Abstract
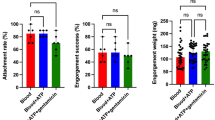
The rearing of ticks is an important technique for studies aiming to elucidate the course and pathogenesis of tick-borne diseases (TBDs). TBDs caused by protozoans (Theileria, Babesia) and bacteria (Anaplasma/Ehrlichia) impose a serious constraint upon livestock health and production in tropical and sub-tropical regions where the distributions of host, pathogen, and vector overlap. This study focuses on Hyalomma marginatum, one of the most important Hyalomma species in the Mediterranean region, being a vector of the virus that causes Crimean-Congo haemorrhagic fever in humans, together with H. excavatum, a vector of Theileria annulata, an important protozoan of cattle. The adaptation of ticks to feeding on artificial membranes allows the creation of model systems that can be put to use examining the underlying mechanisms of pathogen transmission by ticks. Silicone membranes, in particular, offer researchers the flexibility to adjust membrane thickness and content during artificial feeding. The aim of the present study was to develop an artificial feeding technique using silicone-based membranes for all developmental stages of H. excavatum and H. marginatum ticks. Attachment rates after feeding on silicone membranes for females H. marginatum and H. excavatum were 8.33% (8/96) and 7.95% (7/88), respectively. The use of cow hair as a stimulant increased the attachment rate of H. marginatum adults in comparison to other stimulants. The engorgement of H. marginatum and H. excavatum females took 20.5 and 23 days with average weights of 307.85 and 260.64 mg, respectively. Although both tick species could complete egg-laying, and this was followed by hatching of larvae; their larvae and nymphs could not be fed artificially. Taken together, the results of the present study clearly indicate that silicone membranes are suitable for feeding of H. excavatum and H. marginatum adult ticks, supporting engorgement, laying of eggs, and hatching of the larvae. They thus represent a versatile tool for studying transmission mechanisms of tick-borne pathogens. Further studies are warranted to examine attachment and feeding behaviours in order to increase the success of artificial feeding of larvae and nymphal stages.
Graphical abstract



Similar content being viewed by others
Data availability
The authors declare that the data are available.
References
Ahmed BM, Taha KM, Hussein AME (2011) Life cycle of Hyalomma anatolicum Koch, 1844 (Acari: Ixodidae) fed on rabbits, sheep and goats. Vet Parasitol 177:353–358. https://doi.org/10.1016/j.vetpar.2010.12.012
Allan SA (2014) Tick rearing and in vitro feeding. In: Sonenshine DE, Roe RM (eds) Volume II: Biology of ticks, 2nd edn. Oxford University Press, New York, pp 445–473
Antunes S, Merino O, Mosqueda J, Moreno-Cid JA, Bell-Sakyi L, Fragkoudis R, Weisheit S, Pérez de la Lastra JM, Alberdi P, Domingos A, de la Fuente J (2014) Tick capillary feeding for the study of proteins involved in tick-pathogen interactions as potential antigens for the control of tick infestation and pathogen infection. Parasit Vectors 7:42. https://doi.org/10.1186/1756-3305-7-42
Apanaskevich DA, Oliver JH (2014) Life cycles and natural history of ticks. In: Sonenshine DE, Roe RM (eds) Volume II: Biology of ticks, 2nd edn. Oxford University Press, New York, pp 59–73
Bakırcı S, Bilgiç HB, Hacılarlıoğlu S, Ünlü AH, Köse O, Aksulu A, Eren H, Karagenç T (2015) The breeding and colonization of Hyalomma excavatum in laboratory condition. Ankara Univ Vet Fak Derg 62:99–104. https://doi.org/10.1501/Vetfak_0000002665
Bente DA, Forrester NL, Watts DM, McAuley AJ, Whitehouse CA, Bray M (2013) Crimean-Congo hemorrhagic fever: history, epidemiology, pathogenesis, clinical syndrome and genetic diversity. Antivir Res 100(1):159–189. https://doi.org/10.1016/j.antiviral.2013.07.006
Billeter SA, Kasten RW, Killmaster LF, Breitschwerdt EB, Levin ML, Levy MG, Kosoy MY, Chomel BB (2012) Experimental infection by capillary tube feeding of Rhipicephalus sanguineus with Bartonella vinsonii subspecies berkhoffii. Comp Immunol Microbiol Infect Dis 35(1):9–15. https://doi.org/10.1016/j.cimid.2011.09.004
