Abstract
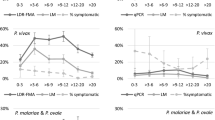
Agroecosystems have been associated with risk of malaria. The aim of this study was to determine the relationship between three agroecosystems: (i) rubber plantation (RP); (ii) oil palm plantation (OPP); (iii) no cash crop plantation (NCCP) and the prevalence of Plasmodium falciparum infection among children living in the Aboisso region. In the three villages within (Ehania-V5) or close (N’zikro) or far from (Ayébo) to each agroecosystem (RP, OPP, and NCCP), two cross-sectional parasitological surveys were carried out during the dry and the peak of the long wet seasons. A total of 586 children aged 1–14 years were recruited in the three villages to determine the prevalence of malaria using conventional microscopy. Plasmodium falciparum was the dominant species with an overall infection prevalence of 40.8%. There was a significant difference in prevalence between agroecosystems, during both the dry (p = 0.002) and wet seasons (p < 0.001), which was higher in agricultural settings compared with the NCCP environment, whatever the season. The prevalence of P. falciparum infection increased from the dry to the wet season in agricultural settings (RP and OPP), whereas no difference was noted for NCCP. Less than 18% of children use insecticide-treated nets (ITNs) in the three villages, ranging from 6 (in RP) to 30% (in OPP). Multivariate analysis indicated that age (1–4; 5–9; and 10–14 years) was not associated with malaria risk, but the season and living in agricultural villages were associated with a greater risk of malaria infection. Risk of malaria exposure was fourfold higher in children from agricultural villages than their counterpart from the non-agricultural area. Our findings highlight significant variations in the prevalence of P. falciparum according to agroecosystem and season. The findings will be useful in designing and implementing malaria control interventions by the National Malaria Control Program.

Similar content being viewed by others
References
Adja AM, N’goran EK, Koudou BG et al (2011) Contribution of Anopheles funestus, An. gambiae and An. nili (Diptera: Culicidae) to the perennial malaria transmission in the southern and western forest areas of Côte d’Ivoire. Ann Trop Med Parasitol 105:13–24. https://doi.org/10.1179/136485910X12851868780388
Adja AM, N’Goran KE, Kengne P et al (2006) Vectorial transmission of malaria in shrubby Savannah area at Ganse, Ivory Coast. Med Trop Rev Corps Sante Colon 66:449–455
Afrane Y, Klinkenberg E, Drechsel P et al (2004) Does irrigated urban agriculture influence the transmission of malaria in the city of Kumasi, Ghana? Acta Trop 89:125–134. https://doi.org/10.1016/j.actatropica.2003.06.001
Afrane YA, Lawson BW, Brenya R et al (2012) The ecology of mosquitoes in an irrigated vegetable farm in Kumasi, Ghana: abundance, productivity and survivorship. Parasit Vectors 5:233. https://doi.org/10.1186/1756-3305-5-233
Antonio-Nkondjio C, Atangana J, Ndo C et al (2008) Malaria transmission and rice cultivation in Lagdo, northern Cameroon. Trans R Soc Trop Med Hyg 102:352–359. https://doi.org/10.1016/j.trstmh.2007.12.010
Antonio-Nkondjio C, Demanou M, Etang J, Bouchite B (2013) Impact of cyfluthrin (Solfac EW050) impregnated bed nets on malaria transmission in the city of Mbandjock : lessons for the nationwide distribution of long-lasting insecticidal nets (LLINs) in Cameroon. Parasit Vectors 6:10
Assako Assako RJ, Bley D, Simard F (2005) Apports des sciences sociales et de l’entomologie dans l’analyse de l’endémicité du paludisme à HEVECAM, une agro-industrie du Sud-Cameroun. https://doi.org/10.1186/1756-3305-6-10
Assi S-B, Henry M-C, Rogier C et al (2013) Inland valley rice production systems and malaria infection and disease in the forest region of western Côte d’Ivoire. Malar J 12:233. https://doi.org/10.1186/1475-2875-12-233
