Abstract
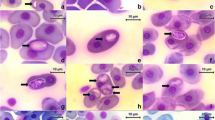
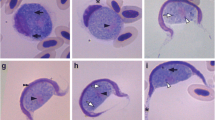
Based on morphology and morphometry of gametocytes in blood and molecular phylogenetic analysis, we described a new species of hemoparasite from the genus Haemogregarina isolated from Lepidosiren paradoxa in the eastern Amazon region. Haemogregarina daviesensis sp. nov. is characterized by monomorphic gametocytes of varying maturity stage and their dimensions were 16 ± 0.12 μm (range 13–18) in length and 6 ± 0.97 μm (range 5–8) in width. The morphological and morphometric data were not identical with other haemogregarine species from fish. All specimens of L. paradoxa analyzed were infected by H. daviesensis sp. nov. and the parasitemia level was moderate (1–28/2000 blood erythrocytes). Two sequences were obtained from L. paradoxa, and these constituted a monophyletic sister clade to the Haemogregarina species. In addition, H. daviesensis sp. nov. detected here grouped with Haemogregarina sp. sequences isolated from chelonian Macrochelys temminckii, with 99% bootstrap support. This study provides the first data on the molecular phylogeny of an intraerythrocytic haemogregarine of freshwater fish and highlights the importance of obtaining additional information on aspects of the general biology of these hemoparasites in fish populations, in order to achieve correct taxonomic classification.


Similar content being viewed by others
References
Alhaboubi AR, Pollard DA, Holman PJ (2017) Molecular and morphological characterization of a haemogregarine in the alligator snapping turtle, Macrochelys temminckii (Testudines: Chelydridae). J Parasitol 116:207–215. https://doi.org/10.1007/s00436-016-5280-2
Almeida-Val VMF, Nozawa SR, Lopes NP, Rocha APH, Mesquita-Saad LS, Silva MNP, Val AL (2010) Biology of the South American lungfish, Lepidosiren paradoxa. In: Jorgensen JM, Joss J (eds) The biology of lungfishes, 1th edn. CRC Press, New York, pp 129–138
Darriba D, Taboada GL, Doallo R, Posada D (2012) jModelTest 2: more models, new heuristics and parallel computing. Nat Methods 9:772. https://doi.org/10.1038/nmeth.2109
Davies AJ (1995) The biology of fish haemogregarines. Adv Parasitol 36:117–203. https://doi.org/10.1016/S0065-308X(08)60491-1
Davies AJ, Johnston MRL (2000) The biology of some intraerythrocytic parasites of fishes, amphibia and reptiles. Adv Parasitol 45:1–107. https://doi.org/10.1016/S0065-308X(00)45003-7
Davies AJ, Smit NJ (2001) The life cycle of Haemogregarina bigemina (Adeleina: Haemogregarinidae) in South African hosts. Folia Parasitol 48:169–177. https://doi.org/10.14411/fp.2001.029
Davies A, Amado LL, Cook R, Bianchini A, Eiras JC (2008) Potential environmental and host gender influences on prevalence of Haemogregarina platessae (Adeleorina: Haemogregarinidae) and suspected Haemohormidium terraenovae (incertae sedis) in Brazilian flounder from the Patos Lagoon Estuary, Southern Brazil. Folia Parasitol 55:161–170. https://doi.org/10.14411/fp.2008.023
Desser SS (1993) The Haemogregarinidae and Lankesterellidae. In: Kreier JP (ed) Parasitic Protozoa. Academic Press, London, pp 247–272
Diniz J, Silva E, De Souza W, Lainson R (2002) Some observations on the fine structure of trophozoites of the haemogregarine Cyrilia lignieresi (Adeleina: Haemogregarinidae) in erythrocytes of the fish Synbranchus marmoratus (Synbranchidae). Parasitol Res 88:593–597. https://doi.org/10.1007/s00436-002-0603-x
Duval L, Robert V, Csorba G, Hassanin A, Randrianarivelojosia M, Walston J, Nhim T, Goodman SM, Ariey F (2007) Multiple host-switching of Haemosporidia parasites in bats. Malar J 6:157. https://doi.org/10.1186/1475-2875-6-157
Eiras JC, Ranzani-Paiva MJT, Davies AJ (1995) Observations on Haemogregarina mugili (Apicomplexa) and Trypanosoma froesi (Sarcomastigophora) from the blood of Mugil platanus Günther, 1880 (Pisces: Mugilidae) in Brazil. Res Rev Parasitol 55:173–176
Eiras JC, Takemoto RM, Pavanelli GC, Luque JL (2012) Checklist of protozoan parasites of fishes from Brazil. Zootaxa 3221:1–25
Elsey RM (2006) Food habits of Macrochelys temminckii (alligator snapping turtle) from Arkansas and Louisiana. Southeast Nat 5:443–452. https://doi.org/10.1656/1528-7092(2006)5[443:FHOMTA]2.0.CO;2
Godfrey RD Jr, Fedynich AM, Pence DB (1987) Quantification of hematozoa in blood smear. J Wildl Dis 23:558–565. https://doi.org/10.7589/0090-3558-23.4.558
Godfrey RD Jr, Pence DB, Fedynich AM (1990) Effects of host and spatial factors on a haemoproteid community in mourning doves from western Texas. J Wildl Dis 26:435–441. https://doi.org/10.7589/0090-3558-26.4.435
Guindon S, Dufayard JF, Lefort V, Anisimova M, Hordijk W, Gascuel O (2010) New algorithms and methods to estimate maximum-likelihood phylogenies: assessing the performance of PhyML 3.0. Syst Biol 59:307–321. https://doi.org/10.1093/sysbio/syq010
