Abstract
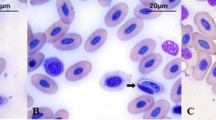
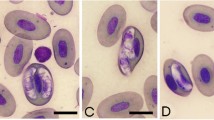
Hepatozoon sp. are parasites that commonly infect frogs and arthropod vectors. This species has variability in the morphological and morphometric characteristics. Due to these variations, the naming of the species is thus impaired and only by visualizing the sporogonic cycle in vector and by molecular studies this problem can be solved. Recently, the use of molecular genetics has helped the species denomination. In this work, we collected 145 frogs (68 Leptodactylus chaquensis and 77 Leptodactylus podicipinus) in different sampling sites, where were found 18 (26.47 %) L. chaquensis and 24 (31.17 %) L. podicipinus parasitized; besides of gamonts, schizogonic forms were also seen in animals organs. The positivity difference between the collection sites for both frog species was not significant (p = 0.958). Comparing gamonts found in each species of anuran, we observed differences in morphology. The comparison in the molecular level for L. podicipinus was not possible due to small amount of blood obtained, just L. chaquensis had their parasites DNA sequenced. The amplified and sequenced samples, named HEP1 to HEP10, are presented in the phylogenetic tree as a different branch from other haemogregarines described on other hosts. Therefore, we have seen that, although the morphology and morphometry of the collected parasites at each site showed differences, the sequencing of these samples revealed identical species of Hepatozoon, and different compared to those from GenBank, thereby demonstrating that the species of Hepatozoon in L. chaquensis observed in this study probably represent a new species.




Similar content being viewed by others
References
Bardsley JE, Harmsen R (1973) The trypanosomes of anura. In: Dawes B (ed) Advance parasitology, vol 7. Academic Press, London, pp 1–73
Barta JR, Desser SS (1984) Blood parasites of amphibians from Algonquin Park, Ontario. J Wildl Dis 20(3):180–189. doi:10.7589/0090-3558-20.3.180
Cai JMD, Collins MAC, Donald V, Thompson DE (1992) PCR cloning and nucleotide sequence determination of the 18S rRNA genes and internal transcribed spacer 1 of the protozoan parasites Cryptosporidium parvum and Cryptosporidium muris. Biochim Biophys Acta 1131:317–320. doi:10.1016/0167-4781(92)90032-U
Costa SCG, Pessoa SB, Pereira NM, Colombo T (1973) The life history of Hepatozoon leptodactyli (Lesage, 1908) Pessoa, 1970—a parasite of the common laboratory animal—the frog of the genus Leptodactylus. Mem Inst Oswaldo Cruz 71(1/2):1–8. doi:10.1590/S0074-02761973000100001
Costa Neto PLO (1994) Estatística. Edgard Blucher LTDA, São paulo
Clark GW, Bradford J (1969) Blood parasites of some reptiles of the Pacific northwest. J Protozool 16:576–581. doi:10.1111/j.1550-7408.1969.tb.02316.x
Desser SS, Hong H, Martin DS (1995) The life history, ultrastructure, and experimental transmission of Hepatozoon catesbianae n. comb., an apicomplexan parasite on the bullfrog, Rana catesbeiana and the mosquito, Culex territans in Algoquin Park, Ontário. J Parasitol 81:212–222. doi:10.2307/3283922
Desser SS (2001) The blood parasites of anurans from Costa Rica with reflections on the taxonomy of their trypanosomes. J Parasitol 87(1):152–160. doi:10.1645/0022-3395(2001)087[0152:TBPOAF]2.0.CO;2
Fantham HB, Porter A, Richardson LR (1942) Some Haematozoa observed in vertebrates in earsten Canada. Parasitol 34:199–226. doi:10.1017/S0031182000016176
Felsenstein J (1985) Confidence limits on phylogenies: an approach using the bootstrap. Evol 39(4):783–791. doi:10.2307/2408678
Ferreira RC, Campaner M, Viola LB, Takata CSA, Takeda GF, Teixeira MMG (2007) Morphological and molecular diversity and phylogenetic relationships among trypanosomes from the Amazonia, Atlantic Forest and Pantanal biomes in Brazil. Parasitol 19:1–16. doi:10.1017/S0331182007003058