Böhme B, Krull C, Clausen PH, Nijhof AM (2018) Evaluation of a semi-automated in vitro feeding system for Dermacentor reticulatus and Ixodes ricinus adults. Parasitol Res 117(2):565–570. https://doi.org/10.1007/s00436-017-5648-y
Bonnet S, Jouglin M, Malandrin L, Becker C, Agoulon A, L’Hostis M, Chauvin A (2007) Transstadial and transovarial persistence of Babesia divergens DNA in Ixodes ricinus ticks fed on infected blood in a new skin-feeding technique. Parasitology 134:197–207. https://doi.org/10.1017/S0031182006001545
Bonnet S, Liu XY (2012) Laboratory artificial infection of hard ticks: a tool for the analysis of tick-borne pathogen transmission. Acarologia 52:453–464. https://doi.org/10.1051/Acarologia/20122068
Broadwater AH, Sonenshine DE, Hynes WL, Ceraul S, De SA (2002) Glass capillary tube feeding: a method for infecting nymphal Ixodes scapularis (Acari: Ixodidae) with the lyme disease spirochete Borrelia burgdorferi. J Med Entomol 39(2):285–292. https://doi.org/10.1603/0022-2585-39.2.285
de la Fuente J, Estrada Pena A, Venzal JM, Kocan KM, Sonenshine DE (2008) Overview: ticks as vectors of pathogens that cause disease in humans and animals. Front Biosci 13:6938–6946. https://doi.org/10.2741/3200
de la Vega R, Diaz G, Finlay R (2000) Artificial feeding in Boophilus microplus (Acari:Ixodidae) through micropipettes. Ann N Y Acad Sci 916:315–319. https://doi.org/10.1111/j.1749-6632.2000.tb05307.x
Dumanlı N (1983) Elazığ ve yöresinde Hyalomma excavatum (Koch, 1844)’un biyo-ekolojisi üzerinde araştırmalar. TÜBİTAK Doğa Bilim Derg 7(1):23–31
Dusbábek F, Simek P, Jegorov A, Tríska J (1991) Identification of xanthine and hypoxanthine as components of assembly pheromone in excreta of argasid ticks. Exp Appl Acarol 11:307–316. https://doi.org/10.1007/BF01202877
Elhachimi L, Valcárcel F, Olmeda AS, Elasatey S, Khattat SE, Daminet S, Sahibi H, Duchateau L (2021) Rearing of Hyalomma marginatum Acarina: Ixodidae) under laboratory conditions in Morocco. Exp Appl Acarol 84:785–794. https://doi.org/10.1007/s10493-021-00641-3
Gargili A, Thangamani S, Bente D (2013) Influence of laboratory animal hosts on the life cycle of Hyalomma marginatum and implications for an in vivo transmission model for Crimean-Congo hemorrhagic fever virus. Front Cell Infect Microbiol 3:39. https://doi.org/10.3389/fcimb.2013.00039
Gonzalez J, Valcarcel F, Aguilar A, Olmeda AS (2017) In vitro feeding of Hyalomma lusitanicum ticks on artificial membrane. Exp Appl Acarol 72:449–459. https://doi.org/10.1007/s10493-017-0167-1
González J, Bickerton M, Toledo A (2021) Applications of artificial membrane feeding for ixodid ticks. Acta Trop 215:105818. https://doi.org/10.1016/j.actatropica.2020.105818
Graham ML, Prescott MJ (2015) The multifactorial role of the 3Rs in shifting the harm-benefit analysis in animal models of disease. Eur J Pharmacol 759:19–29
Hatta T, Miyoshi T, Matsubayashi M, Islam MdK, Alim MA, Anisuzzaman Yamaji K, Fujisaki K, Tsuji N (2012) Semi-artificial mouse skin membrane feeding technique for adult tick, Haemaphysalis Longicornis. Parasit Vectors 5:263. https://doi.org/10.1186/1756-3305-5-263
Jongejan F, Uilenberg G (2004) The global importance of ticks. Parasitology 129(1):3–14. https://doi.org/10.1017/s0031182004005967
Kar S, Yımazer N, Akyıldız G, Gargılı A (2017) The human infesting ticks in the city of Istanbul and its vicinity with reference to a new species for Turkey. Syst Appl Acarol 22(14):2245–2255. https://doi.org/10.11158/saa.22.12.14
Klompen JSH, Black WC, Keirans JE, Oliver JH (1996) Evolution of ticks. Annu Rev Entomol 41:141–161. https://doi.org/10.1146/annurev.en.41.010196.001041
Korshus JB, Munderloh UG, Bey RF, Kurtti TJ (2004) Experimental infection of dogs with Borrelia burgdorferi sensu stricto using Ixodes scapularis ticks artificially infected by capillary feding. Med Microbiol Immunol 193:27–34. https://doi.org/10.1007/s00430-003-0178-x
