Abstract
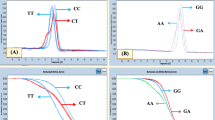
The study of pathogenesis mechanisms of larval stages in the Taeniidae has recently focused on host genetic factors, particularly toll-like receptor (TLR) variations. However, the potential role of TLR4 polymorphism in hydatidosis has not yet been sufficiently elucidated in postoperative patients. In this case-control investigation, 80 patients from Iran, including 40 with acute hydatidosis (AH) and 40 with recurrent hydatidosis (RH), and 80 ethnically matched controls were evaluated from February 2015 to February 2017. Hydatidosis patients were confirmed using radiological, immunological, and histopathological examinations. Genotyping of Asp299Gly and Thr399Ile of TLR4 single-nucleotide polymorphisms was determined by restriction fragment length polymorphism, sequencing, and phylogenetic strategies. The heterozygous mutant-type TLR4 Asp299Gly genotype indicated a tendency to be associated with the occurrence of RH (P = 0.060) and conferred a 3-fold risk for susceptibility. There was no difference in genotype frequency of Asp299Gly between patients with AH and healthy controls (P = 0.42; OR, 1.82; 95% CI, 0.11–30.1%). Interestingly, a frequency of the G allele (12%: Gly) was observed to be a risk factor for susceptibility to RH patients (P = 0.050; OR, 7.08; 95% CI, 0.97–51.5%). A relative genetic variability of TLR4 Asp299Gly was found in RH patients (haplotype diversity: 0.700) compared to AH patients and healthy controls (Hd: 0.000). The Asp299Gly genotype was dominantly identified in patients with hepatic hydatid cysts. The TLR4 Thr399Ile codon was not detected except in a patient with a pulmonary hydatid cyst. The current findings enhance our knowledge regarding the TLR4 Asp299Gly polymorphism potentially leading to the development of RH, by skewing the immune system towards a Th2 response. Identification of the Asp299Gly codon may be a diagnostic hallmark in RH patients who have undergone unsuccessful postoperative intervention. However, further studies with a higher case number are needed on ethnic population from various geographic regions, in order to confirm this hypothesis.





Similar content being viewed by others
References
Ajdary S, Ghamilouie MM, Alimohammadian MH, Riazi-Rad F, Pakzad SR (2011) Toll-like receptor 4 polymorphisms predispose to cutaneous leishmaniasis. Microbes Infect 13:226–231. https://doi.org/10.1016/j.micinf.2010.10.018
Akira S (2003) Mammalian toll-like receptors. Curr Opin Immunol 15:5–11. https://doi.org/10.1016/S0952-7915(02)00013-4
Amirmajdi MM, Sankian M, Mashhadi IE, Varasteh A, Vahedi F, Sadrizadeh A, Spotin A (2011) Apoptosis of human lymphocytes after exposure to hydatid fluid. Iran J Parasitol 6(2):9–16
Apinjoh TO, Anchang-Kimbi JK, Njua-Yafi C, Mugri RN, Ngwai AN, Rockett KA, Mbunwe E, Besingi RN, Clark TG, Kwiatkowski DP (2013) Association of cytokine and toll-like receptor gene polymorphisms with severe malaria in three regions of Cameroon. PLoS One 27: 8(11):e81071. doi:https://doi.org/10.1371/journal.pone.0081071.
Bali P, Pradhan S, Sharma D, Adak T (2013) Toll like receptor 2 and 4 polymorphisms in malaria endemic populations of India. Hum Immunol 74:223–229. https://doi.org/10.1016/j.humimm.2012.11.006
Basu M, Maji AK, Chakraborty A, Banerjee R, Mullick S, Saha P, Das S, Kanjilal SD, Sengupta S (2010) Genetic association of toll-like-receptor 4 and tumor necrosis factor-α polymorphisms with Plasmodium falciparum blood infection levels. Infect Genet Evol 10: 686–696. doi:https://doi.org/10.1016/j.meegid.2010.03.008.
