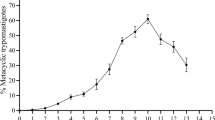
Abstract
Trypanosoma cruzi has a complex life cycle consisting of four morphological and distinct biological stages. Although some authors suggest that T. cruzi primarily follows clonal reproduction, recent genomic and transcriptomic studies indicate an unorthodox capacity for recombination. We aimed to estimate the differential gene expression of 10 meiosis/homologous recombination-related genes during the T. cruzi life cycle, including epimastigotes, under two different types of stress (oxidative stress and pH changes). We performed RT-qPCR tests using novel-designed primers to estimate the differential gene expression (∆Ct and ∆∆Ct) of nine genes (SPO11, HAP2, RAD50, MRN complex, BRCA2, DMC1, MND1, and RPA1) and RAD51, which was previously reported. Our results show basal expression of all genes during the life cycle, indicating their hypothetical role in several cellular processes but with specific signatures of differential gene expression during the life cycle (HAP2, RPA, RAD50, BRCA2, MND1, and DMC1) and oxidative stress (RPA, MRE11, NBS1, BRCA2, MND1, and RAD51). Additionally, we found that the MRN complex has an independent level of expression in T. cruzi, with profiles of MRE11 and NBS1 upregulated in some stages. Recent studies on other trypanosomatids have highlighted the influence of HAP2 and RPA in recombination and hybridization. If T. cruzi uses the same repertoire of genes, our findings could suggest that metacyclogenesis may be the putative step that the parasite uses to undergo recombination. Likewise, our study reveals the differential profiles of genes expressed in response to oxidative and pH stress. Further studies are necessary to confirm our findings and understand the recombination mechanism in T. cruzi.



