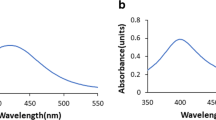
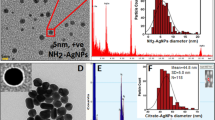
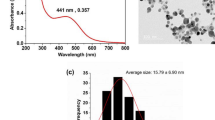
Abstract
Infectious diseases are the leading cause of morbidity and mortality, killing more than 15 million people worldwide. This is despite our advances in antimicrobial chemotherapy and supportive care. Nanoparticles offer a promising technology to enhance drug efficacy and formation of effective vehicles for drug delivery. Here, we conjugated amphotericin B, nystatin (macrocyclic polyenes), and fluconazole (azole) with silver nanoparticles. Silver-conjugated drugs were synthesized successfully and characterized by ultraviolet-visible spectrophotometry, Fourier transform infrared spectroscopy, and atomic force microscopy. Conjugated and unconjugated drugs were tested against Acanthamoeba castellanii belonging to the T4 genotype using amoebicidal assay and host cell cytotoxicity assay. Viability assays revealed that silver nanoparticles conjugated with amphotericin B (Amp-AgNPs) and nystatin (Nys-AgNPs) exhibited significant antiamoebic properties compared with drugs alone or AgNPs alone (P < 0.05) as determined by Trypan blue exclusion assay. In contrast, conjugation of fluconazole with AgNPs had limited effect on its antiamoebic properties. Notably, AgNP-coated drugs inhibited amoebae-mediated host cell cytotoxicity as determined by measuring lactate dehydrogenase release. Overall, here we present the development of a new formulation of more effective antiamoebic agents based on AgNPs coated with drugs that hold promise for future applications.




Similar content being viewed by others
References
Ali SM, Siddiqui R, Ong SK, Shah MR, Anwar A, Heard PJ, Khan NA (2017) Identification and characterization of antibacterial compound (s) of cockroaches (Periplaneta americana). Appl Microbiol Biotechnol 101(1):253–286. https://doi.org/10.1007/s00253-016-7872-2
Anselmo A, Mitragotri S (2016) Nanoparticles in the clinic. Bioeng Transl Med 1:10–29
Anwar A, Shah MR, Muhammad SP, Afridi S, Ali K (2016) Thio-pyridinium capped silver nanoparticle based supramolecular recognition of Cu (I) in real samples and T-lymphocytes. New J Chem 40(7):6480–6486. https://doi.org/10.1039/C5NJ03609G
Aqeel Y, Siddiqui R, Anwar A, Shah MR, Khan NA (2016) Gold nanoparticle conjugation enhances the antiacanthamoebic effects of chlorhexidine. Antimicrob Agents Chemother 60(3):1283–1288. https://doi.org/10.1128/AAC.01123-15
Bourichi H, Brik Y, Hubert P, Cherrah Y, Bouklouze A (2012) Solid-state characterization and impurities determination of fluconazol generic products marketed in Morocco. J Pharm Anal 2(6):412–421. https://doi.org/10.1016/j.jpha.2012.05.007
Dakal TC, Kumar A, Majumdar RS, Yadav V (2016) Mechanistic basis of antimicrobial actions of silver nanoparticles. Front Microbiol 7:1831. https://doi.org/10.3389/fmicb.2016.01831
Fuerst PA, Booton GC, Crary M (2015) Phylogenetic analysis and the evolution of the 18S rRNA gene typing system of Acanthamoeba. J Eukaryot Microbiol 62(1):69–84. https://doi.org/10.1111/jeu.12186
Girois SB, Chapuis F, Decullier E, Revol BGP (2006) Adverse effects of antifungal therapies in invasive fungal infections: review and meta-analysis. Eur J Clin Microbiol Infect Dis 25(2):138–149. https://doi.org/10.1007/s10096-005-0080-0
Grace AN, Pandian K (2007) Antibacterial efficacy of aminoglycosidic antibiotics protected gold nanoparticles—a brief study. Colloid Surf A: Physicochem Eng ASp 297(1-3):63–70. https://doi.org/10.1016/j.colsurfa.2006.10.024
Ishibashi Y, Matsumoto Y, Kabata T, Watanabe R, Hommura S, Yasuraoka K, Ishii K (1990) Oral itraconazole and topical miconazole with débridement for Acanthamoeba keratitis. Am J Ophtalmol 109(2):121–126. https://doi.org/10.1016/S0002-9394(14)75974-4
Khan NA (2006) Acanthamoeba: biology and increasing importance in human health. FEMS Microbiol Rev 30(4):564–595. https://doi.org/10.1111/j.1574-6976.2006.00023.x
Khan NA, Siddiqui R (2009) Acanthamoeba affects the integrity of human brain microvascular endothelial cells and degrades the tight junction proteins. Int J Parasitol 39(14):1611–1616. https://doi.org/10.1016/j.ijpara.2009.06.004
Li X, Robinson SM, Gupta A, Saha K, Jiang Z, Moyano DF, Sahar A, Riley MA, Rotello VM (2014) Functional gold nanoparticles as potent antimicrobial agents against multi-drug-resistant bacteria. ACS Nano 8(10):10682–10686. https://doi.org/10.1021/nn5042625
