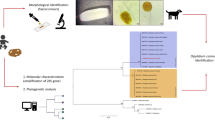
Abstract
Paratanaisia are eucotylidae digeneans that affect the upper urinary tract of birds. This genus contains three species (Paratanaisia bragai, P. robusta, and P. confusa) with similar morphological features. Macroscopic and microscopic damage caused by these parasites ranges from the irrelevant to significant lesions. This study aimed to describe the histological, morphological, and molecular features of the renal tissues and parasite specimens obtained from naturally infected free-ranging and captive wild birds in Brazil. Histopathological evaluations were performed on 103 slides containing kidney tissue sections from parasitized birds. Parasites were observed inside the collecting ducts, causing the dilation and destruction of the lining epithelial cells and alterations in other structures of the renal parenchyma. Such findings indicate that Paratanaisia have pathogenic potential in a wide range of hosts, suggesting low host specificity. The parasites recovered from the kidneys of 10 birds, including Columbiformes, Galliformes, Strigiformes, and Cuculiformes, were morphologically evaluated and identified as Paratanaisia sp. Formalin-fixed paraffin-embedded kidney fragments were subjected to conventional PCR assays targeting the 18S and 28S rDNA genes. A Bayesian inference analysis based on an 800-bp 18S rDNA gene fragment separated the trematode genus accurately, clustering all of the parasites tested with a previously described P. bragai specimen. Analyses on a small fragment of the 28S rDNA gene did not allow for accurately differentiating the Paratanaisia species. Therefore, further morphological studies with additional molecular markers are necessary to improve our understanding of the alpha-taxonomy of this group.



Similar content being viewed by others
References
Abdo W, Sultan K (2013) Histopathological findings of the kidney Trematoda Paratanaisia spp. (Digenea: Eucotylidae) in cattle egret (Bubulcus ibis). Bras J Vet Parasitol 22(2):312–313. https://doi.org/10.1590/S1984-29612013000200050
Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ (1990) Basic local alignment search tool. J Mol Biol 215:403–410. https://doi.org/10.1006/jmbi.1990.9999
Birkenheuer AJ, Levy MG, Breitschwerdt EB (2003) Development and evaluation of a seminested PCR for detection and differentiation of Babesia gibsoni (Asian genotype) and B. canis DNA in canine blood samples. J Clin Microbiol 41(9):4172–4177. https://doi.org/10.1128/JCM.41.9.4172-4177.2003
Brandolini SVPB, Amato SB (2007) Morfologia externa de espécimes adultos de Paratanaisia bragai (Santos, 1934) (Digenea: Eucotylidae). Bras J Vet Parasitol 16(4):129–132. https://doi.org/10.1590/S0101-81752006000400017
Brener B, Tortelly R, Menezes RC, Muniz-Pereira LC, Pinto RM (2006) Prevalence and pathology of the nematode Heterakis gallinarum, the trematode Paratanaisia bragai, and the protozoan Histomonas meleagridis in the turkey, Meleagris gallopavo. Mem Inst Oswaldo Cruz 101(6):677–681. https://doi.org/10.1590/S0074-02762006000600017
Ewing B, Hillier L, Wendl MC, Green P (1998) Base-calling of automated sequencer traces using phred. I. Accuracy assessment. Genome Res 8:175–185. https://doi.org/10.1101/gr.8.3.175
Giovannoni M, Malheiro DM (1952) Incidência de parasitas em Columba livia domestica. Rev Fac Med Vet 4(4):595–598. https://doi.org/10.11606/issn.2318-5066.v4i4p595-598
Gomes DC, Menezes RC, Tortelly R, Pinto RM (2005) Pathology and first occurrence of the kidney trematode Paratanaisia bragai (Santos, 1934) Freitas, 1959 (Digenea: Eucotylidae) in Phasianus colchicus L., 1758, from Brazil. Mem Inst Oswaldo Cruz 100(3):285–288. https://doi.org/10.1590/S0074-02762005000300013
Hudson PJ, Dobson AP, Newborn D (1998) Prevention of population cycles by parasite removal. Science 282(5397):2256–2258. https://doi.org/10.1126/science.282.5397.2256
Huelsenbeck JP, Ronquist F (2001) MRBAYES: Bayesian inference of phylogenetic trees. Bioinformatics 17(8):754–755. https://doi.org/10.1093/bioinformatics/17.8.754
Kazutaka K, Misawa K, Kuma K, Miyata T (2002) MAFFT: a novel method for rapid multiple sequence alignment based on fast Fourier transform. Nucleic Acids Res 30(14):3059–3066. https://doi.org/10.1093/nar/gkf436
Keller DG, Araújo JLB (1992) Ciclo evolutivo de Paratanaisia bragai (Santos, 1934) (Trematoda, Eucotylidae) com novo hospedeiro intermediário no Brasil: Leptinaria inilamellata (D’Orbigny, 1835) (Gastropoda, Pulmonata, Sebulinidae) em condições de laboratório. Bras J Vet Parasitol 1:89–92
Luppi MM, Melo AL, Motta ROC, Malta MCC, Gardiner CH, Santos LR (2007) Granulomatous nephritis in psittacines associated with parasitism by the trematode Paratanaisia spp. Vet Parasitol 146(3-4):363–366. https://doi.org/10.1016/j.vetpar.2007.03.011
