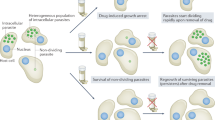
Abstract
Leishmaniasis is a neglected parasitic disease for which the current antileishmania therapeutics are hampered by drug toxicity, high cost, need for parenteral administration, increasing treatment failure rates, and emergence of drug resistance. The R&D pipeline had run fairly dry for several years, but fortunately some new drug candidates are now under (pre)clinical development. Identification of novel drugs will nevertheless remain essential to adequately sustain and improve effective disease control in the future. In this review, a package of standard and accessible R&D approaches is discussed with expansion to some alternative strategies focusing on parasite–host and vector–host interactions.
Similar content being viewed by others
References
Abdeladhim M, Kamhawi S, Valenzuela JG (2014) What’s behind a sand fly bite? The profound effect of sand fly saliva on host hemostasis, inflammation and immunity. Infect Genet Evol 28:691–703. https://doi.org/10.1016/j.meegid.2014.07.028
Alcantara LM, Ferreira TCS, Gadelha FR, Miguel DC (2018) Challenges in drug discovery targeting TriTryp diseases with an emphasis on leishmaniasis. Int J Parasitol Drugs Drug Resist 8(3):430–439. https://doi.org/10.1016/j.ijpddr.2018.09.006
Alrajhi AA, Ibrahim EA, De Vol EB, Khairat M, Faris RM, Maguire JH (2002) Fluconazole for the treatment of cutaneous leishmaniasis caused by Leishmania major. N Engl J Med 346(12):891–895. https://doi.org/10.1056/NEJMoa011882
Alvar J, Vélez ID, Bern C, Herrero M, Desjeux P, Cano J, Jannin J, Boer M, the WHO Leishmaniasis Control Team (2012) Leishmaniasis worldwide and global estimates of its incidence. PLoSOne 7(5):e35671. https://doi.org/10.1371/journal.pone.0035671
Alves F, Bilbe G, Blesson S, Goyal V, Monnerat S, Mowbray C, Muthoni Ouattara G, Pécoul B, Rijal S, Rode J, Solomos A, Strub-Wourgaft N, Wasunna M, Wells S, Zijlstra EE, Arana B, Alvar J (2018) Recent development of visceral Leishmaniasis treatments: successes, pitfalls, and perspectives. Clin Microbiol Rev 31(4). https://doi.org/10.1128/cmr.00048-18
Andrade BB, Teixeira CR (2012) Biomarkers for exposure to sand flies bites as tools to aid control of leishmaniasis. Front Immunol 3:121. https://doi.org/10.3389/fimmu.2012.00121
Arevalo I, Ward B, Miller R, Meng TC, Najar E, Alvarez E, Matlashewski G, Llanos-Cuentas A (2001) Successful treatment of drug-resistant cutaneous leishmaniasis in humans by use of imiquimod, an immunomodulator. Clin Infect Dis 33(11):1847–1851. https://doi.org/10.1086/324161
Avorn J (2015) The $2.6 billion pill--methodologic and policy considerations. N Engl J Med 372(20):1877–1879. https://doi.org/10.1056/NEJMp1500848
Bern C, Maguire JH, Alvar J (2008) Complexities of assessing the disease burden attributable to leishmaniasis. PLoSNeglTropDis 2(10):e313. https://doi.org/10.1371/journal.pntd.0000313
Brahmachari U (1922) Chemotherapy of antimonial compounds in kala-azar infection. Part I. Indian J Med Res 10(2):492–522
Breder CD, Du W, Tyndall A (2017) What’s the regulatory value of a target product profile? Trends Biotechnol 35(7):576–579. https://doi.org/10.1016/j.tibtech.2017.02.011
Brito G et al (2017) Oral Pentoxifylline associated with pentavalent antimony: a randomized trial for cutaneous Leishmaniasis. Am J Trop Med Hyg 96(5):1155–1159. https://doi.org/10.4269/ajtmh.16-0435
Burza S, Croft SL, Boelaert M (2018) Leishmaniasis. Lancet 392(10151):951–970. https://doi.org/10.1016/s0140-6736(18)31204-2
