Abstract
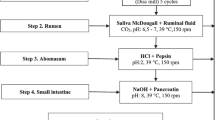
This study developed and evaluated an in vitro rumen incubation (IVRI) method to describe the exsheathment kinetics of Haemonchus contortus third-stage infective larvae (L3) in ruminal liquor (RL). The specific objectives were (i) to standardize the IVRI method to facilitate the contact between L3 and RL as well as the larval recovery, and (ii) to apply the IVRI method to describe the exsheathment kinetics of H. contortus and to select the best fitting nonlinear model. Incubation devices containing H. contortus larvae were incubated according to the IVRI technique in cattle RL or PBS. The incubation conditions included RL mixed with a nitrogen-rich media, maintained at 39 °C, with pH = 7.0, vented with CO2 and manual agitation. The larvae were recovered after 0, 1, 3, 6, 9, 12, and 24 h. The exsheathed and ensheathed larvae were counted to estimate the exsheathment (%) in RL or PBS. Exsheathment in RL was analyzed with nonlinear regression models: Exponential, Gompertz, Logistic, Log-Logistic, and Weibull. The models’ fit was compared to select the one that best described the exsheathment kinetics. The exsheathment in RL reached 6.52%, 20.65%, 58.22%, 69.24%, 73.08%, and 77.20% in 1, 3, 6, 9, 12, and 24 h, respectively. Although the Gompertz, Weibull, and Logistic models were adequate to describe the observed exsheathment, the Log-Logistic model had the best fit. The IVRI method using bovine RL represents a suitable tool for the study of the in vitro exsheathment kinetics of H. contortus L3.



Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Agricultural and Food Research Council (AFRC) (1993) Energy and protein requirements of ruminants. In: An advisory manual prepared by the AFRC Technical Committee on Responses to Nutrients. CAB International, Wallingford
Amanzougarene Z, Fondevila M (2020) Fitting of the in vitro gas production technique to the study of high concentrate diets. Animals 10:1935. https://doi.org/10.3390/ani10101935
Azzolina NA, Nakles DV, Gorecki CD, Peck WD, Ayash SC, Melzer LS, Chatterjee S (2015) CO2 storage associated with CO2 enhanced oil recovery: a statistical analysis of historical operations. Int J Greenh Gas Control 37:384–397. https://doi.org/10.1016/j.ijggc.2015.03.037
Barros-Rodríguez M, Solorio-Sánchez J, Ku-Vera J, Ayala-Burgos A, Sandoval-Castro C, Solís-Pérez G (2012) Productive performance and urinary excretion of mimosine metabolites by hair sheep grazing in a silvopastoral system with high densities of Leucaena leucocephala. Trop Anim Health Prod 44:1873–1878. https://doi.org/10.1007/s11250-012-0150-0
Bekelaar K, Waghorn T, Tavendale M, McKenzie C, Leathwick D (2018a) Carbon dioxide is an absolute requirement for exsheathment of some, but not all, abomasal nematode species. Parasitol Res 117:3675–3678. https://doi.org/10.1007/s00436-018-6094-1
Bekelaar K, Waghorn T, Tavendale M, McKenzie C, Leathwick D (2018b) Heat shock, but not temperature, is a biological trigger for the exsheathment of third-stage larvae of Haemonchus contortus. Parasitol Res 117:2395–2402. https://doi.org/10.1007/s00436-018-5927-2
Bekelaar K, Waghorn T, Tavendale M, McKenzie C, Leathwick D (2019) Abomasal nematode species differ in their in vitro response to exsheathment triggers. Parasitol Res 118:707–710. https://doi.org/10.1007/s00436-018-6183-1
Brunet S, Aufrere J, El Babili F, Fouraste I, Hoste H (2007) The kinetics of exsheathment of infective nematode larvae is disturbed in the presence of a tannin-rich plant extract (sainfoin) both in vitro and in vivo. Parasitol 134:1253–1262. https://doi.org/10.1017/S0031182007002533
Bueno ICS, Cabral-Filho SLS, Gobbo SP, Louvandini H, Vitti DMSS, Abdalla AL (2005) Influence of inoculum source in a gas production method. Anim Feed Sci Technol 123-124:95–105. https://doi.org/10.1016/j.anifeedsci.2005.05.003