Mapeli EB, Nascimento AA, Szabó MPJ, Tebaldi JH (2003) Infecções naturais por helmintos em perdizes (Rhynchotus rufescens Temminck, 1815) de cativeiro, no município de Jaboticabal, estado de São Paulo. Arq Inst Biol 70:415–418
Menezes RC, Mattos DG Jr, Gomes DC, Tortelly R, Muniz-Pereira LC, Pinto RM (2001) Trematodes of free range reared guinea fowls (Numida meleagris Linnaeus, 1758) in the state of Rio de Janeiro, Brazil: morphology and pathology. Avian Pathol 30(3):209–214. https://doi.org/10.1080/03079450124448
Miller MA, Pfeiffer W, Schwartz T (2010) Creating the CIPRES science gateway for inference of large phylogenetic trees. In: Proceedings of the Gateway Computing Environments Workshop (GCE), IEEE, pp 1–8
Momo C, Werther K (2013) Parasitismo renal em perdizes (Rhynchotus rufescens) criadas em cativeiro. PUBVET 7:1571–1652
Momo C, Garrido E, Werther K (2016) Anatomopathological findings in captive-raised winged tinamou (Rhynchotus rufescens). Braz J Vet Res Anim Sci 53(3):227–234. https://doi.org/10.11606/issn.1678-4456.v53i3p227-234
Pinto RM, Menezes RC, Tortelly R (2004) Systematic and pathologic study of Paratanaisia bragai (Santos, 1934) Freitas, 1959 (Digenea, Eucotylidae) infestation in ruddy ground dove Columbina talpacoti (Temminck, 1811). Arq Bras Med Vet Zootec 56(4):472–479. https://doi.org/10.1590/S0102-09352004000400008
Prastowo J, Sahara A, Marganingsih C, Ariyadi B (2014) Identification of renal parasite and its blood urea-creatinine profile on the Indonesian indigenous pigeons. Int J Poult Sci 13:385–389
Routtu J, Grunberg D, Izhar R, Dagan Y, Guttel Y, Ucko M, Ben-ami F (2014) Selective and universal primers for trematode barcoding in freshwater snails. Parasitol Res 113(7):2535–2540. https://doi.org/10.1007/s00436-014-3903-z
Sanger F, Nicklen S, Coulson AR (1977) Dna sequencing with chain-terminating inhibitors. Proc Natl Acad Sci U S A 74(12):5463–5467. https://doi.org/10.1073/pnas.74.12.5463
Santi M, André MR, Hoppe EGL, Werther K (2017) Occurrence of Paratanaisia confusa Freitas, 1951 in free-living guira cuckoo (Guira guira, Cuculiformes: Crotophagidae). Bras J Vet Parasitol 26(2):248–251. https://doi.org/10.1590/S1984-29612017014
Sick H (1997) Ornitologia brasileira. Nova Fronteira, Rio de Janeiro
Stöver BC, Müller KF (2010) TreeGraph 2: combining and visualizing evidence from different phylogenetic analyses. BMC Bioinf 11(1):1–9. https://doi.org/10.1186/1471-2105-11-7
Taroda A, Barros LD, Zulpo DL, Cunha IAL, Paiva MCDC, Sammi AS, Santos JR, Yamamura MH, Vidoto O, Garcia JL (2013) Occurrence of gastrointestinal and renal helminths in Zenaida auriculata (Des Murs, 1847) trap-captured from Brazil. Bras J Vet Parasitol 22(3):415–419. https://doi.org/10.1590/S1984-29612013000300016
Tavela AO, Carretta M Jr, Oliveira AR, Carneiro FT, Silva VHD, Braga FR, Peixoto JV, Carvalho GD, Araújo JV, Paula TAR (2014) Parasitism by Paratanaisia bragai (Digenea, Eucotylidae) in common waxbill (Estrilda astrild). Arq Bras Med Vet Zootec 66(4):1276–1280. https://doi.org/10.1590/1678-7136
Travassos L (1950) Introdução ao estudo da helmintologia. Rev Bras Biol 173 p
Travassos L, Freitas JFT, Kohn A (1969) Gênero Paratanaisia Freitas, 1969. Mem Inst Oswaldo Cruz 67:340–343
Unwin S, Chantrey J, Chatterton J, Aldhoun JA, Littlewood DTJ (2013) Renal trematode infection due to Paratanaisia bragai in zoo housed Columbiformes and a red bird-of-paradise (Paradisaea rubra). Int J Parasitol Parasites Wildl 2:32–41. https://doi.org/10.1016/j.ijppaw.2012.11.001
Xavier VB, Oliveira-Menezes A, Santos MAJ, Amato SB, Torres EJL, Pinheiro J, Brandolini SVPB (2015) Histopathological changes in the kidneys of vertebrate hosts infected naturally and experimentally with Paratanaisia bragai (Trematoda, Digenea). Bras J Vet Parasitol 24(2):241–246. https://doi.org/10.1590/S1984-29612015017
Funding
This work was supported by the National Counsel of Technological and Scientific Development–CNPq and São Paulo Research Foundation–FAPESP (Process No. 2015/22851-8).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Animal procedures and management protocols were approved by the Ethics Committee on Animal Use (CEUA) of the School of Agricultural and Veterinarian Sciences (FCAV/Unesp) (protocol number 009414/15) and by Institute Chico Mendes for Conservation of Biodiversity (ICMBio no. 49343-1
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Section Editor: Christoph G. Grevelding
Rights and permissions
About this article
Cite this article
De Santi, M., André, M.R., Lux Hoppe, E.G. et al. Renal trematode infection in wild birds: histopathological, morphological, and molecular aspects. Parasitol Res 117, 883–891 (2018). https://doi.org/10.1007/s00436-018-5767-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-018-5767-0