Cha Y, Erez T, Reynolds IJ, Kumar D, Ross J, Koytiger G, Kusko R, Zeskind B, Risso S, Kagan E, Papapetropoulos S, Grossman I, Laifenfeld D (2018) Drug repurposing from the perspective of pharmaceutical companies. Br J Pharmacol 175(2):168–180. https://doi.org/10.1111/bph.13798
Chakravarty J, Sundar S (2010) Drug resistance in leishmaniasis. J Global Infect Dis 2(2):167–176. https://doi.org/10.4103/0974-777X.62887
Charlton RL, Rossi-Bergmann B, Denny PW, Steel PG (2018) Repurposing as a strategy for the discovery of new anti-leishmanials: the-state-of-the-art. Parasitology 145(2):219–236. https://doi.org/10.1017/s0031182017000993
ClinicalTrials.gov (2015) US National Library of medicine, 1997, NCT01980199. Trial to Determine Efficacy of Fexinidazole in Visceral Leihmaniasis Patients in Sudan. Registered Nov 2013, https://clinicaltrials.gov/ct2/show/NCT01980199. Accessed 6/6/2019
Cohen JP, Silva L, Cohen A, Awatin J, Sturgeon R (2016) Progress report on neglected tropical disease drug donation programs. Clin Ther 38(5):1193–1204. https://doi.org/10.1016/j.clinthera.2016.02.031
Cojean S et al (2012) Leishmania resistance to miltefosine associated with genetic marker. Emerg Infect Dis 18(4):704–706. https://doi.org/10.3201/eid1804.110841
Croft SL (1986) In vitro screens in the experimental chemotherapy of leishmaniasis and trypanosomiasis. Parasitol Today (Personal ed) 2(3):64–69
Croft SL (2001) Monitoring drug resistance in leishmaniasis. Tropical Med Int Health 6(11):899–905
Croft SL, Engel J (2006) Miltefosine--discovery of the antileishmanial activity of phospholipid derivatives. Trans R Soc Trop Med Hyg 100 Suppl 1:S4–S8. https://doi.org/10.1016/j.trstmh.2006.03.009
Croft SL, Olliaro P (2011) Leishmaniasis chemotherapy—challenges and opportunities. Clin Microbiol Infect 17(10):1478–1483. https://doi.org/10.1111/j.1469-0691.2011.03630.x
Croft SL, Sundar S, Fairlamb AH (2006) Drug resistance in leishmaniasis. Clin Microbiol Rev 19(1):111–126. https://doi.org/10.1128/CMR.19.1.111-126.2006
Dalton JE, Kaye PM (2010) Immunomodulators: use in combined therapy against leishmaniasis. Expert Rev Anti-Infect Ther 8(7):739–742. https://doi.org/10.1586/eri.10.64
Davies CR, Kaye P, Croft SL, Sundar S (2003) Leishmaniasis: new approaches to disease control. Bmj 326(7385):377–382
De Rycker M et al (2013) Comparison of a high-throughput high-content intracellular Leishmania donovani assay with an axenic amastigote assay. Antimicrob Agents Chemother 57(7):2913–2922. https://doi.org/10.1128/aac.02398-12
de Souza W, Rodrigues JCF (2009) Sterol biosynthesis pathway as target for anti-trypanosomatid drugs. Interdiscip Perspect Infect Dis 2009:1–19. https://doi.org/10.1155/2009/642502
Dey R et al (2018) Gut microbes egested during bites of infected sand flies augment severity of Leishmaniasis via Inflammasome-derived IL-1beta. Cell Host Microbe 23(1):134–143.e6. https://doi.org/10.1016/j.chom.2017.12.002
DNDi (2016) Fexinidazole/miltefosine combination (VL). http://www.dndi.org/diseases-projects/portfolio/fexinidazole-vl/. Accessed 6/6/2019
DNDi (2019) https://www.dndi.org/about-dndi/business-model/lessons-learned/. Accessed 6/6/2019
Durieu E et al (2016a) From drug screening to target deconvolution: a target-based drug discovery pipeline using Leishmania casein kinase 1 isoform 2 to identify compounds with Antileishmanial activity. Antimicrob Agents Chemother 60(5):2822–2833. https://doi.org/10.1128/aac.00021-16