Budke CM, Deplazes P, Torgerson PR (2006) Global socioeconomic impact of cystic echinococcosis. Emerg Infect Dis 12:296–303. https://doi.org/10.3201/eid1202.050499
Craig PS, McManus DP, Lightowlers MW, Chabalgoity JA, Garcia HH, Gavidia CM, Gilman RH, Gonzalez AE, Lorca M, Naquira C (2007) Prevention and control of cystic echinococcosis. Lancet Infect Dis 7:385–394. https://doi.org/10.1016/S1473-3099(07)70134-2
Ejghal R, Hida M, Bennani ML, Meziane M, Aurag R, Lemrani M (2016) The TLR2 and TLR4 gene polymorphisms in Moroccan visceral leishmaniasis patients. Acta Trop 158:77–82. https://doi.org/10.1016/j.actatropica.2016.02.020
Esposito S, Molteni CG, Zampiero A, Baggi E, Lavizzari A, Semino M, Daleno C, Groppo M, Scala A, Terranova L (2012) Role of polymorphisms of toll-like receptor (TLR) 4, TLR9, toll-interleukin 1 receptor domain containing adaptor protein (TIRAP) and FCGR2A genes in malaria susceptibility and severity in Burundian children. Malar J 11:196. https://doi.org/10.1186/1475-2875-11-196
Ghabouli-Mehrabani N, Kousha A, Khalili M, Mahami-Oskouei M, Mohammadzadeh M, Alizadeh S, Maleksabet A, Hamidi F (2014) Hydatid cyst surgeries in patients referred to hospitals in East Azerbaijan province during 2009-2011. Iran J Parasitol 9(2):233–238
Galeh TM, Spotin A, Mahami-Oskouei M, Carmena D, Rahimi MT, Barac A, Ghoyounchi R, Berahmat R, Ahmadpour E (2018) The seroprevalence rate and population genetic structure of human cystic echinococcosis in the Middle East: a systematic review and meta-analysis. Int J Surg 51(2018):39–48. https://doi.org/10.1016/j.ijsu.2018.01.025
Hajizadeh M, Ahmadpour E, Sadat ATE, Spotin A (2013) Hydatidosis as a cause of acute appendicitis: a case report. Asian Pac J Trop Dis 3:71–73. https://doi.org/10.1016/S2222-1808(13)60015-8
Hise A, Hazlett F, Bockarie M, Zimmerman P, Tisch D, Kazura J (2003) Polymorphisms of innate immunity genes and susceptibility to lymphatic filariasis. Genes Immun 4:524–527. https://doi.org/10.1038/sj.gene.6364015
Iwalokun BA, Oluwadun A, Iwalokun SO, Agomo P (2015) Toll-like receptor (TLR4) Asp299Gly and Thr399Ile polymorphisms in relation to clinical falciparum malaria among Nigerian children: a multisite cross-sectional immunogenetic study in Lagos. Genes Enviro 37:3. https://doi.org/10.1186/s41021-015-0002-z
Kabelitz D (2007) Expression and function of toll-like receptors in T lymphocytes. Curr Opin Immunol 19:39–45. https://doi.org/10.1016/j.coi.2006.11.007
Karimi M, Rostami A, Spotin A, Rouhani S (2017) Ruptured pulmonary hydatid cyst: a case report. J Parasit Dis 41(3):899–902. https://doi.org/10.1007/s12639-017-0880-z
Kawai T, Akira S (2010) The role of pattern-recognition receptors in innate immunity: update on toll-like receptors. Nature Immunol 11:373–384. https://doi.org/10.1038/ni.1863
Kesar V, Odin JA (2014) Toll-like receptors and liver disease. Liver Int 34:184–196. https://doi.org/10.1111/liv.12315
Koshki MH, Nourian A, Rahimi MT, Daryani A, Spotin A, Ahmadpour E (2017) Natural products applied against hydatid cyst protoscolices: a review of past to present. Acta Trop 176:385–394. https://doi.org/10.1016/j.actatropica.2017.09.013
Kosik-Bogacka D, Wojtkowiak-Giera A, Kolasa A, Czernomysy-Furowicz D, Lanocha N, Wandurska-Nowak E, Salamatin R, Jagodzinski P (2013) Hymenolepis diminuta: analysis of the expression of toll-like receptor genes (TLR2 and TLR4) in the small and large intestines of rats. Part II. Exp Parasitol 135:437–445. https://doi.org/10.1016/j.exppara.2013.08.002
Kropf P, Freudenberg MA, Modolell M, Price HP, Herath S, Antoniazi S, Galanos C, Smith DF, Müller I (2004) Toll-like receptor 4 contributes to efficient control of infection with the protozoan parasite Leishmania major. Infect Immun 72:1920–1928. https://doi.org/10.1128/IAI.72.4.1920-1928.2004
Lachuriya G, Garg RK, Jain A, Malhotra HS, Singh AK, Jain B, Kumar N, Verma R, Sharma PK (2016) Toll-like receptor-4 polymorphisms and serum matrix metalloproteinase-9 in newly diagnosed patients with calcified neurocysticercosis and seizures. Medicine 95(17):e3288. https://doi.org/10.1097/MD.0000000000003288
Leoratti FM, Farias L, Alves FP, Suarez-Mútis MC, Coura JR, Kalil J, Camargo EP, Moraes SL Ramasawmy R (2008) Variants in the toll-like receptor signaling pathway and clinical outcomes of malaria. J Infect Dis 198:772–780. https://doi.org/10.1086/590440
Lipoldová M, Demant P (2006) Genetic susceptibility to infectious disease: lessons from mouse models of leishmaniasis. Nat Rev Genet 7:294–305. https://doi.org/10.1038/nrg1832
Little JM, Hollands MJ, Ekberg H (1988) Recurrence of hydatid disease. World J Surg12: 700–703.
