Abstract
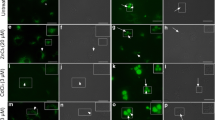
This study aimed to explore Toxoplasma gondii nucleus coding apicoplast protein acyl carrier protein (ACP) synthesis and trafficking in delayed death. The recombinant T. gondii ACP was expressed by prokaryotic expression method, and anti-ACP polyclonal antibody was obtained from rabbit immune. T. gondii “delayed death” was induced by clindamycin (CLDM), and ACP transcription was determined by real-time PCR assay. The expression of ACP with transit type (t-ACP) and mature type (m-ACP) was determined by Western blotting with anti-ACP polyclonal antibody. The mutant-expressed ACP fused with green fluorescent protein (GFP) tag was constructed by pHX-ACP-GFP. The distribution of ACP in “delayed death” was observed by ACP-GFP fusion protein with a confocal microscope. T. gondii ACP transcription and t-ACP expression had no significant decrease in the early 4 h of “delayed death,” but there has been a significant decrease in 6 h. The expression of m-ACP had a significant decrease in 4 h which occurred earlier than the t-ACP expression. The number of brightly dot green fluorescence in ACP-GFP mutant decreased with prolonged time. There was very little brightly dot green fluorescence in ACP-GFP mutant when treated with CLDM for 6 h. CLDM could suppress apicoplast proliferation and induce T. gondii “delayed death”; however, it could not directly suppress nucleus coding ACP transcription and expression. T. gondii lacking of apicoplast had a barrier of transit peptide cleavage and t-ACP could not be transformed into m-ACP. The reason for the decrease in ACP expression could be due to excessive t-ACP synthesis in tachyzoites resulting in a negative feedback for the ACP coding gene transcription.






Similar content being viewed by others
References
Botté CY, Dubar F, McFadden GI, Maréchal E, Biot C (2012) Plasmodium falciparum apicoplast drugs: targets or off-targets? Chem Rev 112(3):1269–1283. doi:10.1021/ cr200258w
Choi SR, Mukherjee P, Avery MA (2008) The fight against drug-resistant malaria: novel plasmodial targets and antimalarial drugs. Curr Med Chem 15(2):161–171
Coppens I, Andries M, Liu JL, Cesbron-Delauw MF (1999) Intracellular trafficking of dense granule proteins in Toxoplasma gondii and experimental evidences for a regulated exocytosis. Eur J Cell Biol 78(7):463–472
Crawford MJ, Thomsen-Zieger N, Ray M, Schachtner J, Roos DS, Seeber F (2006) Toxoplasma gondii scavenges host-derived lipoic acid despite its de novo synthesis in the apicoplast. Embo J 25(13):3214–3222
Dahl EL, Rosenthal PJ (2008) Apicoplast translation, transcription and genome replication: targets for antimalarial antibiotics. Trends Parasitol 24(6):279–284. doi:10.1016/j. pt.2008. 03.007
Dautu G, Ueno A, Munyaka B, Carmen G, Makino S, Kobayashi Y, Igarashi M (2008) Molecular and biochemical characterization of Toxoplasma gondii beta-hydroxyacyl-acyl carrier protein dehydratase (FABZ). Parasitol Res 102(6):1301–1309. doi:10.1007/ s00 436-008-0909-4
DeRocher A, Hagen CB, Froehlich JE, Feagin JE, Parsons M (2000) Analysis of targeting sequences demonstrates that trafficking to the Toxoplasma gondii plastid branches off the secretory system. J Cell Sci 113(Pt22):3969–3977
DeRocher A, Gilbert B, Feagin JE, Parsons M (2005) Dissection of brefeldin A-sensitive and-insensitive steps in apicoplast protein targeting. J Cell Sci 118(Pt3):565–574
Fichera ME, Roos DS (1997) A plastid organelle as a drug target in apicomplexan parasites. Nature 390:407–409