Durieu E, Prina E, Leclercq O, Oumata N, Gaboriaud-Kolar N, Vougogiannopoulou K, Aulner N, Defontaine A, No JH, Ruchaud S, Skaltsounis AL, Galons H, Späth GF, Meijer L, Rachidi N (2016b) From drug screening to target deconvolution: a target-based drug discovery pipeline using Leishmania casein kinase 1 isoform 2 to identify compounds with Antileishmanial activity. Antimicrob Agents Chemother 60(5):2822–2833. https://doi.org/10.1128/aac.00021-16
El-On J, Bazarsky E, Sneir R (2007) Leishmania major: in vitro and in vivo anti-leishmanial activity of paromomycin ointment (Leshcutan) combined with the immunomodulator Imiquimod. Exp Parasitol 116(2):156–162. https://doi.org/10.1016/j.exppara.2006.12.004
Ferreira LLG, Andricopulo AD (2018) Chemoinformatics strategies for Leishmaniasis drug discovery. Front Pharmacol 9:1278–1278. https://doi.org/10.3389/fphar.2018.01278
Fraihi W et al (2017) An integrated overview of the midgut bacterial flora composition of Phlebotomus perniciosus, a vector of zoonotic visceral leishmaniasis in the Western Mediterranean Basin. PLoS Negl Trop Dis 11(3):e0005484. https://doi.org/10.1371/journal.pntd.0005484
Freitas-Junior LH, Chatelain E, Kim HA, Siqueira-Neto JL (2012) Visceral leishmaniasis treatment: what do we have, what do we need and how to deliver it? Int J Parasitol Drugs Drug Resist 2:11–19. https://doi.org/10.1016/j.ijpddr.2012.01.003
Frezard F, Demicheli C, Ribeiro RR (2009) Pentavalent antimonials: new perspectives for old drugs. Molecules 14(7):2317–2336. https://doi.org/10.3390/molecules14072317
Galán-Puchades MT (2017) WHO delays Guinea-worm disease eradication to 2020: are dogs the sole culprits? Lancet Infect Dis 17(11):1124–1125. https://doi.org/10.1016/S1473-3099(17)30565-0
Gomes R, Oliveira F (2012) The immune response to sand fly salivary proteins and its influence on leishmania immunity. Front Immunol 3:110. https://doi.org/10.3389/fimmu.2012.00110
Goncalves D, Hunziker P (2016) Transmission-blocking strategies: the roadmap from laboratory bench to the community. Malar J 15:95. https://doi.org/10.1186/s12936-016-1163-3
Hendrickx S et al (2014) Experimental selection of paromomycin and miltefosine resistance in intracellular amastigotes of Leishmania donovani and L. infantum. Parasitol Res 113(5):1875–1881. https://doi.org/10.1007/s00436-014-3835-7
Hendrickx S, Beyers J, Mondelaers A, Eberhardt E, Lachaud L, Delputte P, Cos P, Maes L (2016a) Evidence of a drug-specific impact of experimentally selected paromomycin and miltefosine resistance on parasite fitness in Leishmania infantum. J Antimicrob Chemother 71:1914–1921. https://doi.org/10.1093/jac/dkw096
Hendrickx S, Guerin PJ, Caljon G, Croft SL, Maes L (2016b) Evaluating drug resistance in visceral leishmaniasis: the challenges. Parasitology 1-11 doi:https://doi.org/10.1017/s0031182016002031
Hendrickx S, Maes L, Croft SL, Caljon G (2018) The challenges of effective leishmaniasis treatment. In: Sucre AP (ed) Drug Resistance in Leishmania Parasites. Springer: 193–206
Herrera Acevedo C, Scotti L, Feitosa Alves M, Formiga Melo Diniz MF, Scotti MT (2017) Computer-Aided Drug Design Using Sesquiterpene Lactones as Sources of New Structures with Potential Activity against Infectious Neglected Diseases. Molecules (Basel, Switzerland) 22(1) doi:https://doi.org/10.3390/molecules22010079
Hillesland H et al (2008) Identification of aerobic gut bacteria from the kala azar vector, Phlebotomus argentipes: a platform for potential paratransgenic manipulation of sand flies. Am J Trop Med Hyg 79(6):881–886
Hurwitz I, Fieck A, Read A, Hillesland H, Klein N, Kang A, Durvasula R (2011a) Paratransgenic control of vector borne diseases. Int J Biol Sci 7(9):1334–1344
