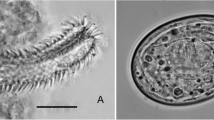
Abstract
The anomaly P is a mass morphological anomaly found in some populations of anuran amphibians (water frogs of the genus Pelophylax and toads of the genera Bufo and Bufotes) caused by the parasitic flatworm Strigea robusta. Minimum dose of cercariae for the appearance of the anomaly P remains unknown. However, it is important information for understanding of host population dynamics after invasion and the effects of the parasite on the second intermediate hosts. Herein, the invasion properties of S. robusta in Pelophylax lessonae tadpoles (Anura: Ranidae) and minimum dose for appearance of mild and severe forms of the anomaly P syndrome were described after direct experiments with certain numbers of cercariae exposure. Experimental groups of tadpoles have been exposed to eight doses of cercariae (2, 4, 6, 8, 10, 12, 14 and 16). A total of 63.8% tadpoles survived to the end of this experiment. It was revealed that a mild form of the anomaly P (polydactyly) can appear after infection by two cercariae, while the severe form traits appear after infection by four cercariae. The mean number of detected encysted metacercariae was reached to 53.5%. Differences in infection rates can be explained by the presence of an individual immune response in tadpoles or by the presence of different genetic lineages of the parasite infecting the same snail, which have different infectious potential. Low doses of infection leading to the induction of anomalies characterize S. robusta as a highly pathogenic species for amphibian species that are susceptible to infection and show an abnormal phenotype.


Similar content being viewed by others
Data availability
All data used are available upon request from the corresponding author.
References
Bellakhal M, Neveu A, Fartouna-Bellakhal M, Missaoui H, Aleya L (2014) Effects of temperature, density and food quality on larval growth and metamorphosis in the north African green frog Pelophylax saharicus. J Therm Biol. 45:81–86. https://doi.org/10.1016/j.jtherbio.2014.08.006
Cichy A, Faltýnková A, Żbikowska E (2011) Cercariae (Trematoda, Digenea) in European freshwater snails - a checklist of records from over one hundred years. Folia Malacol 19:165–189. https://doi.org/10.2478/v10125-011-0023-6
Combes C (1980) Atlas mondial des cercaires. Paris: Mémoires du Muséum National d'Histoire Naturelle. Série A, Zoologie. 115:1–236
Dedukh D, Litvinchuk S, Rosanov J, Shabanov D, Krasikova A (2017) Mutual maintenance of di- and triploid Pelophylax esculentus hybrids in R-E systems: results from artificial crossings experiments. BMC Evol Biol 17(1):220. https://doi.org/10.1186/s12862-017-1063-3
Dubois A (1979) Anomalies and mutations in natural populations of the Rana “esculenta” complex (Amphibia, Anura). Mitteilungen aus dem zoologischen Museum in Berlin 55:59–87
Dubois A (2017) Rostand’s anomaly P in Palaearctic green frogs (Pelophylax) and similar anomalies in amphibians. Mertensiella 25:49–56
Faltýnková A, Niewiadomska K, Santos M, Valtonen E (2007) Furcocercous cercariae (Trematoda) from freshwater snails in Central Finland. Acta Parasit 52:310–317. https://doi.org/10.2478/s11686-007-0050-z
Faltýnková A, Našincová V, Kablásková L (2008) Larval trematodes (Digenea) of planorbid snails (Gastropoda: Pulmonata) in Central Europe: a survey of species and key to their identification. Syst Parasitol 69:155–178. https://doi.org/10.1007/s11230-007-9127-1
Gosner KL (1960) A simplifed table for staging anuran embryos and larvae with notes on identifcation. Herpetologica 16:183–190. https://doi.org/10.2307/3890061
Hamann MI, Fernández MV, González CE (2019) Metacercariae of Strigeidae parasitizing amphibians of the Chaco Region in Argentina. An Acad Bras Ciênc 91:e20180044. https://doi.org/10.1590/0001-3765201920180044
Heneberg P, Sitko J, Těšínský M, Rząd I, Bizos J (2018) Central European Strigeidae Railliet, 1919 (Trematoda: Strigeidida): molecular and comparative morphological analysis suggests the reclassification of Parastrigea robusta Szidat, 1928 into Strigea Abildgaard, 1790. Parasitol Int 67:688–701. https://doi.org/10.1016/j.parint.2018.07.003
Henle K, Dubois A, Vershinin VL (2017a) A review of anomalies in natural populations of amphibians and their potential causes. Mertensiella 25:57–164
Henle K, Dubois A, Vershinin V (2017b) Commented glossary, terminology and synonymies of anomalies in natural populations of amphibians. Mertensiella 25:9–48
Johnson PTJ, Lunde KB, Ritchie EG, Launer EU (1999) The effect of trematode infection on amphibian limb development and survivorship. Science 284:802–804. https://doi.org/10.1126/science.284.5415.802