Mahami-Oskouei M, Kaseb-Yazdanparast A, Spotin A, Shahbazi A, Adibpour M, Ahmadpour E, Ghabouli-Mehrabani N (2016) Gene flow for Echinococcus granulosus metapopulations determined by mitochondrial sequences: a reliable approach for reflecting epidemiological drift of parasite among neighboring countries. Exp Parasitol 171:77–83. https://doi.org/10.1016/j.exppara.2016.10.017
Mahami-Oskouei M, Ghabouli-Mehrabani N, Miahipour A, Fallah E (2014) Molecular characterization and sequence analysis of Echinococcus granulosus from sheep isolates in East Azerbaijan province, northwest of Iran. J Parasit Dis 40(3):785–790. https://doi.org/10.1007/s12639-014-0579-3.
Mahami-Oskouei M, Ghabouli-Mehrabani N, Miahipour A, Fallah E, Shahbazi A, Mazhari N, Hamidi F (2015) Genotypic characterization of Echinococcus granulosus isolates based on the mitochondrial cytochrome c oxidase 1 (cox1) gene in Northwest Iran. Trop Biomed 32(4):717–725
Maldonado C, Trejo W, Ramirez A, Carrera M, Sánchez J, López-Macias C, Isibasi A (2000) Lipophosphopeptidoglycan of Entamoeba histolytica induces an antiinflammatory innate immune response and downregulation of toll-like receptor 2 (TLR-2) gene expression in human monocytes. Arch Med Res 31:S71–S73. https://doi.org/10.1016/S0188-4409(00)00199-5
Misch EA, Hawn TR (2008) Toll-like receptor polymorphisms and susceptibility to human disease. Clin Sci 114:347–360. https://doi.org/10.1042/CS20070214
Mockenhaupt FP, Cramer JP, Hamann L, Stegemann MS, Eckert J, Oh NR, Otchwemah RN, Dietz E, Ehrhardt S, Schröder NW (2006) Toll-like receptor (TLR) polymorphisms in African children: common TLR-4 variants predispose to severe malaria. Proc Natl AcadSci U S A 103:177–182. https://doi.org/10.1073/pnas.0506803102
Moro P, Schantz PM (2009) Echinococcosis: a review. Int J Infect Dis 13(2):125–133. https://doi.org/10.1016/j.ijid.2008.03.037
Mun HS, Aosai F, Norose K, Piao LX, Fang H, Akira S, Yano A (2005) Toll-like receptor 4 mediates tolerance in macrophages stimulated with Toxoplasma gondii-derived heat shock protein 70. Infect Immun 73:4634–4642. https://doi.org/10.1128/IAI.73.8.4634-4642.2005
Naseri M, Akbarzadeh A, Spotin A, Akbari NAR, Mahami-Oskouei M, Ahmadpour E (2016) Scolicidal and apoptotic activities of albendazole sulfoxide and albendazole sulfoxide-loaded PLGA-PEG as a novel nanopolymeric particle against Echinococcus granulosus protoscoleces. Parasitol Res 115:4595–4603. https://doi.org/10.1007/s00436-016-5250-8
Netea MG, Wijmenga C, O'neill LA (2012) Genetic variation in toll-like receptors and disease susceptibility. Nat Immunol 13:535–542. https://doi.org/10.1038/ni.2284
Pappas PW, Read CP (1975) Membrane transport in helminth parasites: a review. Exp Parasitol 37:469–530. https://doi.org/10.1016/0014-4894(75)90016-8
Possenti A, Manzano-Román R, Sánchez-Ovejero C, Boufana B, La Torre G, Siles-Lucas M, Casulli A (2016) Potential risk factors associated with human cystic echinococcosis: systematic review and meta-analysis. PLOS Negl Trop 10:e0005114. https://doi.org/10.1371/journal.pntd.0005114
Prousalidis J, Kosmidis C, Anthimidis G, Kapoutzis K, Karamanlis E, Fachantidis E (2012) Postoperative recurrence of cystic hydatidosis. Can J Surg 55:15–20. https://doi.org/10.1503/cjs.013010
Qavi A, Garg RK, Malhotra HS, Jain A, Kumar N, Malhotra KP, Srivastava PK, Verma R, Sharma PK (2016) Disseminated cysticercosis: clinical spectrum, Toll-like receptor-4 gene polymorphisms and role of albendazole: A prospective follow-up of 60 cases with a review of 56 published cases. Medicine 95. doi: https://doi.org/10.1097/MD.0000000000004882.