Maincent P, Le Verge R, Sado P, Couvreur P, Devissaguet JP (1986) Disposition kinetics and oral bioavailability of vincamine-loaded polyalkyl cyanoacrylate nanoparticles. J Pharm Sci 75(10):955–958. https://doi.org/10.1002/jps.2600751009
Marciano-Cabral F, Cabral G (2003) Acanthamoeba spp. as agents of disease in humans. Clin Microbiol Rev 16(2):273–307. https://doi.org/10.1128/CMR.16.2.273-307.2003
Martinez AJ, Visvesvara GS (1991) Laboratory diagnosis of pathogenic free-living amoebas: Naegleria, Acanthamoeba, and Leptomyxid. Clin Lab Med 11(4):861–872
Martín-Navarro CM, López-Arencibia A, Sifaoui I, Reyes-Batlle M, Valladares B, Martínez-Carretero E, Piñero JE, Maciver SK, Lorenzo-Morales J (2015) Statins and voriconazole induce programmed cell death in Acanthamoeba castellanii. Antimicrob Agents Chemother 59(5):2817–2824. https://doi.org/10.1128/AAC.00066-15
O'Connell KM, Hodgkinson JT, Sore HF, Welch M, Salmond GP, Spring DR (2013) Combating multidrug-resistant bacteria: current strategies for the discovery of novel antibacterials. Angew Chem Int Ed 52(41):10706–10733. https://doi.org/10.1002/anie.201209979
Radwan MA, AlQuadeib BT, Šiller L, Wright MC, Horrocks B (2017) Oral administration of amphotericin B nanoparticles: antifungal activity, bioavailability and toxicity in rats. Drug Deliv 24(1):40–50. https://doi.org/10.1080/10717544.2016.1228715
Rodino S, Butu M, Negoescu C, Caunii A, Cristina RT, Butnariu M (2014) Spectrophotometric method for quantitative determination of nystatin antifungal agent in pharmaceutical formulations. Digest J Nanomater Biostruct 9:1215–1222
Seal DV, Hay J, Kirkness CM (1995) Chlorhexidine or polyhexamethylene biguanide for Acanthamoeba keratitis. Lancet 345(8942):136. https://doi.org/10.1016/S0140-6736(95)90106-X
Singh A, Sharma PK, Majumdar DK (2011) Development and validation of different UV-spectropjotometric methods for the estimation of fluconazole in bulk and in solid dosage form. Indian J Chem Technol 18:357–362
Sissons J, Alsam S, Stins M, Rivas AO, Morales JL, Faull J, Khan NA (2006) Use of in vitro assays to determine effects of human serum on biological characteristics of Acanthamoeba castellanii. J Clin Microbiol 44(7):2595–2600. https://doi.org/10.1128/JCM.00144-06
Thomson S, Rice CA, Zhang T, Edrada-Ebel R, Henriquez FL, Roberts CW (2017) Characterisation of sterol biosynthesis and validation of 14α-demethylase as a drug target in Acanthamoeba. Sci Rep 7(1):8247. https://doi.org/10.1038/s41598-017-07495-z
Visvesvara GS, Moura H, Schuster FL (2007) Pathogenic and opportunistic free-living amoebae: Acanthamoeba spp., Balamuthia mandrillaris, Naegleria fowleri, and Sappinia diploidea. FEMS Immunol Med Microbiol 50(1):1–26. https://doi.org/10.1111/j.1574-695X.2007.00232.x
Walsh MD, Hanna SK, Sen J, Rawal S, Cabral CB, Yurkovetskiy AV, Fram RJ, Lowinger TB, Zamboni WC (2012) Pharmacokinetics and antitumor efficacy of XMT-1001, a novel, polymeric topoisomerase I inhibitor, in mice bearing HT-29 human colon carcinoma xenografts. Clin Cancer Res 18(9):2591–2602. https://doi.org/10.1158/1078-0432.CCR-11-1554
Wilczewska AZ, Niemirowicz K, Markiewicz KH, Car H (2012) Nanoparticles as drug delivery systems. Pharmacol Rep 64(5):1020–1037. https://doi.org/10.1016/S1734-1140(12)70901-5
Yang X, Yang J, Wang L, Ran B, Jia Y, Zhang L, Yang G, Shao H, Jiang X (2017) Pharmaceutical intermediate modified gold nanoparticles: against multidrug-resistant bacteria and wound-healing application via electrospun ccaffold. ACS Nano 11(6):5737–5745. https://doi.org/10.1021/acsnano.7b01240
Zazo H, Colino CI, Lanao JM (2016) Current applications of nanoparticles in infectious diseases. J Control Release 224:86–102. https://doi.org/10.1016/j.jconrel.2016.01.008
Zhao G, Stevens SE (1998) Multiple parameters for the comprehensive evaluation of the susceptibility of Escherichia coli to the silver ion. Biometals 11(1):27–32. https://doi.org/10.1023/A:1009253223055
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Anwar, A., Siddiqui, R., Hussain, M.A. et al. Silver nanoparticle conjugation affects antiacanthamoebic activities of amphotericin B, nystatin, and fluconazole. Parasitol Res 117, 265–271 (2018). https://doi.org/10.1007/s00436-017-5701-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-017-5701-x