Hurwitz I, Hillesland H, Fieck A, Das P, Durvasula R (2011b) The paratransgenic sand fly: a platform for control of Leishmania transmission. Parasit Vectors 4:82. https://doi.org/10.1186/1756-3305-4-82
Johnston KL, Ford L, Taylor MJ (2014) Overcoming the challenges of drug discovery for neglected tropical diseases: the A.WOL experience. J Biomol Screen 19(3):335–343. https://doi.org/10.1177/1087057113511270
Jones NG, Catta-Preta CMC, Lima A, Mottram JC (2018a) Genetically validated drug targets in Leishmania: current knowledge and future prospects. ACS Infect Dis 4(4):467–477. https://doi.org/10.1021/acsinfecdis.7b00244
Jones NG, Catta-Preta CMC, Lima APCA, Mottram JC (2018b) Genetically validated drug targets in Leishmania: current knowledge and future prospects. ACS Infect Dis 4(4):467–477. https://doi.org/10.1021/acsinfecdis.7b00244
Kelly PH, et al. (2017) The gut microbiome of the vector <em>Lutzomyia longipalpis</em> is essential for survival of <em>Leishmania infantum</em>. mBio 8(1) doi:https://doi.org/10.1128/mBio.01121-16
Khalili G, Dobakhti F, Mahmoudzadeh-Niknam H, Khaze V, Partovi F (2011) Immunotherapy with Imiquimod increases the efficacy of Glucantime therapy of Leishmania major infection. Iran J Immunol 8(1):45–51
Khare S, Nagle AS, Biggart A, Lai YH, Liang F, Davis LC, Barnes SW, Mathison CJN, Myburgh E, Gao MY, Gillespie JR, Liu X, Tan JL, Stinson M, Rivera IC, Ballard J, Yeh V, Groessl T, Federe G, Koh HXY, Venable JD, Bursulaya B, Shapiro M, Mishra PK, Spraggon G, Brock A, Mottram JC, Buckner FS, Rao SPS, Wen BG, Walker JR, Tuntland T, Molteni V, Glynne RJ, Supek F (2016) Proteasome inhibition for treatment of leishmaniasis, Chagas disease and sleeping sickness. Nature 537(7619):229–233. https://doi.org/10.1038/nature19339
Khraiwesh M, Leed S, Roncal N, Johnson J, Sciotti R, Smith P, Read L, Paris R, Hudson T, Hickman M, Grogl M (2016) Antileishmanial activity of compounds derived from the Medicines for Malaria Venture open access box against intracellular Leishmania major amastigotes. Am J Trop Med Hyg 94(2):340–347. https://doi.org/10.4269/ajtmh.15-0448
Klinkert MQ, Heussler V (2006) The use of anticancer drugs in antiparasitic chemotherapy. Mini-Rev Med Chem 6(2):131–143
Knockaert M, Gray N, Damiens E, Chang YT, Grellier P, Grant K, Fergusson D, Mottram J, Soete M, Dubremetz JF, le Roch K, Doerig C, Schultz PG, Meijer L (2000) Intracellular targets of cyclin-dependent kinase inhibitors: identification by affinity chromatography using immobilised inhibitors. Chem Biol 7(6):411–422
Kulkarni MM, Jones EA, McMaster WR, McGwire BS (2008) Fibronectin binding and proteolytic degradation by Leishmania and effects on macrophage activation. Infect Immun 76(4):1738–1747. https://doi.org/10.1128/iai.01274-07
Kulshrestha A et al (2013) Validation of a simple resazurin-based promastigote assay for the routine monitoring of miltefosine susceptibility in clinical isolates of Leishmania donovani. Parasitol Res 112(2):825–828. https://doi.org/10.1007/s00436-012-3212-3
Kumar GA, Roy S, Jafurulla M, Mandal C, Chattopadhyay A (2016) Statin-induced chronic cholesterol depletion inhibits Leishmania donovani infection: relevance of optimum host membrane cholesterol. Biochim Biophys Acta 1858(9):2088–2096. https://doi.org/10.1016/j.bbamem.2016.06.010
Kumar A, Pandey SC, Samant M (2018) Slow pace of antileishmanial drug development. Parasitol Open 4:e4. https://doi.org/10.1017/pao.2018.1
Lamotte S, Spath GF, Rachidi N, Prina E (2017) The enemy within: targeting host-parasite interaction for antileishmanial drug discovery. PLoS Negl Trop Dis 11(6):e0005480. https://doi.org/10.1371/journal.pntd.0005480