Johnson PTJ, Kellermanns E, Bowerman J (2011) Critical windows of disease risk: amphibian pathology driven by developmental changes in host resistance and tolerance. Funct Ecol 25:726–734
Kuzmin Y, Dmytrieva I, Marushchak O, Morozov-Leonov S, Oskyrko O, Nekrasova O (2020) Helminth species and infracommunities in frogs Pelophylax ridibundus and P. esculentus (Amphibia: Ranidae) in Northern Ukraine. Acta Parasitol 65:341–353. https://doi.org/10.2478/s11686-019-00164-3
Lunaschi L, Drago F (2012) Digenean parasites of Cariama cristata (Aves, Gruiformes) from Formosa Province, Argentina, with the description of a new species of the genus Strigea. Acta Parasitol 57:26–33. https://doi.org/10.2478/s11686-012-0004-y
Lunaschi LI, Drago FB (2013) Digenean parasites of the great antshrike, Taraba major (Aves: Thamnophilidae), from Argentina, with a description of a new species of the genus Strigea (Strigeidae). Folia Parasit 60:331–338. https://doi.org/10.14411/fp.2013.034
Matushkina KA, Kidov AA, Seryakova AA (2017) Captive breeding of larvae of narrowly distributedtriploid toads, Bufotes baturae (Stöck, Schmid, SteinleinetGrosse, 1999) with the use of complete feeds for aquariumfish. Vestn. Tambov. Gos. Univ. Estestv. Tekhn. Nauki 22(5 – 1):960–964
Nekrasova OD (2008) Classification of amphibian anomalies. Proceedings of the Ukrainian Herpetological Society, Kyiv 1:55–58
Ouellet M (2000) Amphibian deformities: current state of knowledge. In: Ecotoxicology of Amphibians and Reptiles: Society of Environmental Toxicology and Chemistry (SETAC), pp 617–661
Patrelle C, Portier J, Jouet D, Delorme D, Ferté H (2015) Prevalence and intensity of Alaria alata (Goeze, 1792) in water frogs and brown frogs in natural conditions. Parasitol Res 114:4405–4412. https://doi.org/10.1007/s00436-015-4680-z
Rostand J (1971) Les étangs à monstres. In: Histoire d’une recherche (1947-1970). Stock, Paris
Sinsch U, Kaschek J, Wiebe J (2018) Heavy metacercariae infestation (Parastrigea robusta) promotes the decline of a smooth newt population (Lissotriton vulgaris). Salamandra 54:210–221
Sinsch U, Heneberg P, Těšínský M, Balczun C, Scheid P (2019) Helminth endoparasites of the smooth newt Lissotriton vulgaris: linking morphological identification and molecular data. J Helminthol 93:332–341. https://doi.org/10.1017/S0022149X18000184
Sitko J, Heneberg P (2021) Long-term dynamics of trematode infections in common birds that use farmlands as their feeding habitats. Parasites Vectors 14:383. https://doi.org/10.1186/s13071-021-04876-2
Sudarikov VE (1984) Trematode fauna of the USSR. Strigeidae. Nauka, Moscow
Svinin AO, Bashinskiy IV, Litvinchuk SN, Neymark LA, Osipov VV, Katsman EA, Ermakov OA, Ivanov AY, Vedernikov AA, Drobot GP, Dubois A (2019) First record of the Jean Rostand’s “anomaly P” in the marsh frog, Pelophylax ridibundus, in central Russia. Alytes 37:31–45
Svinin AO, Bashinskiy IV, Litvinchuk SN, Ermakov OA, Ivanov AY, Neymark LA, Vedernikov AA, Osipov VV, Drobot GP, Dubois A (2020) Strigea robusta causes polydactyly and severe forms of Rostand’s anomaly P in water frogs. Parasites Vectors 13:381. https://doi.org/10.1186/s13071-020-04256-2
Vojtek J (1972) Observations on the life cycle of Parastrigea robusta Szidat, 1928 (Trematoda: Strigeidae) in Czechoslovakia. Folia Parasitol (Praha) 19:210
Zhigileva ON, Kirina IY (2015) Helminth infestation of the moor frog (Rana arvalis Nilsson, 1842) and the Siberian tree frog (Rana amurensis Boulenger, 1886) in Western Siberia. Contemp Probl Ecol 8:232–236. https://doi.org/10.1134/S1995425515020171
Acknowledgements
We are thankful to anonymous reviewer for the valuable comments and suggestions for improving the manuscript. We are thankful to L.A. Neymark and V.V. Osipov for help in field.
Funding
The research was supported by the Russian Science Foundation grant No. 21-74-00079, https://rscf.ru/en/project/21-74-00079/
Author information
Authors and Affiliations
Contributions
A.S., O.E. and S.L. designed the study; A S. provided data curation and made an original draft preparation; O.E. and S.L. reviewed and edited the manuscript. All the authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Ethics approval
The Mari State University Ethics Committee (Yoshkar-Ola, Russia) (#2/22.10.2021) approved experimental design and procedures.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Section Editor: David Bruce Conn
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Svinin, A., Bashinskiy, I., Ermakov, O. et al. Effects of minimum Strigea robusta (Digenea: Strigeidae) cercariae doses and localization of cysts on the anomaly P manifestation in Pelophylax lessonae (Anura: Ranidae) tadpoles. Parasitol Res 122, 889–894 (2023). https://doi.org/10.1007/s00436-022-07778-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-022-07778-z