Kröber T, Guerin PM (2007a) In vitro feeding assays for hard ticks. Trends Parasitol 23:445–449. https://doi.org/10.1016/j.pt.2007.07.010
Kröber T, Guerin PM (2007b) An in vitro feeding assay to test acaricides for control of hard ticks. Pest Manag Sci 63:17–22. https://doi.org/10.1002/ps.1293
Krull C, Böhme B, Clausen PH, Nijhof AM (2017) Optimization of an artificial tick feeding assay for Dermacentor reticulatus. Parasit Vectors 10:60. https://doi.org/10.1186/s13071-017-2000-4
Lew-Tabor AE, Bruyeres AG, Zhang B, Valle MR (2014) Rhipicephalus (Boophilus) microplus tick in vitro feeding methods for functional (dsRNA) and vaccine candidate (antibody) screening. Ticks Tick Borne Dis 5:500–510. https://doi.org/10.1016/j.ttbdis.2014.03.005
Midilli K, Gargili A, Ergonul O, Sengöz G, Ozturk R, Bakar M, Jongejan F (2007) Imported Crimean-Congo hemorrhagic fever cases in Istanbul. BMC Infect Dis 7:54. https://doi.org/10.1186/1471-2334-7-54
Militzer N, Bartel A, Clausen PH, Hoffmann-Köhler P, Nijhof AM (2021) Artificial feeding of all consecutive life stages of Ixodes ricinus. Vaccine 9:385. https://doi.org/10.3390/vaccines9040385
Musyoki JM, Osir EO, Kiara HK, Kokwaro ED (2004) Comparative studies on the infectivity of Theileria parva in ticks fed in vitro and those fed on cattle. Exp Appl Acarol 32:51–67. https://doi.org/10.1023/B:APPA.0000018159.47700.e4
Ölmez N (2019) In vitro feeding of Dermacentor reticulatus (Fabricius, 1794) and Hyalomma marginatum (Koch, 1844). PhD Thesis, Kafkas University, Kars, Turkey
Orkun O, Karaer Z, Cakmak A, Nalbantoglu S (2014) Identification of tick-borne pathogens in ticks feeding on humans in Turkey. PLoS Negl Trop Dis 8:e3067. https://doi.org/10.1371/journal.pntd.0003067
Orkun O, Karaer Z, Cakmak A, Nalbantoglu S (2014b) Spotted fever group rickettsiae in ticks in Turkey. Ticks Tick Borne Dis 5:213–218. https://doi.org/10.1016/j.ttbdis.2012.11.018
Ouhelli H (1994) Comparative development of Hyalomma marginatum (Koch, 1844), H. detritum (Schulze, 1919), H. anatolicum excavatum (Koch, 1844), H. lusitanicum (Koch, 1844) and H. dromedarii (Koch, 1844) under laboratory conditions. Acta Parasitol 39:153–157
Parola P, Paddock CD, Socolovschi C, Labruna MB, Mediannikov O, Kernif T, Abdad MY, Stenos J, Bitam I, Fournier PE, Raoult D (2013) Update on tick-borne rickettsioses around the world: a geographic approach. Clin Microbiol Rev 26:657–702
Psaroulaki A, Germanakis A, Gikas A, Scoulica E, Tselentis Y (2005) Simultaneous detection of “Rickettsia mongolotimonae” in a patient and in a tick in Greece. J Clin Microbiol 43:3558–3559
Sajid MS, Kausar A, Iqbal A, Abbas H, Iqbal Z, Jones MK (2018) An insight into the ecobiology, vector significance and control of Hyalomma ticks (Acari: Ixodidae): a review. Acta Trop 187:229–239. https://doi.org/10.1016/j.actatropica.2018.08.016
Santos-Silva MM, Vatansever Z (2017) Hyalomma marginatum. In: Estrada-Peña A, Mihalca AD, Petney TN (eds) Ticks of Europe and North Africa, a guide to species identification. Springer, Switzerland, pp 349–354
Soares CAG, Lima CMR, Dolan MC, Piesman J, Beard CB, Zeidner NS (2005) Capillary feeding of specific dsRNA induces silencing of the isac gene in nymphal Ixodes scapularis ticks. Insect Mol Biol 14:443–452. https://doi.org/10.1111/j.1365-2583.2005.00575.x
Sonenshine DE (2004) Pheromones and other semiochemicals of ticks and their use in tick control. Parasitology 129:405–425. https://doi.org/10.1017/S003118200400486X
Sonenshine DE (2006) Tick pheromones and their use in tick control. Annu Rev Entomol 51:557–580. https://doi.org/10.1146/annurev.ento.51.110104.151150
Sonenshine DE, Roe RM (2014) Overview: ticks, people, and animals. In: Sonenshine DE, Roe RM (eds) Volume I: Biology of ticks, 2nd edn. Oxford University Press, New York, pp 3–16
Sutherst RW (2004) Global change and human vulnerability to vector-borne diseases. Clin Microbiol Rev 17:136–173. https://doi.org/10.1128/CMR.17.1.136-173.2004