Langhorne J, Duffy PE (2016) Expanding the antimalarial toolkit: targeting host-parasite interactions. J Exp Med 213(2):143–153. https://doi.org/10.1084/jem.20151677
Lestinova T, Rohousova I, Sima M, de Oliveira CI, Volf P (2017) Insights into the sand fly saliva: blood-feeding and immune interactions between sand flies, hosts, and Leishmania. PLoS Negl Trop Dis 11(7):e0005600. https://doi.org/10.1371/journal.pntd.0005600
Louradour I, Monteiro CC, Inbar E, Ghosh K, Merkhofer R, Lawyer P, Paun A, Smelkinson M, Secundino N, Lewis M, Erram D, Zurek L, Sacks D (2017) The midgut microbiota plays an essential role in sand fly vector competence for Leishmania major. Cell Microbiol 19(10):e12755. https://doi.org/10.1111/cmi.12755
Marr AK, MacIsaac JL, Jiang R, Airo AM, Kobor MS, McMaster WR (2014) Leishmania donovani infection causes distinct epigenetic DNA methylation changes in host macrophages. PLoS Pathog 10(10):e1004419. https://doi.org/10.1371/journal.ppat.1004419
Martin-Martin I, Chagas AC, Guimaraes-Costa AB, Amo L, Oliveira F, Moore IN, DeSouza-Vieira TS, Sanchez EE, Suntravat M, Valenzuela JG, Ribeiro JMC, Calvo E (2018) Immunity to LuloHya and Lundep, the salivary spreading factors from Lutzomyia longipalpis, protects against Leishmania major infection. PLoS Pathog 14(5):e1007006. https://doi.org/10.1371/journal.ppat.1007006
Maurya R, Bhattacharya P, Ismail N, Dagur PK, Joshi AB, Razdan K, McCoy JP, Ascher J, Dey R, Nakhasi HL (2016) Differential role of leptin as an Immunomodulator in controlling visceral Leishmaniasis in Normal and leptin-deficient mice. Am J Trop Med Hyg 95(1):109–119. https://doi.org/10.4269/ajtmh.15-0804
McDowell MA (2015) Vector-transmitted disease vaccines: targeting salivary proteins in transmission (SPIT). Trends Parasitol 31(8):363–372. https://doi.org/10.1016/j.pt.2015.04.011
McGwire BS, Satoskar AR (2014) Leishmaniasis: clinical syndromes and treatment. QJM 107(1):7–14. https://doi.org/10.1093/qjmed/hct116
Mesa CVB, Gustavo A, Muñoz DL, Muskus CE, Flórez AF, Ochoa R, Vélez ID, Robledo SM (2015) In silico screening of potential drug with Antileishmanial Activty and validation of their activity by in vitro and in vivo studies. J Chem Chem Eng 9:375–402
Murray HW (2010) Treatment of visceral leishmaniasis in 2010: direction from Bihar state, India. Future Microbiol 5(9):1301–1303. https://doi.org/10.2217/fmb.10.92
Nagle AS, Khare S, Kumar AB, Supek F, Buchynskyy A, Mathison CJN, Chennamaneni NK, Pendem N, Buckner FS, Gelb MH, Molteni V (2014) Recent developments in drug discovery for leishmaniasis and human African trypanosomiasis. Chem Rev 114(22):11305–11347. https://doi.org/10.1021/cr500365f
Nwaka S, Hudson A (2006) Innovative lead discovery strategies for tropical diseases. Nat Rev Drug Discov 5(11):941–955. https://doi.org/10.1038/nrd2144
Ouakad M et al (2011) Increased metacyclogenesis of antimony-resistant Leishmania donovani clinical lines. Parasitology 138(11):1392–1399. https://doi.org/10.1017/S0031182011001120
Pacis A et al (2015) Bacterial infection remodels the DNA methylation landscape of human dendritic cells. Genome Res 25(12):1801–1811. https://doi.org/10.1101/gr.192005.115
Parihar SP, Hartley MA, Hurdayal R, Guler R, Brombacher F (2016) Topical simvastatin as host-directed therapy against severity of cutaneous Leishmaniasis in mice. Sci Rep 6:33458. https://doi.org/10.1038/srep33458
