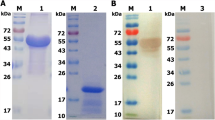
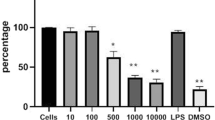
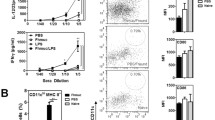
Abstract
Dendritic cells play a crucial role in inducing antigen-specific immunity to pathogens. During host-parasite interaction, host immune response to the parasite molecules is considered essential for recognizing novel antigens for control strategies. Therefore, in the present study, chicken dendritic cells (DCs) (ChDCs), derived from spleens were used to evaluate their capacity to proliferate and differentiate autologous T lymphocytes in response to actin-depolymerizing factor from Eimeria tenella (EtADF). Immunoblot analysis showed that recombinant EtADF protein (rEtADF) was able to interact with rat anti-rEtADF antibodies. The immunofluorescence test confirmed rEtADF binding on ChDCs surface. Flow cytometric analysis revealed that phenotypes for MHCII, CD1.1, CD11c, CD80, and CD86 were increased in ChDCs after rEtADF treatment. qRT-PCR results indicated that ChDCs triggered TLR signaling in response to rEtADF, and suppressed Wnt signaling. Transcript levels of CD83, CCL5, and CCR7 in ChDCs were improved following rEtADF treatment. In addition, rEtADF promoted DC-directed T cell proliferation and differentiation of naïve T cells into CD3+/CD4+ T cells in DC/T cell co-incubation system. Cytokine analysis of rEtADF-pulsed ChDCs showed increased levels of IL-12 and IFN-γ, while IL-10 and TGF-β remained unchanged. Moreover, rEtADF-treated ChDCs enhanced production of IFN-γ when incubated with T cells, and IL-4 secretion remained unchanged. Our findings indicted that rEtADF could facilitate the polarization of Th1 immune cells by triggering both host DCs and T cells. Our findings provide useful insights into future work aimed at anticoccidial vaccine strategies.









Similar content being viewed by others
Data availability
The data generated or analyzed during this study are included in this published article.
References
Banchereau J, Steinman RM (1998) Dendritic cells and the control of immunity. Nature 392:245–252. https://doi.org/10.1038/32588
Blake DP, Tomley FM (2014) Securing poultry production from the ever-present Eimeria challenge. Trends Parasitol 30:12–19. https://doi.org/10.1016/j.pt.2013.10.003
Boks MA, Kager-Groenland JR, Haasjes MS, Zwaginga JJ, van Ham SM, ten Brinke A (2012) IL-10-generated tolerogenic dendritic cells are optimal for functional regulatory T cell induction--a comparative study of human clinical-applicable DC. Clin Immunol 142:332–342. https://doi.org/10.1016/j.clim.2011.11.011
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Breed DG, Dorrestein J, Schetters TP, Waart LV, Rijke E, Vermeulen AN (1997) Peripheral blood lymphocytes from Eimeria tenella infected chickens produce gamma-interferon after stimulation in vitro. Parasite Immunol 19:127–135. https://doi.org/10.1046/j.1365-3024.1997.d01-191.x
Brisbin JT et al (2008) Gene expression profiling of chicken lymphoid cells after treatment with Lactobacillus acidophilus cellular components. Dev Comp Immunol 32:563–574. https://doi.org/10.1016/j.dci.2007.09.003
Clevers H, Nusse R (2012) Wnt/beta-catenin signaling and disease. Cell 149:1192–1205. https://doi.org/10.1016/j.cell.2012.05.012
Cristina Castañeda-Patlán M, Fuentes-García G, Robles-Flores M (2019) Wnt signaling as a master regulator of immune tolerance in a tumor microenvironment. In: Cell signalling - thermodynamics and molecular control. https://doi.org/10.5772/intechopen.81619
Dalloul RA, Lillehoj HS (2006) Poultry coccidiosis: recent advancements in control measures and vaccine development. Expert Rev Vaccines 5:143–163. https://doi.org/10.1586/14760584.5.1.143
Goronzy JJ, Weyand CM (2012) 44 - The innate and adaptive immune systems. In: Goldman L, Schafer AI (eds) Goldman’s Cecil medicine (twenty fourth edition). W.B. Saunders, Philadelphia, pp 214–222. https://doi.org/10.1016/B978-1-4377-1604-7.00044-0
Heufler C et al (1996) Interleukin-12 is produced by dendritic cells and mediates T helper 1 development as well as interferon-gamma production by T helper 1 cells. Eur J Immunol 26:659–668. https://doi.org/10.1002/eji.1830260323
Higgs R et al (2006) Induction of a novel chicken Toll-like receptor following Salmonella enterica serovar Typhimurium infection. Infect Immun 74:1692–1698. https://doi.org/10.1128/iai.74.3.1692-1698.2006