Fichera ME, Bhopale MK, Roos DS (1995) In vitro assays elucidate peculiar kinetics of clindamycin action against Toxoplasma gondii. Antimicrob Agents Chemother 39(7):1530–1537
Fleige T, Limenitakis J, Soldati-Favre D (2010) Apicoplast: keep it or leave it. Microbes Infect 12(4):253–262
Foth BJ, Ralph SA, Tonkin CJ, Struck NS, Fraunholz M, Roos DS, Cowman AF, McFadden GI (2003) Dissecting apicoplast targeting in the malaria parasite Plasmodium falciparum. Science 299:705–708
Harb OS, Chatterjee B, Fraunholz MJ, Crawford MJ, Nishi M, Roos DS (2004) Multiple functionally redundant signals mediate targeting to the apicoplast in the apicomplexan parasite Toxoplasma gondii. Eukaryot Cell 3(3):663–674
Kim K, Soldati D, Boothroyd JC (1993) Gene replacement in Toxoplasma gondii with chloramphenicol acetyltransferase as selectable marker. Science 262:911–914
Kohler S, Delwiche CF, Denny PW, Tilney LG, Webster P, Wilson RJ, Palmer JD, Roos DS (1997) A plastid of probable green algal origin in apicomplexan parasites. Science 275:1485–1489
Mazumdar J, Wilson HE, Masek KA, Hunter C, Striepen B (2006) Apicoplast fatty acid synthesis is essential for organelle biogenesis and parasite survival in Toxoplasma gondii. Proc Natl Acad Sci U S A 103(35):13192–13197
Parsons M, Karnataki A, Derocher AE (2009) Evolving insights into protein trafficking to the multiple compartments of the apicomplexan plastid. J Eukaryot Microbiol 56(3):214–220. doi:10.1111/j.1550-7408.2009.00405.x
Pfluger SL, Goodson HV, Moran JM, Ruggiero CJ, Ye X, Emmons KM, Hager KM (2005) Receptor for retrograde transport in the apicomplexan parasite Toxoplasma gondii. Eukaryot Cell 4(2):432–442
Ridley RG (2002) Medical need, scientific opportunity and the drive for antimalarial drugs. Nature 415:686–693
Soldati D, Boothroyd JC (1993) Transient transfection and expression in the obligate intracellular parasite Toxoplasma gondii. Science 260:349–352
Striepen B, He CY, Matrajt M, Soldati D, Roos DS (1998) Expression, selection, and organellar targeting of the green fluorescent protein in Toxoplasma gondii. Mol Biochem Parasitol 92(2):325–338
Waller RF, McFadden GI (2005) The apicoplast: a review of the derived plastid of apicomplexan parasites. Curr Issues Mol Biol 7(1):57–79
Wiesner J, Reichenberg A, Heinrich S, Schlitzer M, Jomaa H (2008) The plastid-like organelle of apicomplexan parasites as drug target. Curr Pharm Des 14(9):855–871
Acknowledgments
We thank Professor Dominique Soldati-Favre of the Department of Microbiology and Molecular Medicine, Faculty of Medicine, University of Geneva and Professor Marilyn Parsons and Amy DeRocher for the kind gift of experimental material and direction. This study received financial support from the National Natural Science Foundation of China (grant number 81301453), the Laboratory of Parasite and Vector Biology of China, MOPH (grant number WSBKTKT201302), the China Postdoctoral Science Foundation (grant number 2014 M561598), Jiangsu Postdoctoral Science Foundation (grant number 1402171C), the Senior Talent Studying Initial Funding of Jiangsu University (grant number 13JDG023, 13JDG127), and the Graduate Student Creative project of Jiangsu Province (grant number CX10B_282Z).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wu, L., Shen, J., Zhou, Y. et al. Toxoplasma gondii nucleus coding apicoplast protein ACP synthesis and trafficking in delayed death. Parasitol Res 114, 1099–1105 (2015). https://doi.org/10.1007/s00436-014-4281-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-014-4281-2