Haag J, O’Huigin C, Overath P (1998) The molecular phylogeny of trypanosomes: evidence for an early divergence of the salivaria. Mol Biochem Parasitol 91:37–49. doi:10.1016/S0166-6851(97)00185-0
Harkness LM, Drohan AE, Dickson CM, Smith TG (2010) Experimental transmission of Hepatozoon clamatae (Apicomplexa: Adeleida) to the wood frog, Rana sylvatica, and to the mosquito Culex pipiens. J Parasitol 96(2):434–436. doi:10.1645/GE-2317.1
Hull RW, Camin JH (1960) Haemogregarines in snakes: The incidence of the erythrocytic stages. J Parasitol 46(4):515–523. doi:10.2307/3275151
Jakes K, O’Donoghue PJ, Cameron SL (2003) Phylogenetic relationship of Hepatozoon (Haemogregarina) boigae, Hepatozoon sp., Haemogregarina clelandi and Haemoproteus chelodina form Australian reptilies to other Apicomplexa based on cladistic analyses of ultrastructural and life-cycle characters. Parasitol 126:555–559. doi:10.1017/S0031182003003111
Kim B, Smith TG, Desser SS (1998) The life history and host specificity of Hepatozoon clamatae (Apicomplexa: Adeleorina) and ITS-1 nucleotide sequence variation of Hepatozoon species of frogs and mosquitoes from Ontario. J Parasitol 84:789–797. doi:10.2307/3284589
Leal DDM, O’Dwyer LH, Ribeiro VC, Silva RJ, Ferreira VL, Rodrigues RB (2009) Hemoparasites of the genus Trypanosoma (Kinetoplastida: Trypanosomatidae) and hemogregarines in anurans of the São Paulo and Mato Grosso do Sul States - Brazil. An Acad Bras Cienc 81(2):199–206. doi:10.1590/S0001-37652009000200006
Levine ND, Nye RR (1977) A survey of blood tissue parasites of leopard frogs Rana pipiens in the United States. J Wildl Dis 13:17–23. doi:10.7589/0090-3558-13.1.17
Levine ND (1988) The protozoan phylum Apicomplexa. CRC, Boca Raton, FL
Maia da Silva F, Rodrigues AC, Campaner M, Takata CS, Brigido MC, Junqueira AC, Coura JR, Takeda GF, Shaw JJ, Teixeira MM (2004) Randomly amplified polimorphic DNA analysis of Trypanosoma rangeli and allied species from human, monkeys and other sylvatic mammals of the Brazilian Amazon disclosed a new group and a species-specific marker. Parasitol 128:283–294. doi:10.1017/S003118203004554
Mansour NS, Mohammed AH (1966) Development of Haemogregarina pestanae in the toad Bufo regularis. J Protozool 13(2):265–269. doi:10.1111/j.1550-7408.1966.tb.01905.x
Martin DS, Wright ADG, Barta JR, Desser SS (2002) Phylogenetic position of the giant trypanosomes Trypanosoma chanttoni, Trypanosoma fallisi, Trypanosoma mega, Trypanosoma neveulemairei, and Trypanosoma ranarum inferred from 18S rRNA gene sequences. J Parasitol 88(3):566–571. doi:10.1645/0022-3395(2002)088[0566:PPOTGA]2.0.CO:2
Moço TC, Silva RJ, Madeira NG, Paduan KS, Rubini AS, Leal DDM, O’Dwyer LH (2012) Morphological, morphometric, and molecular characterization of Hepatozoon spp. (Apicomplexa, Hepatozoidae) from naturally infected Caudisona durissa terrifica (Serpentes, Viperidae). Parasitol Res 110:1393–1401. doi:10.1007/s00436-011-2639-2
Mohammed AH, Mansour NS (1966) Development of Haemogregarina boueti in the toad Bufo regularis. J Protozool 13(2):259–264. doi:10.1111/j1550-7408.1966.tb01904.x
O’Dwyer LH, Moço TC, Paduan KS, Spenassatto C, Silva RJ, Ribolla PEM (2013) Description of three new species of Hepatozoon (Apicomplexa, Hepatozoidae) from Rattlesnakes (Crotalus durissus terrificus) based on molecular, morphometric and morphologic characters. Exp Parasitol 135:200–207. doi:10.1016/j.exppara.2013.06.019
Perkins SL, Keller AK (2001) Phylogeny of Nuclear subunit rRNA genes of hemogregarines amplified with specific primers. J Parasitol 87(4):870–876. doi:10.1645/0022-3395(2001)087[0870:PONSSR]2.0.CO:2