Burnham KP, Anderson DR (2004) Multimodel inference. Sociol Methods Res 33:261–304. https://doi.org/10.1177/0049124104268644
Castagna F, Palma E, Cringoli G, Bosco A, Nisticò N, Caligiuri G, Britti D, Musella V (2019) Use of complementary natural feed for gastrointestinal nematodes control in sheep: effectiveness and benefits for animals. Animals 9:1037. https://doi.org/10.3390/ani9121037
Castañeda-Ramírez GS, Mathieu C, Vilarem G, Hoste H, Mendoza-de-Gives P, González-Pech PG, Torres-Acosta JFJ, Sandoval-Castro CA (2017) Age of Haemonchus contortus third stage infective larvae is a factor influencing the in vitro assessment of anthelmintic properties of tannin containing plants. Vet Parasitol 243:130–134. https://doi.org/10.1016/j.vetpar.2017.06.019
Chan-Pérez JI, Torres-Acosta JFJ, Sandoval-Castro CA, Castañeda-Ramírez GS, Vilarem G, Mathieu C, Hoste H (2017) Susceptibility of ten Haemonchus contortus isolates from different geographical origins towards acetone:water extracts of polyphenol-rich plants. Part 2: infective L3 larvae. Vet Parasitol 240:11–16. https://doi.org/10.1016/j.vetpar.2017.04.023
Church DC (1993) The ruminant animal: digestive physiology and nutrition. Waveland Press, New Jersey
Czerkawski JW, Breckenridge G (1977) Design and development of a long-term rumen simulation technique (Rusitec). Brit J Nutr 38:371–384. https://doi.org/10.1079/BJN19770102
Do DN, Miar Y (2019) Evaluation of growth curve models for body weight in american mink. Animals 10:22. https://doi.org/10.3390/ani10010022
Galicia-Aguilar HH, Rodríguez-González LA, Capetillo-Leal CM, Cámara-Sarmiento R, Aguilar-Caballero AJ, Sandoval-Castro CA, Torres-Acosta JFJ (2012) Effects of Havardia albicans supplementation on feed consumption and dry matter digestibility of sheep and the biology of Haemonchus contortus. Anim Feed Sci Technol 176:178–184. https://doi.org/10.1016/j.anifeedsci.2012.07.021
García E (1973) Modificaciones al sistema de clasificación climática de Köppen. Instituto de Geografía. Universidad Nacional Autónoma de México, México
Getachew G, DePeters E, Robinson PH, Fadel JG (2005) Use of an in vitro rumen gas production technique to evaluate microbial fermentation of ruminant feeds and its impact on fermentation products. Anim Feed Sci Technol 123-124:547–559. https://doi.org/10.1016/j.anifeedsci.2005.04.034
GraphPad Prism version 8.0.0 for Windows (2018) GraphPad Software, California. https://www.graphpad.com
Henderson G, Cox F, Ganesh S, Jonker A, Young W, Janssen PH (2015) Rumen microbial community composition varies with diet and host, but a core microbiome is found across a wide geographical range. Sci Rep 5:14567. https://doi.org/10.1038/srep14567
Hertzberg H, Huwyler U, Kohler L, Rehbein S, Wanner M (2002) Kinetics of exsheathment of infective ovine and bovine strongylid larvae in vivo and in vitro. Parasitol 125:65–70. https://doi.org/10.1017/S0031182002001816
Hojjati F, Hossein-Zadeh NG (2017) Comparison of non-linear growth models to describe the growth curve of Mehraban sheep. J Appl Animal Res 46:499–504. https://doi.org/10.1080/09712119.2017.1348949
Jackson F, Hoste H (2010) In vitro methods for the primary screening of plant products for direct activity against ruminant. In: Vercoe PE, Makkar HPS, Schlink AC (eds) in vitro screening of plant resources for extra-nutritional attributes in ruminant: Nuclear and related methodologies. Springer Science+Business Media, USA, pp 25–46
Lonngren KJ, Barone CD, Zajac AM, Brown RN, Reed JD, Krueger CG, Petersson KH (2020) Effect of birdsfoot trefoil cultivars on exsheathment of Haemonchus contortus in fistulated sheep. Vet Parasitol 287:109271. https://doi.org/10.1016/j.vetpar.2020.109271
