Ines Hammami: investigation, methodology, data analyses, writing—original draft. Lavina Ciuca: methodology, writing—review and editing. Maria Paola Maurelli: methodology, writing—review and editing. Rihab Romdhane: investigation. Limam Sassi: investigation. Mohamed Ridha Rjeibi: methodology. Nadia Farhat: investigation. Alain Kouam Simo: investigation, methodology. Laura Rinaldi: funding acquisition, writing—review and editing. Mourad Rekik: funding acquisition. Mohamed Gharbi: conceptualization, supervision, funding acquisition, writing—review and editing.
Corresponding author
Ethics declarations
Ethical approval
The present study was performed in a certified slaughterhouse under the supervision of an officially certified veterinarian by the Tunisian state.
Consent to participate
Informed consent was obtained from the slaughterhouse staff prior to sampling the Fasciola flukes.
Consent for publication
All authors read and consent to the publication of the manuscript.
Conflict of interest
The authors declare no competing interests.
Additional information
Section Editor: Robin Flynn
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hammami, I., Ciuca, L., Maurelli, M.P. et al. First morphometric and molecular characterization of Fasciola spp. in Northwest Tunisia. Parasitol Res 122, 2467–2476 (2023). https://doi.org/10.1007/s00436-023-07933-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-023-07933-0