Tajeri S, Razmi G, Haghparast A (2016) Establishment of an artificial tick feeding system to study Theileria lestoquardi infection. PloS One 11(12):e0169053. https://doi.org/10.1371/journal.pone.0169053
Tajeri S, Razmi GR (2011) Hyalomma anatolicum anatolicum and Hyalomma dromedarii (Acari: Ixodidae) imbibe ovine blood in vitro by utilizing an artificial feeding system. Vet Parasitol 180:332–335. https://doi.org/10.1016/j.vetpar.2011.03.014
Troughton DR, Levin ML (2007) Life cycles of seven ixodid tick species (Acari: Ixodidae) under standardized laboratory conditions. J Med Entomol 44(5):732–740. https://doi.org/10.1093/jmedent/44.5.732
Turell MJ (2007) Role of ticks in the transmission of Crimean-Congo hemorrhagic fever virus. In: Ergonul O, Whitehouse CA (eds) Crimean-Congo Hemorrhagic Fever: a global perspective, 1st edn. Springer, Berlin, Germany, pp 143–154
Valcárcel F, González J, González MG, Sánchez M, Tercero JM, Elhachimi L, Carbonell JD, Olmeda AS (2020) Comparative ecology of Hyalomma lusitanicum and Hyalomma marginatum Koch, 1844 (Acarina: Ixodidae). Insects 11(5):303. https://doi.org/10.3390/insects11050303
Waladde SM, Kemp DH, Rice MJ (1979) Feeding electrograms and fluid uptake measurements of cattle tick Boophilus microplus attached on artificial membranes. Int J Parasitol 9:89–95
Waladde SM, Ochieng SA, Gichuhi PM (1991) Artificial-membrane feeding of the ixodid tick, Rhipicephalus appendiculatus, to repletion. Exp Appl Acarol 11:297–306
Weidmann M, Avsic-Zupanc T, Bino S et al (2016) Biosafety standards for working with Crimean-Congo hemorrhagic fever virus J Gen Virol 97(11):2799–2808. https://doi.org/10.1099/jgv.0.000610
Wikel SK (2014) Tick-host interactions. In: Sonenshine DE, Roe RM (eds) Volume II: Biology of ticks, 2nd edn. Oxford University Press, New York, pp 88–128
Yukarı BA (1993) Laboratuarda Hyalomma anatolicum excavatum (Koch, 1844) Kolonisinin elde edilmesi ve muhafazası. Ankara Üniv Vet Fak Derg 40:99–114
Yukarı BA, Nalbantoğlu S, Karaer Z, İnci A, Eren H, Sayın F (2011) Some biological features of Hyalomma marginatum in the laboratory. Turkiye Parazitol Derg 35:40–42. https://doi.org/10.5152/tpd.2011.10
Acknowledgements
The authors would like to thank Prof. Willie Weir from the Univ. of Glasgow (https://www.gla.ac.uk/schools/bohvm/staff/willieweir/#) for his valuable contribution to the manuscript.
Funding
Financial support for this study was provided by a grant from TUBITAK (grant number: TUBITAK-118O904).
Author information
Authors and Affiliations
Contributions
Conception and design of study: Serkan Bakirci, Hüseyin Bilgin Bilgiç, and Tülin Karagenç; acquisition of data: Serkan Bakirci, Hüseyin Bilgin Bilgiç, Selin Hacilarlioğlu, and Metin Pekağirbaş; analysis and/or interpretation of data: Serkan Bakirci, Hüseyin Bilgin Bilgiç, and Tülin Karagenç; drafting the manuscript: Serkan Bakirci, Hüseyin Bilgin Bilgiç, and Tülin Karagenç; revising the manuscript critically for important intellectual content: Hüseyin Bilgin Bilgiç, Hasan Eren, and Serkan Bakirci; approval of the version of the manuscript to be published: Serkan Bakirci, Hüseyin Bilgin Bilgiç, Tülin Karagenç, Selin Hacilarlioğlu, Metin Pekağirbaş, and Hasan Eren.
Corresponding author
Ethics declarations
Ethics approval
All the clinical/laboratory experiments with laboratory animals (gerbil and rabbits) were approved by the Animal Experiment Ethics Committee of Aydin Adnan Menderes University on the 31st May 2018 in accordance with decision number 64583101/2018/074.
Consent to participate
The authors declare their consent to participate.
Consent for publication
The authors declare their consent for publication.
Competing interests
The authors declare no competing interests.
Additional information
Section Editor: Van Lun Low
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Bilgiç, H.B., Hacilarlioğlu, S., Pekağirbaş, M. et al. In vitro feeding of Hyalomma excavatum and Hyalomma marginatum tick species. Parasitol Res 122, 1641–1649 (2023). https://doi.org/10.1007/s00436-023-07867-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-023-07867-7