Patterson S, Wyllie S (2014) Nitro drugs for the treatment of trypanosomatid diseases: past, present, and future prospects. Trends Parasitol 30(6):289–298. https://doi.org/10.1016/j.pt.2014.04.003
Patterson S, Wyllie S, Stojanovski L, Perry MR, Simeons FRC, Norval S, Osuna-Cabello M, de Rycker M, Read KD, Fairlamb AH (2013) The R enantiomer of the Antitubercular drug PA-824 as a potential Oral treatment for visceral Leishmaniasis. Antimicrob Agents Chemother 57(10):4699–4706. https://doi.org/10.1128/aac.00722-13
Patterson S, et al. (2016) The anti-tubercular drug delamanid as a potential oral treatment for visceral leishmaniasis. eLife 5. doi:https://doi.org/10.7554/eLife.09744
Pena I et al (2015) New compound sets identified from high throughput phenotypic screening against three kinetoplastid parasites: an open resource. Sci Rep 5:8771. https://doi.org/10.1038/srep08771
Pina-Vazquez C, Reyes-Lopez M, Ortiz-Estrada G, de la Garza M, Serrano-Luna J (2012) Host-parasite interaction: parasite-derived and -induced proteases that degrade human extracellular matrix. J Parasitol Res 2012:748206–748224. https://doi.org/10.1155/2012/748206
Purkait B et al (2012) Mechanism of amphotericin B resistance in clinical isolates of Leishmania donovani. Antimicrob Agents Chemother 56(2):1031–1041. https://doi.org/10.1128/AAC.00030-11
Rachidi N, Taly JF, Durieu E, Leclercq O, Aulner N, Prina E, Pescher P, Notredame C, Meijer L, Späth GF (2014) Pharmacological assessment defines Leishmania donovani casein kinase 1 as a drug target and reveals important functions in parasite viability and intracellular infection. Antimicrob Agents Chemother 58(3):1501–1515. https://doi.org/10.1128/aac.02022-13
Ready PD (2014) Epidemiology of visceral leishmaniasis. ClinEpidemiol 6:147–154. https://doi.org/10.2147/CLEP.S44267
Rijal S et al (2013) Increasing failure of miltefosine in the treatment of kala-azar in Nepal and the potential role of parasite drug resistance, reinfection, or noncompliance. Clin Infect Dis 56(11):1530–1538. https://doi.org/10.1093/cid/cit102
Roy S, Kumar GA, Jafurulla M, Mandal C, Chattopadhyay A (2014) Integrity of the actin cytoskeleton of host macrophages is essential for Leishmania donovani infection. Biochim Biophys Acta 1838(8):2011–2018. https://doi.org/10.1016/j.bbamem.2014.04.017
Sangshetti JN, Kalam Khan FA, Kulkarni AA, Arote R, Patil RH (2015) Antileishmanial drug discovery: comprehensive review of the last 10 years. RSC Adv 5(41):32376–32415. https://doi.org/10.1039/C5RA02669E
Sant’Anna MRV, Diaz-Albiter H, Aguiar-Martins K, al Salem WS, Cavalcante RR, Dillon VM, Bates PA, Genta FA, Dillon RJ (2014) Colonisation resistance in the sand fly gut: Leishmania protects Lutzomyia longipalpis from bacterial infection. Parasit Vectors 7(1):329. https://doi.org/10.1186/1756-3305-7-329
Santiago MEB, Neto LS, Alexandre EC, Munari DP, Andrade MMC, Somenzari MA, Ciarlini PC, V.M.F. L (2013) Improvement in clinical signs and cellular immunity of dogs with visceral leishmaniasis using the immunomodulator P-MAPA. Acta Trop 127(3):174–180. https://doi.org/10.1016/j.actatropica.2013.04.005
Silverman JM, Chan SK, Robinson DP, Dwyer DM, Nandan D, Foster LJ, Reiner NE (2008) Proteomic analysis of the secretome of Leishmania donovani. Genome Biol 9(2):R35. https://doi.org/10.1186/gb-2008-9-2-r35
Silverman JM, Clos J, de'Oliveira CC, Shirvani O, Fang Y, Wang C, Foster LJ, Reiner NE (2010) An exosome-based secretion pathway is responsible for protein export from Leishmania and communication with macrophages. J Cell Sci 123(Pt 6):842–852. https://doi.org/10.1242/jcs.056465