Bassa FK, Ouattara M, Silué KD et al (2016) Epidemiology of malaria in the Taabo health and demographic surveillance system, south-central Côte d’Ivoire. Malar J 15:9. https://doi.org/10.1186/s12936-015-1076-6
Betsi AN, Tchicaya ES, Koudou BG (2012) [High proliferation of An. gambiae and An. funestus larvae in irrigated and non-irrigated rice fields in the Western forest region of Côte-d’Ivoire]. Bull Soc Pathol Exot 105:220–229. https://doi.org/10.1007/s13149-012-0219-z
Betsi N, Koua H, FouaBi K (2003) “Anopheles funestus” (Giles, 1900) la riziculture et le paludisme dans la région forestière ouest de la Côte d’Ivoire. Cah Agric 12(1):341–346
Bigoga JD, Manga L, Titanji VPK et al (2007) Malaria vectors and transmission dynamics in coastal south-western Cameroon. Malar J 6:5. https://doi.org/10.1186/1475-2875-6-5
Bigoga JD, Nanfack FM, Awono-Ambene PH et al (2012) Seasonal prevalence of malaria vectors and entomological inoculation rates in the rubber cultivated area of Niete South Region of Cameroon. Parasit Vectors 5:197. https://doi.org/10.1186/1756-3305-5-197
Chang MS, Hii J, Buttner P, Mansoor F (1997) Changes in abundance and behaviour of vector mosquitoes induced by land use during the development of an oil palm plantation in Sarawak. Trans R Soc Trop Med Hyg 91:382–386. https://doi.org/10.1016/s0035-9203(97)90248-0
Doannio JMC, Dossou-Yovo J, Diarrassouba S et al (2002) [Dynamics of malaria transmission in Kafiné, a rice growing village in a humid savannah area of Côte d’Ivoire]. Bull Soc Pathol Exot 95:11–16
Doannio JM, Konan YL, Amalaman K, Attiah J (2004) Connaissances, attitudes et pratiques des populations visà-vis des moustiques dans la zone urbaine et périurbaine de Bouaké et dans les villages de Kafiné et de Kabolo (Côte d’Ivoire - Afrique de l’Ouest). Bulletin de la Société de pathologie exotique. 97(4):295–301
Henry M-C, Rogier C, Nzeyimana I et al (2003) Inland valley rice production systems and malaria infection and disease in the savannah of Côte d’Ivoire. Trop Med Int Health TM IH 8:449–458. https://doi.org/10.1046/j.1365-3156.2003.01053.x
Houngbedji CA, N’Dri PB, Hürlimann E, et al (2015) Disparities of Plasmodium falciparum infection, malaria-related morbidity and access to malaria prevention and treatment among school-aged children: a national cross-sectional survey in Côte d’Ivoire. Malar J 14. https://doi.org/10.1186/1475-2875-14-7
Koudou BG, Adja AM, Matthys B et al (2007) [Agricultural activities and malaria transmission in two eco-epidemiological settings in central Côte d’Ivoire]. Bull Soc Pathol Exot 100:124–126
Koudou BG, Tano Y, Doumbia M et al (2005) Malaria transmission dynamics in central Côte d’Ivoire: the influence of changing patterns of irrigated rice agriculture. Med Vet Entomol 19:27–37. https://doi.org/10.1111/j.0269-283X.2005.00530.x
Koudou BG, Tano Y, Keiser J et al (2009) Effect of agricultural activities on prevalence rates, and clinical and presumptive malaria episodes in central Côte d’Ivoire. Acta Trop 111:268–274. https://doi.org/10.1016/j.actatropica.2009.05.006
Lengeler C (2004) Insecticide-treated bed nets and curtains for preventing malaria. Cochrane Database Syst Rev CD000363. https://doi.org/10.1002/14651858.CD000363.pub2
Matthys B, Vounatsou P, Raso G et al (2006) Urban farming and malaria risk factors in a medium-sized town in Cote d’Ivoire. Am J Trop Med Hyg 75:1223–1231
Mmbando BP, Vestergaard LS, Kitua AY et al (2010) A progressive declining in the burden of malaria in north-eastern Tanzania. Malar J 9:216. https://doi.org/10.1186/1475-2875-9-216
Mueller I, Taime J, Ibam E et al (2002) Complex patterns of malaria epidemiology in the highlands region of Papua New Guinea. P N G Med J 45:200–205
Mutero C.M, Kabutha C, Kimani V, et al. (2004) A transdisciplinary perspective on the links between malaria and agroecosystems in Kenya. Acta Tropica 89:171–186. https://doi.org/10.1016/j.actatropica.2003.07.003.