Rahimi MT, Ahmadpour E, Esboei BR, Spotin A, Koshki MHK, Alizadeh, A, Honary S, Barabadi H, Mohammadi MA (2015) Scolicidal activity of biosynthesized silver nanoparticles against Echinococcus granulosus protoscolices. Int J Surg 19: 128–133. doi:https://doi.org/10.1016/j.ijsu.2015.05.043.
Rasouli M, Keshavarz M, Kalani M, Moravej A, Kiany S, Badiee P (2012) Toll-like receptor 4 (TLR4) polymorphisms in Iranian patients with visceral leishmaniasis. Mol Biol Rep 39:10795–10802. https://doi.org/10.1007/s11033-012-1973-5
Rezazadeh M, Hajilooi M, Haidari M, Rafiei A, Ahmad Alavi S, Keramat F (2006) Association of susceptibility to brucellosis and interleukin-4 promoter polymorphism. Scand J Infect Dis 1:11–12. https://doi.org/10.1080/00365540600786473
Rigano R, Profumo E, Ioppolo S, Notargiacomo S, Teggi A, Siracusano A (1999) Serum cytokine detection in the clinical follow up of patients with cystic echinococcosis. J. Clin Exp Immunol 115:503–507. https://doi.org/10.1046/j.1365-2249.1999.00843.x
Rouhani S, Parvizi P, Spotin A (2013) Using specific synthetic peptide (p176) derived AgB 8/1-kDa accompanied by modified patient’s sera: a novel hypothesis to follow-up of cystic echinococcosis after surgery. Med Hypotheses 81:557–560. https://doi.org/10.1016/j.mehy.2013.07.003
Schröder NW, Schumann RR (2005) Single nucleotide polymorphisms of toll-like receptors and susceptibility to infectious disease. Lancet Infect Dis 5:156–164. https://doi.org/10.1016/S1473-3099(05)01308-3
Shan JY, Ji WZ, Li HT, Tuxun T, Lin RY, Wen H (2011) TLR2 and TLR4 expression in peripheral blood mononuclear cells of patients with chronic cystic echinococcosis and its relationship with IL-10. Parasite Immunol 33:692–696. https://doi.org/10.1111/j.1365-3024.2011.01335.x
Shariatzadeh SA, Spotin A, Gholami S, Fallah E, Hazratian T, Mahami-Oskouei M, Montazeri F, Moslemzadeh HR, Shahbazi A (2015) The first morphometric and phylogenetic perspective on molecular epidemiology of Echinococcus granulosus sensu lato in stray dogs in a hyperendemic Middle East focus, northwestern Iran. Parasit Vectors 8:409. https://doi.org/10.1186/s13071-015-1025-9
Singh A, Garg RK, Jain A, Malhotra HS, Prakash S, Verma R, Sharma PK (2015) Toll like receptor-4 gene polymorphisms in patients with solitary cysticercus granuloma. J Neurol Sci 355:180–185. https://doi.org/10.1016/j.jns.2015.06.014
Spotin A, Gholami S, Nasab AN, Fallah E, Oskouei MM, Semnani V, Shariatzadeh SA, Shahbazi A (2015) Designing and conducting in silico analysis for identifying of Echinococcus spp. with discrimination of novel haplotypes: an approach to better understanding of parasite taxonomic. Parasitol Res 114:1503–1509. https://doi.org/10.1007/s00436-015-4334-1
Spotin A, Mahami-Oskouei M, Harandi MF, Baratchian M, Bordbar A, Ahmadpour E, Ebrahimi S (2017) Genetic variability of Echinococcus granulosus complex in various geographical populations of Iran inferred by mitochondrial DNA sequences. Acta Trop 165:10–16. https://doi.org/10.1016/j.actatropica.2016.03.002
Spotin A, Majdi MMA, Sankian M, Varasteh A (2012a) The study of apoptotic bifunctional effects in relationship between host and parasite in cystic echinococcosis: a new approach to suppression and survival of hydatid cyst. Parasitol Res 110:1979–1984. https://doi.org/10.1007/s00436-011-2726-4