Hall TA (1999) BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp Ser 41:95–98
Hayes PM, Smit NJ (2019) Molecular insights into the identification and phylogenetics of the cosmopolitan marine fish blood parasite, Haemogregarina bigemina (Adeleorina: Haemogregarinidae). Int J Parasitol - Par 8:216–220. https://doi.org/10.1016/j.ijppaw.2019.01.006
Jepps MW (1927) Note on a haemogregarine in Lepidosiren paradoxa. Parasitology 19:285–287. https://doi.org/10.1017/S0031182000005722
Jepps MW (1929) Further note on Haemogregarina Lepidosirenis. Parasitology 21:282–287. https://doi.org/10.1017/S0031182000022976
Lainson R (1981) On Cyrilia gomesi (Neiva & Pinto, 1926) gen. nov. (Haemogregarinidae) and Trypanosoma bourouli Neiva & Pinto, in the fish Synbranchus marmoratus: simultaneous transmission by the leech Haementeria lutzi in: canning EU (ed) parasitological topics. (Society of Protozoologists special publication 1) Society of Protozoologists, London 150-158
Machado LP, Wellendorf H, Brito PM (2010) On the type material of Lepidosiren paradoxa Fitzinger, 1837 (Sarcopterygii, Dipnoi). C R Biol 333:56–60. https://doi.org/10.1016/j.crvi.2009.10.005
Magro NM, de-Oliveira AT, O'Dwyer LH (2016) First report and description of a Cyrilia sp.(Apicomplexa: Haemogregarinidae) from a freshwater Cururu Stingray Potamotrygon cf. histrix (Elasmobranchii: Potamotrygonidae), from the Amazon Region, Brazil. J Fish Dis 39: 907–911. https://doi.org/10.1111/jfd.12425
Mathew JS, Bussche RAVD, Ewing SA, Malayer JR, Latha BR, Panciera RJ (2000) Phylogenetic relationships of Hepatozoon (Apicomplexa: Adeleorina) based on molecular, morphologic, and life-cycle characters. J Parasitol 86:366–372. https://doi.org/10.1645/0022-3395(2000)086[0366:PROHAA]2.0.CO;2
Nelson JS, Grande TC, Wilson MVH (2016) Fishes of the world. John Wiley & Sons, New Jersey
Oliveira AT, Araújo MLG, Pantoja-Lima J, Aride PHR, Tavares-Dias M, Brinn RP, Marcon JL (2017) Cyrilia sp. (Apicomplexa: Haemogregarinidae) in the Amazonian freshwater stingray Potamotrygon wallacei (cururu stingray) in different hydrological phases of the Rio Negro. Braz J Biol 77: 413–416. https://doi.org/10.1590/1519-6984.00416
Ricklefs RE, Fallon SM, Bermingham E (2004) Evolutionary relationships, cospeciation, and host switching in avian malaria parasites. Syst Biol 53:111–119. https://doi.org/10.1080/10635150490264987
Schouten GB (1941) Haemogregarina bertonii n. sp. hematozoario de Lepidosiren paradoxa Fitinger. Rev Soc Cien Paraguay 5:113–115
Siddall ME (1995) Phylogeny of adeleid blood parasites with a partial systematic revision of the haemogregarine complex. J Eukaryot Microbiol 42:116–125. https://doi.org/10.1111/j.1550-7408.1995.tb01551.x
Siddall ME, Desser SS (1993) Ultrastructure of merogonic development of Haemogregarina (sensu lato) myoxocephali (Apicomplexa: Adeleina) in the marine leech Malmiana scorpii and localization of infective stages in the salivary cells. Eur J Protistol 29:191–201. https://doi.org/10.1016/S0932-4739(11)80273-7
Sloboda M, Kamler M, Bulantová J, Votýpka J, Modrý D (2007) A new species of Hepatozoon (Apicomplexa: Adeleorina) from Python regius (Serpentes: Pythonidae) and its experimental transmission by a mosquito vector. J Parasitol 93:1189–1198. https://doi.org/10.1645/GE-1200R.1
Tamura K, Stecher G, Peterson D, Filipski A, Kumar S (2013) MEGA6: molecular evolutionary genetics analysis version 6.0. Mol Biol Evol 30:2725–2729
Ujvari B, Madsen T, Olsson M (2004) High prevalence of Hepatozoon spp. (Apicomplexa, Hepatozoidae) infection in water pythons (Liasis fuscus) from tropical Australia. J Parasitol 90:670–672. https://doi.org/10.1645/GE-204R
Úngari LP, Santos ALQ, O’Dwyer LH, da Silva MRL, de Melo Fava NN, Paiva GCM, Cury MC (2018) Haemogregarina podocnemis sp. nov.: description of a new species of Haemogregarina Danilewsky 1885 (Adeleina: Haemogregarinaidae) in free-living and captive yellow-spotted river turtles Podocnemis unifilis (Testudines: Podocnemididae) from Brazil. J Parasitol Res 117:1535–1548. https://doi.org/10.1007/s00436-018-5817-7
Acknowledgements
The authors thank the National Council for Scientific and Technological Development (Conselho Nacional de Desenvolvimento Científico e Tecnológico, CNPq), Brazil for the research productivity grant awarded to Dr. M. Tavares-Dias (# 303013/2015-0). To Jackson Cleiton de Souza for help in fieldwork.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Section Editor: Astrid Holzer
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Esteves-Silva, P.H., da Silva, M.R.L., O’Dwyer, L.H. et al. Haemogregarina daviesensis sp. nov. (Apicomplexa: Haemogregarinidae) from South American lungfish Lepidosiren paradoxa (Sarcopterygii: Lepidosirenidae) in the eastern Amazon region. Parasitol Res 118, 2773–2779 (2019). https://doi.org/10.1007/s00436-019-06430-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-019-06430-7