López S, Prieto M, Dijkstra J, Dhanoa MS, France J (2004) Statistical evaluation of mathematical models for microbial growth. Int J Food Microbiol 96:289–300. https://doi.org/10.1016/j.ijfoodmicro.2004.03.026
Makkar HPS (2005) In vitro gas methods for evaluation of feeds containing phytochemicals. Anim Feed Sci Technol 123-124:291–302. https://doi.org/10.1016/j.anifeedsci.2005.06.003
Mauricio RM, Mould FL, Dhanoa M, Owen E, Channa KS, Theodorou MK (1999) A semi-automated in vitro gas production technique for ruminant feedstuff evaluation. Anim Feed Sci Technol 79:321–330. https://doi.org/10.1016/S0377-8401(99)00033-4
Menke K, Steingass H (1988) Estimation of the energetic feed value obtained from chemical analysis and in vitro gas production using rumen liquid. Anim Res Dev 28:55
Ministry of Agriculture, Fisheries and Food (MAFF) (1986) Manual of veterinary parasitological laboratory techniques (Reference Book; 418), 3rd edn. Her Majesty’s Stationery Office (HMSO), London, p 160
Minitab 18 Statistical Software (2017) Minitab, Inc, Pennsylvania. https://www.minitab.com
Miranda-Miranda DY, Mancilla-Montelongo G, González-Pech PG, Sandoval-Castro CA, Torres-Acosta JFJ (2021) La combinación de extractos de plantas nutraceúticas pueden inducir efectos de sinergia o antagonismo contra Haemonchus contortus en condiciones in vitro. Cartel. XXVI Congreso Panamericano de Ciencias Veterinarias 2021. Virtual (México) 24-26 nov 2021
Monforte-Briceño GE, Sandoval-Castro CA, Ramírez-Avilés L, Capetillo Leal CM (2005) Defaunating capacity of tropical forage trees. Effect of PEG and its relationship with in vitro gas production. Anim Feed Sci Tech 123-124:313–327. https://doi.org/10.1016/j.anifeedsci.2005.04.016
Motulsky HJ, Christopoulos A (2003) Fitting models to biological data using linear and nonlinear regression. A practical guide to curve fitting. GraphPad Software Inc., California
Noel ZA, Wang J, Chilvers MI (2018) significant influence of EC50 estimation by model choice and EC50 type. Plant Dis 102:708–714. https://doi.org/10.1094/PDIS-06-17-0873-SR
Ortíz-Domínguez GA, Marin-Tun CG, Ventura-Cordero J, González-Pech PG, Capetillo-Leal CM, Torres-Acosta JFJ, Sandoval-Castro CA (2021) Comparing the in vitro digestibility of leaves from tropical trees when using the rumen liquor from cattle, sheep or goats. Small Rumin Res 205:106561. https://doi.org/10.1016/j.smallrumres.2021.106561
Peleg M, Corradini MG, Normand MD (2004) Kinetic models of complex biochemical reactions and biological processes. Chem Ing Tech 76:413–423. https://doi.org/10.1002/cite.200406157
Sandoval-Castro CA, Capetillo C, Cetina R, Ramirez L (2002) A mixture simplex design to study associative effects with the in vitro gas production technique. Anim Feed Sci Tech. 101:191–200. https://doi.org/10.1016/S0377-8401(02)00137-2
Schabenberger O, Tharp BE, Kells JJ, Penner D (1999) Statistical tests for hormesis and effective dosages in herbicide dose response. Agron J 91:713. https://doi.org/10.2134/agronj1999.914713x
Seefeldt SS, Jensen JE, Fuerst EP (1995) Log-Logistic analysis of herbicide dose-response relationships. Weed Tech 9:218–227. https://doi.org/10.1017/S0890037X00023253
Sepúlveda-Vázquez J, Lara-Del Rio MJ, Vargas-Magaña JJ, Quintal-Franco JA, Alcaraz-Romero RA, Ojeda-Chi R-VRI, Mancilla-Montelongo G, González-Pech PG, Torres-Acosta JFJ (2021) Frequency of sheep farms with anthelmintic resistant gastrointestinal nematodes in the Mexican Yucatán peninsula. Vet Parasitol: Reg Stud Rep 24:100549. https://doi.org/10.1016/j.vprsr.2021.100549
Sommerville RI (1957) The exsheathing mechanism of nematode infective larvae. Exp Parasitol 6:18–30. https://doi.org/10.1016/0014-4894(57)90004-8
Sprouffske K, Wagner A (2016) Growthcurver: an R package for obtaining interpretable metrics from microbial growth curves. BMC Bioinform 17:172. https://doi.org/10.1186/s12859-016-1016-7