Nzeyimana I, Henry M, Dossou-Yovo J et al (2002) [The epidemiology of malaria in the southwestern forests of the Ivory Coast (Tai region)]. Bull Société Pathol Exot 95:89–94
Pluess B, Mueller I, Levi D et al (2009) Malaria–a major health problem within an oil palm plantation around Popondetta Papua New Guinea. Malar J 8:56. https://doi.org/10.1186/1475-2875-8-56
Sadia-Kacou CAM, Yobo CM, Adja MA et al (2019) Use of Anopheles salivary biomarker to assess seasonal variation of human exposure to Anopheles bites in children living near rubber and oil palm cultivations in Côte d’Ivoire. Parasite Epidemiol Control 5. https://doi.org/10.1016/j.parepi.2019.e00102
Tanga MC, Ngundu WI, Tchouassi PD (2011) Daily survival and human blood index of major malaria vectors associated with oil palm cultivation in Cameroon and their role in malaria transmission. Trop Med Int Health 16:447–457. https://doi.org/10.1111/j.1365-3156.2011.02726.x
Yadouléton A, N’Guessan R, Allagbé H et al (2010) The impact of the expansion of urban vegetable farming on malaria transmission in major cities of Benin. Parasit Vectors 3:118. https://doi.org/10.1186/1756-3305-3-118
Acknowledgements
We thank the study participants for their consent to participate in the study. We are grateful to PALMCI health, local authorities, and particularly the children and their parents/guardians. We are grateful to local assistants and to Issa Coulibaly from Institut Pierre Richet for his technical assistance. This study was integrated with malaria surveys and has the support of the National Malaria Control Program, which has provided anti-malarial and facilitated collaboration with the study site health agents. This study was also supported by the Aboisso Departmental and Regional Health Director.
We thank Isabella Athanassiou (Bonn, Germany) for language editing.
Funding
The study received part support from the National Malaria Control Programme (NMCP) of Côte d’Ivoire. The NMCP is currently implementing a lot of interventions against malaria. The parasitology surveys implemented in this study were supported and approved by the NMCP. The data generated were integrated into the routine monitoring and evaluation survey activities of NMCP.
Author information
Authors and Affiliations
Contributions
Cécile Agnimou Malanfoua Sadia-Kacou, Maurice Akré Adja, Benjamin Guibehi Koudou, and Yao Tano conceived and designed the study. Cécile Agnimou Malanfoua Sadia-Kacou collected data, performed all laboratory activities, and drafted the manuscript. Benjamin Guibehi Koudou and Maurice Akré Adja coordinated the field activities and read the manuscript. Anne poinsignon, Jean T. Coulibaly, Allassane F. Ouattara, Serge- Brice Assi and Franck Remoé interpreted the data and revised the manuscript. Tano Yao supervised the study. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics statement
This proposal was reviewed and approved by the Scientific Committee of Institut Pierre-Richet (IPR) and PNLP (Côte d’Ivoire) prior to implementation. The health district and village authorities were informed about the objectives, procedures, and benefits of the study. Verbal approval from the different communities’ members and written consent of districts health officers were obtained before starting data collection. Sick children were treated when they showed any symptoms suspected to be caused by malaria.
Conflict of interest
The authors declare no competing interests.
Additional information
Section Editor: Tobili Sam-Yellowe
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sadia-Kacou, C.A.M., Adja, M.A., Assi, S.B. et al. Seasonal prevalence of Plasmodium falciparum infection and use of insecticide-treated nets among children in three agroecosystems in Aboisso, Côte d’Ivoire. Parasitol Res 120, 3663–3671 (2021). https://doi.org/10.1007/s00436-021-07326-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-021-07326-1