Jin P, Han TH, Ren J, Saunders S, Wang E, Marincola FM, Stroncek DF (2010) Molecular signatures of maturing dendritic cells: implications for testing the quality of dendritic cell therapies. J Transl Med 8:4. https://doi.org/10.1186/1479-5876-8-4
Kadykalo S, Roberts T, Thompson M, Wilson J, Lang M, Espeisse O (2018) The value of anticoccidials for sustainable global poultry production. Int J Antimicrob Agents 51:304–310. https://doi.org/10.1016/j.ijantimicag.2017.09.004
Kannaki TR, Reddy MR, Shanmugam M, Verma PC, Sharma RP (2010) Chicken toll-like receptors and their role in immunity. World’s Poult Sci J 66:727–738. https://doi.org/10.1017/s0043933910000693
Kapsenberg ML (2003) Dendritic-cell control of pathogen-driven T-cell polarization. Nat Rev Immunol 3:984–993. https://doi.org/10.1038/nri1246
Kawai T, Akira S (2007) TLR signaling. Semin Immunol 19:24–32. https://doi.org/10.1016/j.smim.2006.12.004
Kim WH, Chaudhari AA, Lillehoj HS (2019) Involvement of T cell immunity in avian coccidiosis. Front Immunol 10:2732–2732. https://doi.org/10.3389/fimmu.2019.02732
Kogut MH, Lange C (1989a) Interferon-gamma-mediated inhibition of the development of Eimeria tenella in cultured cells. J Parasitol 75:313–317
Kogut MH, Lange C (1989b) Recombinant interferon-gamma inhibits cell invasion by Eimeria tenella. J Interf Res 9:67–77. https://doi.org/10.1089/jir.1989.9.67
Kokkinopoulos D, Perez S, Sotiriadou R, Stinios J, Papamichail M (1992) The use of nylon wool for the isolation of T lymphocyte subpopulations. J Immunol Methods 154:1–6. https://doi.org/10.1016/0022-1759(92)90205-8
Laidlaw BJ, Craft JE, Kaech SM (2016) The multifaceted role of CD4(+) T cells in CD8(+) T cell memory. Nat Rev Immunol 16:102–111. https://doi.org/10.1038/nri.2015.10
Lakho SA et al (2020) Recombinant ubiquitin-conjugating enzyme of Eimeria maxima induces immunogenic maturation in chicken splenic-derived dendritic cells and drives Th1 polarization in-vitro. Microb Pathog:104162. https://doi.org/10.1016/j.micpath.2020.104162
Lal K et al (2009) Proteomic comparison of four Eimeria tenella life-cycle stages: unsporulated oocyst, sporulated oocyst, sporozoite and second-generation merozoite. Proteomics 9:4566–4576. https://doi.org/10.1002/pmic.200900305
Lambotin M, Raghuraman S, Stoll-Keller F, Baumert TF, Barth H (2010) A look behind closed doors: interaction of persistent viruses with dendritic cells. Nat Rev Microbiol 8:350–360. https://doi.org/10.1038/nrmicro2332
Liang J, Fu J, Kang H, Lin J, Yu Q, Yang Q (2013) The stimulatory effect of TLRs ligands on maturation of chicken bone marrow-derived dendritic cells. Vet Immunol Immunopathol 155:205–210. https://doi.org/10.1016/j.vetimm.2013.06.014
Liu L et al (2017) Identification of common immunodominant antigens of Eimeria tenella , Eimeria acervulina and Eimeria maxima by immunoproteomic analysis. Oncotarget 8(21)
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-ΔΔCt) method. Methods (San Diego, Calif) 25:402–408. https://doi.org/10.1006/meth.2001.1262
Lowenthal JW, York JJ, O’Neil TE, Rhodes S, Prowse SJ, Strom ADG, Digby MR (1997) In vivo effects of chicken interferon-γ during infection with Eimeria. J Interf Cytokine Res 17:551–558. https://doi.org/10.1089/jir.1997.17.551
Manicassamy S, Pulendran B (2011) Dendritic cell control of tolerogenic responses. Immunol Rev 241:206–227. https://doi.org/10.1111/j.1600-065X.2011.01015.x
Min W, Kim WH, Lillehoj EP, Lillehoj HS (2013) Recent progress in host immunity to avian coccidiosis: IL-17 family cytokines as sentinels of the intestinal mucosa. Dev Comp Immunol 41:418–428. https://doi.org/10.1016/j.dci.2013.04.003
O’Neill LAJ, Fitzgerald KA, Bowie AG (2003) The Toll–IL-1 receptor adaptor family grows to five members. Trends Immunol 24:286–289. https://doi.org/10.1016/S1471-4906(03)00115-7
Oderup C, LaJevic M, Butcher EC (2013) Canonical and noncanonical Wnt proteins program dendritic cell responses for tolerance. J Immunol 190:6126–6134. https://doi.org/10.4049/jimmunol.1203002
Peek HW, Landman WJ (2011) Coccidiosis in poultry: anticoccidial products, vaccines and other prevention strategies. Vet Q 31:143–161. https://doi.org/10.1080/01652176.2011.605247
Prowse SJ, Pallister J (1989) Interferon release as a measure of the T-cell response to coccidial antigens in chickens. Avian Pathol 18:619–630. https://doi.org/10.1080/03079458908418637
Pulendran B, Ahmed R (2006) Translating innate immunity into immunological memory: implications for vaccine development. Cell 124:849–863. https://doi.org/10.1016/j.cell.2006.02.019