Theodorou M, Williams B, Dhanoa M, McAllan A, France J (1994) A simple gas production using a pressure transducer to determine the fermentation kinetics of ruminant feeds. Anim Feed Sci Technol 48:185–197. https://doi.org/10.1016/0377-8401(94)90171-6
Vargas-Magaña JJ, Torres Acosta JFJ, Aguilar-Caballero AJ, Sandoval-Castro CA, Hoste H, Chan-Pérez JI (2014) Anthelmintic activity of acetone-water extracts against Haemonchus contortus eggs: interactions between tannins and other plant secondary compounds. Vet Parasitol 206:322–327. https://doi.org/10.1016/j.vetpar.2014.10.008
Ventura-Cordero J, González-Pech PG, Jaimez-Rodriguez PR, Ortíz-Ocampo GI, Sandoval-Castro CA, Torres-Acosta JFJ (2018) Feed resource selection of Criollo goats artificially infected with Haemonchus contortus: nutritional wisdom and prophylactic self-medication. Anim 12:1269–1276. https://doi.org/10.1017/S1751731117002634
Wang M, Tang SX, Tan ZL (2011) Modeling in vitro gas production kinetics: derivation of logistic–exponential (LE) equations and comparison of models. Anim Feed Sci Technol 165:137–150. https://doi.org/10.1016/j.anifeedsci.2010.09.016
Yang XS (2019) Data fitting and regression. In: Yang XS (ed) introduction to algorithms for data mining and machine learning. Academic Press, London, pp 67–90
Zajac AM, Garza J (2020) Biology, epidemiology, and control of gastrointestinal nematodes of small ruminants. Vet Clinic N Am: Food Anim Pract 36:73–87. https://doi.org/10.1016/j.cvfa.2019.12.005
Acknowledgements
We would like to thank the staff of the nutrition area and the Animal Nutrition Laboratory at FMVZ-UADY for all the support provided. We also thank the team in charge of the L3 larvae production at the FMVZ-UADY. C.G. Marin-Tun. received a doctoral scholarship from CONACYT, México.
Funding
This study was supported by the Consejo Nacional de Ciencia y Tecnología (CONACYT, México): Cátedras CONACYT Project No. 692, and doctoral grant No. 784280.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study’s conception and design. Material preparation and data collection were performed by Cindy G. Marin-Tun and Concepción M. Capetillo-Leal. Data analyses were performed by Cindy G. Marin-Tun and Carlos A. Sandoval Castro. The first draft of the manuscript was written by Cindy G. Marin-Tun, María G. Mancilla-Montelongo, and Juan F.J. Torres-Acosta. All the figures were prepared by Cindy G. Marin-Tun. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
The study was approved by the Bioethics Committee of the Faculty of Veterinary Medicine and Animal Science from the Autonomous University of Yucatán, México (CB-CCBA-D-2022-001).
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Handling Editor: Una Ryan
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary 1
Online resource 1 Haemonchus contortus L3 larvae: sheath (a), larva during the exsheathment process (b) and exsheathed larva (b).
Supplementary 2
Online resource 2 Complete equations for the nonlinear models considered to select the model that best described the in vitro exsheathment kinetics of Haemonchus contortus L3 using the in vitro rumen incubation technique with ruminal liquid.
Supplementary 3
Online resource 3 Log-logistic equations using the original exsheathment values and the data at 30 or 50% of the original data of in vitro exsheathment from H. contortus L3 larvae.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Marin-Tun, C.G., Mancilla-Montelongo, M.G., Torres-Acosta, J.F. et al. An in vitro rumen incubation method to study exsheathment kinetics of Haemonchus contortus third-stage infective larvae. Parasitol Res 122, 833–845 (2023). https://doi.org/10.1007/s00436-023-07780-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-023-07780-z