Similar content being viewed by others
Data availability
All the sequences for meiosis and HR-related genes were deposited in NCBI GenBank under the accession numbers ON746177-ON746187.
References
Alvarez VE, Kosec G, Sant’Anna C et al (2008) Autophagy is involved in nutritional stress response and differentiation in Trypanosoma cruzi. J Biol Chem 283:3454–3464. https://doi.org/10.1074/jbc.M708474200
Alves CL, Repolês BM, da Silva MS et al (2018) The recombinase Rad51 plays a key role in events of genetic exchange in Trypanosoma cruzi. Sci Rep 8:13335. https://doi.org/10.1038/s41598-018-31541-z
Bekker-Jensen S, Mailand N (2010) Assembly and function of DNA double-strand break repair foci in mammalian cells. DNA Repair (amst) 9:1219–1228. https://doi.org/10.1016/j.dnarep.2010.09.010
Berry ASF, Salazar-Sánchez R, Castillo-Neyra R et al (2019) Sexual reproduction in a natural Trypanosoma cruzi population. PLoS Negl Trop Dis 13:e0007392. https://doi.org/10.1371/journal.pntd.0007392
Cruz-Saavedra L, Muñoz M, León C et al (2017) Purification of Trypanosoma cruzi metacyclic trypomastigotes by ion exchange chromatography in sepharose-DEAE, a novel methodology for host-pathogen interaction studies. J Microbiol Methods 142:27–32. https://doi.org/10.1016/j.mimet.2017.08.021
Cruz-Saavedra L, Muñoz M, Patiño LH et al (2020) Slight temperature changes cause rapid transcriptomic responses in Trypanosoma cruzi metacyclic trypomastigotes. Parasit Vectors 13:255. https://doi.org/10.1186/s13071-020-04125-y
Cruz-Saavedra L, Vallejo GA, Guhl F et al (2020) Transcriptional remodeling during metacyclogenesis in Trypanosoma cruzi I. Virulence 11:969–980. https://doi.org/10.1080/21505594.2020.1797274
Cruz-Saavedra L, Vallejo GA, Guhl F, Ramírez JD (2020) Transcriptomic changes across the life cycle of Trypanosoma cruzi II. PeerJ 8:e8947. https://doi.org/10.7717/peerj.8947
Cruz-Saavedra L, Schwabl P, Vallejo GA, Carranza JC, Muñoz M, Patino LH, Paniz-Mondolfi A, Llewellyn MS, Ramírez JD (2022) Genome plasticity driven by aneuploidy and loss of heterozygosity in Trypanosoma cruzi. Microb Genom 8(6):mgen000843. https://doi.org/10.1099/mgen.0.000843
da Silva MS (2021) DNA double-strand breaks: a double-edged sword for trypanosomatids. Front Cell Dev Biol 9:669041. https://doi.org/10.3389/fcell.2021.669041
Gaunt MW, Yeo M, Frame IA et al (2003) Mechanism of genetic exchange in American trypanosomes. Nature 421:936–939. https://doi.org/10.1038/nature01438
Gillespie DA, Ryan KM (2015) Autophagy is critically required for DNA repair by homologous recombination. Mol Cell Oncol 3:e1030538. https://doi.org/10.1080/23723556.2015.1030538
Gomes LR, Menck CFM, Leandro GS (2017) Autophagy roles in the modulation of DNA repair pathways. Int J Mol Sci 18:2351. https://doi.org/10.3390/ijms18112351
Gomes Passos Silva D, da SilvaSantos S, Nardelli SC et al (2018) The in vivo and in vitro roles of Trypanosoma cruzi Rad51 in the repair of DNA double strand breaks and oxidative lesions. PLoS Negl Trop Dis 12:e0006875. https://doi.org/10.1371/journal.pntd.0006875
Howick VM, Peacock L, Kay C et al (2022) Single-cell transcriptomics reveals expression profiles of Trypanosoma brucei sexual stages. PLoS Pathog 18:e1010346. https://doi.org/10.1371/journal.ppat.1010346
Keeney S (2008) Spo11 and the formation of DNA double-strand breaks in meiosis. Genome Dynamics Stab 2:81. https://doi.org/10.1007/7050_2007_026
Louradour I, Ferreira TR, Duge E et al (2022) Stress conditions promote Leishmania hybridization in vitro marked by expression of the ancestral gamete fusogen HAP2 as revealed by single-cell RNA-seq. Elife 11:e73488. https://doi.org/10.7554/eLife.73488
Maréchal A, Zou L (2015) RPA-coated single-stranded DNA as a platform for post-translational modifications in the DNA damage response. Cell Res 25:9–23. https://doi.org/10.1038/cr.2014.147
Matos GM, Lewis MD, Talavera-López C et al (2022) Microevolution of Trypanosoma cruzi reveals hybridization and clonal mechanisms driving rapid genome diversification. Elife 11:e75237. https://doi.org/10.7554/eLife.75237
Messenger LA, Miles MA (2015) Evidence and importance of genetic exchange among field populations of Trypanosoma cruzi. Acta Trop 151:150–155. https://doi.org/10.1016/j.actatropica.2015.05.007
Paes MC, Cosentino-Gomes D, de Souza CF et al (2011) The role of heme and reactive oxygen species in proliferation and survival of Trypanosoma cruzi. J Parasitol Res 2011:174614. https://doi.org/10.1155/2011/174614
Paula JIO, da Pinto JS, Rossini A et al (2020) New perspectives for hydrogen peroxide in the amastigogenesis of Trypanosoma cruzi in vitro. Biochim Biophys Acta Mol Basis Dis 1866:165951. https://doi.org/10.1016/j.bbadis.2020.165951
Peacock L, Kay C, Farren C et al (2021) Sequential production of gametes during meiosis in trypanosomes. Commun Biol 4:555. https://doi.org/10.1038/s42003-021-02058-5
Pedra-Rezende Y, Fernandes MC, Mesquita-Rodrigues C et al (2021) Starvation and pH stress conditions induced mitochondrial dysfunction, ROS production and autophagy in Trypanosoma cruzi epimastigotes. Biochim Biophys Acta Mol Basis Dis 1867:166028. https://doi.org/10.1016/j.bbadis.2020.166028
Petukhova GV, Pezza RJ, Vanevski F et al (2005) The Hop2 and Mnd1 proteins act in concert with Rad51 and Dmc1 in meiotic recombination. Nat Struct Mol Biol 12:449–453. https://doi.org/10.1038/nsmb923