Raker VK, Domogalla MP, Steinbrink K (2015) Tolerogenic dendritic cells for regulatory T cell induction in man. Front Immunol 6. https://doi.org/10.3389/fimmu.2015.00569
Rothwell L, Muir W, Kaiser P (2000) Interferon-gamma is expressed in both gut and spleen during Eimeria tenella infection. Avian Pathol 29:333–342. https://doi.org/10.1080/03079450050118467
Sallusto F, Lanzavecchia A (2002) The instructive role of dendritic cells on T-cell responses. Arthritis Res 4(Suppl 3):S127–S132. https://doi.org/10.1186/ar567
Shrestha A, Sadeyen J-R, Iqbal M (2018) Enhancing protective efficacy of poultry vaccines through targeted delivery of antigens to antigen-presenting cells. Vaccines 6. https://doi.org/10.3390/vaccines6040075
Song X, Huang X, Yan R, Xu L, Li X (2015) Efficacy of chimeric DNA vaccines encoding Eimeria tenella 5401 and chicken IFN-gamma or IL-2 against coccidiosis in chickens. Exp Parasitol 156:19–25. https://doi.org/10.1016/j.exppara.2015.05.003
St Paul M, Brisbin JT, Abdul-Careem MF, Sharif S (2013) Immunostimulatory properties of Toll-like receptor ligands in chickens. Vet Immunol Immunopathol 152:191–199. https://doi.org/10.1016/j.vetimm.2012.10.013
Tellez G, Shivaramaiah S, Barta J, Hernandez-Velasco X, Hargis B (2014) Coccidiosis: recent advancements in the immunobiology of Eimeria species, preventive measures, and the importance of vaccination as a control tool against these Apicomplexan parasites. Vet Med 23. https://doi.org/10.2147/vmrr.s57839
Thakur A, Mikkelsen H, Jungersen G (2019) Intracellular pathogens: host immunity and microbial persistence strategies. J Immunol Res 2019:1356540. https://doi.org/10.1155/2019/1356540
Trout JM, Lillehoj HS (1996) T lymphocyte roles during Eimeria acervulina and Eimeria tenella infections. Vet Immunol Immunopathol 53:163–172. https://doi.org/10.1016/0165-2427(95)05544-4
Wetzel DM, Hakansson S, Hu K, Roos D, Sibley LD (2003) Actin filament polymerization regulates gliding motility by apicomplexan parasites. Mol Biol Cell 14:396–406. https://doi.org/10.1091/mbc.e02-08-0458
Wu Z, Rothwell L, Young JR, Kaufman J, Butter C, Kaiser P (2010) Generation and characterization of chicken bone marrow-derived dendritic cells. Immunology 129:133–145. https://doi.org/10.1111/j.1365-2567.2009.03129.x
Wu Z, Hu T, Kaiser P (2011) Chicken CCR6 and CCR7 are markers for immature and mature dendritic cells respectively. Dev Comp Immunol 35:563–567. https://doi.org/10.1016/j.dci.2010.12.015
Xu JH, Qin ZH, Liao YS, Xie MQ, Li AX, Cai JP (2008) Characterization and expression of an actin-depolymerizing factor from Eimeria tenella. Parasitol Res 103:263–270. https://doi.org/10.1007/s00436-008-0961-0
Zhou Z et al (2013) Upregulation of chicken TLR4, TLR15 and MyD88 in heterophils and monocyte-derived macrophages stimulated with Eimeria tenella in vitro. Exp Parasitol 133:427–433. https://doi.org/10.1016/j.exppara.2013.01.002
Funding
This work is supported by the Joint Research Project between the National Natural Science Foundation China and Pakistan Science Foundation (NSFC-PSF) and Grant Number (Grant No. 31661143017).
Author information
Authors and Affiliations
Contributions
LXR directed the project and participated in the coordination and management of the study. SAL performed the laboratory experiments on DCs and wrote the manuscript. MH analyzed the data. JH, ZY, and ZY assisted in the laboratory experiments. MWH provided some ideas for the experimental design. MTA obtained chicken spleen samples, and MAM cultured and isolated dendritic cells. SXK, YRF, and XLX provided new analytical reagents and tools. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethics approval
Animals treated in this study were in compliance with the guidelines of the Animal Ethics Committee, Nanjing Agricultural University, China. All of the experimental protocols were granted approval from the Science and Technology Agency of Jiangsu Province. The approval ID is SYXK (SU) 2018–0005.
Additional information
Section Editor: Berit Bangoura
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lakho, S.A., Haseeb, M., Huang, J. et al. Actin-depolymerizing factor from Eimeria tenella promotes immunogenic function of chicken dendritic cells. Parasitol Res 120, 579–592 (2021). https://doi.org/10.1007/s00436-020-07016-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-020-07016-4