Polizer M, Lastória G, Rondon MAC (2000) Características físicas da região do Passo do Lontra. In: I Simpósio Brasileiro de Recursos hídricos do Centro Oeste, 2000, Brasília: Distrito Federal
Rodela LG (2006) Unidades de Vegetação e pastagens nativas do Pantanal da Nhecolândia, Mato Grosso do Sul. 252 p. Tese de Doutorado. Universidade de São Paulo
Rodrigues AC, Paiva F, Campaner M, Stevens JR, Noyes HA, Teixeira MMG (2006) Phylogeny of Trypanosoma (Megatrypanum) theileri and related trypanosomes reveals lineages of isolates associated with artiodactyl hosts diverging on SSU and ITS ribossomal sequences. Parasitol 132:215–224. doi:10.1017/S0031182005008929
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4(4):406–425
Scopel KKG, Fontes CJF, Nunes ÁC, Horta MF, Braga ÉM (2004) Low sensitivity of nested PCR using Plasmodium DNA extracted from stained thick blood smears: an epidemiological retrospective study among subjects with low parasitaemia in an endemic area of the Brazilian Amazon region. Malar J 3:1–6. doi:10.1186/1475-2875-3-8
Schlegel M (1991) Protist evolution and phylogeny as discerned from small subunit ribossomal RNA sequence comparisons. Eur J Protistol 27:207–219. doi:10.1016/S0932-4739(11)80059-3
Sloboda M, Kamler M, Bulantová J, Votýpka J, Modrý DA (2007) A new species of Hepatozoon (Apicomplexa: Adeleorina) from Phyton regius (serpentes: Phytonidae) and its experimental transmission by a mosquito vector. J Parasitol 93(5):1189–1198. doi:10.1645/GE-1200R.1
Smith TG, Desser SS, Martim DD (1994) The development of Hepatozoon sipedon n. sp. (Apicomplexa: Adeleina: Hepatozoidae) in its natural host, the Northern water snake (Nerodia sipedon sipedon), culicine vectors, Culex pipiens and Culex territans, and an intermediate host, the Northern leopard frog (Rana pipiens). Parasitol Res 80(7):559–568. doi:10.1007/BF00933003
Smith TG (1996) The genus Hepatozoon (Apicomplexa: Adeleina). J Parasitol 82(4):565–585. doi:10.2307/3283781
Smith TG, Kim B, Hong H, Desser SS (2000) Intraerytrocytic development of species of Hepatozoon infecting ranid frogs: evidence for convergence of life cycle characteristics among apicomplexans. J Parasitol 86(3):451–458. doi:10.2307/3284856
Soriano BMA, Alves MJM (2005) Boletim agrometeorológico ano 2002 para a sub-região da Nhecolãndia, Pantanal, Mato Grosso do Sul. EMBRAPA Pantanal, Corumbá, Brasil
Sykes JE, Owens SD, Terry JC, Lindsay LL, Pusterla N (2008) Use of dried blood smears for detection of feline hemoplasmas using real-time polymerase chain reaction. J Vet Diagn Investig 20(5):616–620. doi:10.1177/1040638708020000513
Uetanabaro M, Almeida Prado CP, Rodrigues DJ, Gordo M, Campos Z (2008) Field guide to the anurans of the Pantanal and surrounding Cerrados. Universidade do Mato Grosso do Sul, Brasil
Viana LA, Soares P, Silva JE, Paiva F, Coutinho ME (2012) Anurans as paratenic host in the transmission of Hepatozoon caimani to caimans Caiman yacare and Caiman latirostris. Parasitol Res 110(2):883–886. doi:10.1007/s00436-011-2570-6
Acknowledgments
This study received financial support from Coordination for the improvement of Higher Education Personnel (CAPES) and Foundation for UNESP Development, Process No. 00237/10-DFP (FUNDUNESP). We gratefully acknowledge Dr. Vanda Lucia Ferreira, Associate Professor of the Biology Department, Biological Sciences and Health Center of Mato Grosso do Sul Federal University, for valuable technical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Menezes Leal, D.D., Dreyer, C.S., da Silva, R.J. et al. Characterization of Hepatozoon spp. in Leptodactylus chaquensis and Leptodactylus podicipinus from two regions of the Pantanal, state of Mato Grosso do Sul, Brazil. Parasitol Res 114, 1541–1549 (2015). https://doi.org/10.1007/s00436-015-4338-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-015-4338-x