Ramírez JD, Duque MC, Montilla M et al (2012a) Natural and emergent Trypanosoma cruzi I genotypes revealed by mitochondrial (Cytb) and nuclear (SSU rDNA) genetic markers. Exp Parasitol 132:487–494. https://doi.org/10.1016/j.exppara.2012.09.017
Ramírez JD, Guhl F, Messenger LA et al (2012b) Contemporary cryptic sexuality in Trypanosoma cruzi. Mol Ecol 21:4216–4226. https://doi.org/10.1111/j.1365-294X.2012.05699.x
Reis-Cunha JL, Baptista RP, Rodrigues-Luiz GF et al (2018) Whole genome sequencing of Trypanosoma cruzi field isolates reveals extensive genomic variability and complex aneuploidy patterns within TcII DTU. BMC Genomics 19:816. https://doi.org/10.1186/s12864-018-5198-4
Schwabl P, Imamura H, Van den Broeck F et al (2019) Meiotic sex in Chagas disease parasite Trypanosoma cruzi. Nat Commun 10:3972. https://doi.org/10.1038/s41467-019-11771-z
Szurman-Zubrzycka M, Baran B, Stolarek-Januszkiewicz M et al (2019) The dmc1 mutant allows an insight into the DNA double-strand break repair during meiosis in barley (Hordeum vulgare L.). Front Plant Sci 10:761. https://doi.org/10.3389/fpls.2019.00761
Teixeira DE, Benchimol M, Crepaldi PH, de Souza W (2012) Interactive multimedia to teach the life cycle of Trypanosoma cruzi, the causative agent of Chagas disease. PLoS Negl Trop Dis 6:e1749. https://doi.org/10.1371/journal.pntd.0001749
Thorslund T, Esashi F, West SC (2007) Interactions between human BRCA2 protein and the meiosis-specific recombinase DMC1. EMBO J 26(12):2915–2922. https://doi.org/10.1038/sj.emboj.7601739
Tibayrenc M, Ayala FJ (1999) Evolutionary genetics of Trypanosoma and Leishmania. Microbes Infect 1:465–472. https://doi.org/10.1016/s1286-4579(99)80050-1
Tibayrenc M, Ayala FJ (2022) Microevolution and subspecific taxonomy of Trypanosoma cruzi. Infect Genet Evol 103:105344. https://doi.org/10.1016/j.meegid.2022.105344
Van Der Heyden N, Docampo R (2000) Intracellular pH in mammalian stages of Trypanosoma cruzi is K+-dependent and regulated by H+-ATPases. Mol Biochem Parasitol 105:237–251. https://doi.org/10.1016/s0166-6851(99)00184-x
Vanrell MC, Losinno AD, Cueto JA et al (2017) The regulation of autophagy differentially affects Trypanosoma cruzi metacyclogenesis. PLoS Negl Trop Dis 11:e0006049. https://doi.org/10.1371/journal.pntd.0006049
Wang W, Peng D, Baptista RP et al (2021) Strain-specific genome evolution in Trypanosoma cruzi, the agent of Chagas disease. PLoS Pathog 17:e1009254. https://doi.org/10.1371/journal.ppat.1009254
Warrenfeltz S, Basenko EY, Crouch K et al (2018) EuPathDB: the eukaryotic pathogen genomics database resource. Methods Mol Biol 1757:69–113. https://doi.org/10.1007/978-1-4939-7737-6_5
Acknowledgements
We thank Duvan Morales for his support during the RT-qPCR analysis. We would like to acknowledge the assistance of ChatGPT, a language model trained by OpenAI, for providing helpful insights and corrections during the process of improving the drafting and readability.
Funding
This work was funded by the Dirección de Investigación e Innovación at the Universidad del Rosario.
Author information
Authors and Affiliations
Contributions
LCS designed the primers, standardized the RT-qPCR, made the RNA extraction, performed the RT-qPCR, analyzed the data, and wrote the manuscript. TC made the survival curves and RNA extraction. NB supported the RT-qPCR standardization. BD participated during the primers design. JDR conceived the manuscript, supervised the project, and wrote, reviewed, and edited the manuscript. All authors approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Handling Editor: Una Ryan
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Below is the link to the electronic supplementary material.
ESM 1
Supplementary file1 Primers and PCR conditions for nine pairs of primers including the previously designed primers that amplify RAD51 (Table. S1) (Alves et al. 2018). (XLSX 11 kb)
ESM 2
Supplementary file2 Anova one-way test comparing the expression of meiosis and HR-related genes during the life cycle of Trypanosoma cruzi. (XLSX 26 kb)

ESM 3
Electrophoresis agarose gel for the ten meiosis and HR related genes. (PNG 1813 kb)

ESM 4
Interaction plot of Meiosis and homologous recombination-related genes during the whole T. cruzi life cycle. The y axes correspond to the values of ΔCt calculated, the x axes list the genes evaluated. (PNG 1032 kb)

ESM 5
Interaction plot of Meiosis and homologous recombination-related genes during oxidative stress in epimastigotes, metacyclic trypomastigotes and cell-derived trypomastigotes. The y axes correspond to the values of ΔΔCt calculated, the x axes list the genes evaluated. A interaction plot per stage was included. (PNG 2181 kb)

ESM 6
Interaction plot of Meiosis and homologous recombination-related genes during pH changes in epimastigotes, metacyclic trypomastigotes and cell-derived trypomastigotes. The y axes correspond to the values of ΔΔCt calculated, the x axes list the genes evaluated. A interaction plot per stage was included. (PNG 2942 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Cruz-Saavedra, L., Caceres, T., Ballesteros, N. et al. Differential expression of meiosis and homologous recombination-related genes in the life cycle of Trypanosoma cruzi. Parasitol Res 122, 1747–1757 (2023). https://doi.org/10.1007/s00436-023-07850-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-023-07850-2