Siqueira-Neto JL, Moon S, Jang J, Yang G, Lee C, Moon HK, Chatelain E, Genovesio A, Cechetto J, Freitas-Junior LH (2012) An image-based high-content screening assay for compounds targeting intracellular Leishmania donovani amastigotes in human macrophages. PLoS Negl Trop Dis 6(6):e1671. https://doi.org/10.1371/journal.pntd.0001671
Sundar S, Chakravarty J (2010) Antimony toxicity. Int J Environ Res Public Health 7(12):4267–4277. https://doi.org/10.3390/ijerph7124267
Tegazzini D, Díaz R, Aguilar F, Peña I, Presa JL, Yardley V, Martin JJ, Coteron JM, Croft SL, Cantizani J (2016) A replicative in vitro assay for drug discovery against Leishmania donovani. Antimicrob Agents Chemother 60(6):3524–3532. https://doi.org/10.1128/aac.01781-15
Telleria EL, Martins-da-Silva A, Tempone AJ, Traub-Csekö YM (2018) Leishmania, microbiota and sand fly immunity. Parasitology 145(10):1336–1353. https://doi.org/10.1017/S0031182018001014
Turner KG, Vacchina P, Robles-Murguia M, Wadsworth M, McDowell MA, Morales MA (2015) Fitness and phenotypic characterization of Miltefosine-resistant Leishmania major. PLoS Negl Trop Dis 9(7):e0003948. https://doi.org/10.1371/journal.pntd.0003948
Uliana SR, Barcinski MA (2009) Repurposing for neglected diseases. Science (New York, NY) 326(5955):935; author reply 935. https://doi.org/10.1126/science.326.5955.935-a
van Griensven J, Zijlstra EE, Hailu A (2014) Visceral Leishmaniasis and HIV coinfection: time for concerted action. PLoS Negl Trop Dis 8(8):e3023. https://doi.org/10.1371/journal.pntd.0003023
Vanaerschot M, De DS, Rijal S, Maes L, Dujardin JC, Decuypere S (2011) Antimonial resistance in Leishmania donovani is associated with increased in vivo parasite burden. PLoSOne 6(8):e23120. https://doi.org/10.1371/journal.pone.0023120
Vianna G (1912) Tratamento da leishmaniose tegumentar por injeções intravenosas de tártaro emético. An 7º Congr Bras Med Cirurg 4:426–428
WHO (2002) New treatment for leishmaniasis is 95% effective. Bull World Health Organ 80:688
WHO (2018) Leishmaniasis fact sheet. http://www.whoint/en/news-room/fact-sheets/detail/leishmaniasis. Accessed 6/5/2019
Wilke ABB, Marrelli MT (2015) Paratransgenesis: a promising new strategy for mosquito vector control. Parasit Vectors 8:342. https://doi.org/10.1186/s13071-015-0959-2
Wyatt PG, Gilbert IH, Read KD, Fairlamb AH (2011) Target validation: linking target and chemical properties to desired product profile. Curr Top Med Chem 11(10):1275–1283
Zulfiqar B, Jones AJ, Sykes ML, Shelper TB, Davis RA, Avery VM (2017a) Screening a natural product-based library against Kinetoplastid parasites. Molecules (Basel) 22(10). doi:https://doi.org/10.3390/molecules22101715
Zulfiqar B, Shelper TB, Avery VM (2017b) Leishmaniasis drug discovery: recent progress and challenges in assay development. Drug Discov Today 22(10):1516–1531. https://doi.org/10.1016/j.drudis.2017.06.004
Funding
This work was funded by the Research Fund Flanders (FWO: project G051812N, G013118N, and 12I0317N), and research funds of the University of Antwerp (TT-ZAPBOF 33049 and TOP-BOF 35017). LMPH is a partner of the Antwerp Drug Discovery Network (ADDN, www.addn.be) and the Excellence Centre “Infla-Med” (www.uantwerpen.be/infla-med).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Section Editor: Marta Teixeira
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hendrickx, S., Caljon, G. & Maes, L. Need for sustainable approaches in antileishmanial drug discovery. Parasitol Res 118, 2743–2752 (2019). https://doi.org/10.1007/s00436-019-06443-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-019-06443-2