Spotin A, Mokhtari Amirmajdi M, Sankian M, Varasteh A, Shamsian AA, Vahedi F (2012b) Expression of apoptosis inducing-ligands, TRAIL and Fas-L in hydatid cyst germinal layer and normal tissue. J Ardabil Univ Med Sci 12:7–15
Torgerson PR, Devleesschauwer B, Praet N, Speybroeck N, Willingham AL, Kasuga F, Rokni MB, Zhou XN, F evre EM, Sripa B, Gargouri N, Furst T, Budke CM, Carabin H, Kirk MD, Angulo FJ, Havelaar A, de Silva N (2015) World Health Organization estimates of the global and regional disease burden of 11 foodborne parasitic diseases, 2010: a data synthesis. PLoS Med 12:e1001920. https://doi.org/10.1371/journal.pmed.1001920
Tuxun T, Ma H, Apaer S, Zhang H, Aierken A, Li YP, Lin RY, Zhao JM, Zhang JH, Wen H (2015) Expression of toll-like receptors 2 and 4 and related cytokines in patients with hepatic cystic and alveolar echinococcosis. Mediat Inflamm 2015:632760. https://doi.org/10.1155/2015/632760
Velasco-Tirado V, Romero-Alegría Á, Belhassen-García M, Alonso-Sardón M, Esteban-Velasco C, López-Bernús A, Carpio-Perez A, López MFJ, Bellido JLM, Muro A (2017) Recurrence of cystic echinococcosis in an endemic area: a retrospective study. BMC Infect Dis 17:455. https://doi.org/10.1186/s12879-017-2556-9
Verma A, Prasad KN, Gupta RK, Singh AK, Nyati KK, Rizwan A, Pandey CM, Paliwal VK (2010) Toll-like receptor 4 polymorphism and its association with symptomatic neurocysticercosis. J Infect Dis 202:1219–1225. https://doi.org/10.1086/656395
Weitzel T, Zulantay I, Danquah I, Hamann L, Schumann RR, Apt W, Mockenhaupt FP (2012) Mannose-binding lectin and toll-like receptor polymorphisms and Chagas disease in Chile. Am J Trop Med Hyg 86:229–232. https://doi.org/10.4269/ajtmh.2012.11-0539
Wujcicka W, Gaj Z, Wilczyński J, Nowakowska D (2015) Possible role of TLR4 and TLR9 SNPs in protection against congenital toxoplasmosis. Eur J Clin Microbiol Infect Dis 34:2121–2129. https://doi.org/10.1007/s10096-015-2461-3
Zafra G, Flórez O, Morillo CA, Echeverría LE, Martín J, González CI (2008) Polymorphisms of toll-like receptor 2 and 4 genes in Chagas disease. Mem Inst Oswaldo Cruz 103:27–30. https://doi.org/10.1590/S0074-02762008000100004
Zakeri S, Pirahmadi S, Mehrizi AA, Djadid ND (2011) Genetic variation of TLR-4, TLR-9 and TIRAP genes in Iranian malaria patients. Malar J 10:77. https://doi.org/10.1186/1475-2875-10-77
Acknowledgments
This study was financially supported by Immunology Research Center, Tabriz University of Medical Sciences, Tabriz, Iran (Grant no. 95/2-5/1). This is a report of a database from the thesis of Jafar Noori registered in Tabriz University of Medical Sciences.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All patients provided written informed consent for participation in the study. The study protocol was approved by the Ethical Review Committee of Tabriz University of Medical Sciences (TUOMS; No: 5/D/56350).
Conflicts of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Noori, J., Spotin, A., Ahmadpour, E. et al. The potential role of toll-like receptor 4 Asp299Gly polymorphism and its association with recurrent cystic echinococcosis in postoperative patients. Parasitol Res 117, 1717–1727 (2018). https://doi.org/10.1007/s00436-018-5850-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-018-5850-6