Abstract
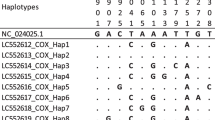
As the most common cause of the human microsporidiosis, Enterocytozoon bieneusi has been found in a wide variety of animal hosts. Deers are the ruminant mammals living in a variety of biomes, and the distribution of deer species differ by geography. To understand the prevalence of natural infection of E. bieneusi in deer and to assess their epidemiological role in the transmission of microsporidiosis caused by E. bieneusi, 91 fecal specimens were collected from 86 sika deers and five red deers in the northeast of China. By PCR and sequencing of the internal transcribed spacer (ITS) region of the ribosomal RNA (rRNA) gene of E. bieneusi, an average infection rate of 31.9 % (29/91) was observed in deer, with 32.6 % (28/86) for sika deer, and 20 % (1/5) for red deer. Six ITS genotypes were identified: one known genotype BEB6 (n = 20) and five novel genotypes HLJD-I to HLJD-IV (one each) and HLJD-V (n = 5). A phylogenetic analysis based on a neighbor-joining tree of the ITS gene sequences of E. bieneusi indicated that genotypes HLJD-II and HLJD-III fell into group 1 of zoonotic potential, while the other genotypes (BEB6, HLJD-I, HLJD-IV, HLJD-V) were clustered into so-called bovine-specific group 2. This is the first report of E. bieneusi in deer in China. The observation of genotype BEB6 in humans previously and in deer here and also the findings of the two novel genotypes (HLJD-II to HLJ-III) belonging to potential zoonotic group 1 suggested the possibility of deer in the transmission of E. bieneusi to humans.


Similar content being viewed by others
References
Akinbo FO, Okaka CE, Omoregie R, Dearen T, Leon ET, Xiao L (2012) Molecular epidemiologic characterization of Enterocytozoon bieneusi in HIV-infected persons in Benin City, Nigeria. Am J Trop Med Hyg 86:441–445
Akinbo FO, Okaka CE, Omoregie R, Adamu H, Xiao L (2013) Unusual Enterocytozoon bieneusi genotypes and Cryptosporidium hominis subtypes in HIV-infected patients on highly active antiretroviral therapy. Am J Trop Med Hyg 89:157–161
Buckholt MA, Lee JH, Tzipori S (2002) Prevalence of Enterocytozoon bieneusi in swine: an 18-month survey at a slaughterhouse in Massachusetts. Appl Environ Microbiol 68:2595–2599
Decraene V, Lebbad M, Botero-Kleiven S, Gustavsson AM, Löfdahl M (1985) Occurrence of a new microsporidan: Enterocytozoon bieneusi n.g., n. sp., in the enterocytes of a human patient with AIDS. J Protozool 32:250–254
Dengjel B, Zahler M, Hermanns W, Heinritzi K, Spillmann T, Thomschke A, Löscher T, Gothe R, Rinder H (2001) Zoonotic potential of Enterocytozoon bieneusi. J Clin Microbiol 39:4495–4499
Deplazes P, Mathis A, Muler C, Webe R (1996) Molecular epidemiology of Encephalitozoon cuniculi and firsts detection of Entercytozoon bieneusi in faecal samples from pigs. J Eukaryot Microbiol 43:93S
Didier ES, Weiss LM (2006) Microsporidiosis: current status. Curr Opin Infect Dis 19:485–492
Fayer R, Santin M, Trout JM (2007) Enterocytozoon bieneusi in mature dairy cattle on farms in the eastern United States. Parasitol Res 102:15–20
Galván-Díaz AL, Magnet A, Fenoy S, Henriques-Gil N, Haro M, Gordo FP, Miró G, Aguila CD, Izquierdo F (2014) Microsporidia detection and genotyping study of human pathogenic E. bieneusi in animals from Spain. PLoS One 9:e92289
Ghosh K, Weiss LM (2009) Molecular diagnostic tests for microsporidia. Interdis Perspect Infect Dis 2009:926521
Guo Y, Alderisio KA, Yang W, Cama V, Feng Y, Xiao L (2014) Host specificity and source of Enterocytozoon bieneusi genotypes in a drinking source watershed. Appl Environ Microbiol 80:218–225
Henriques-Gil N, Haro M, Izquierdo F, Fenoy S, del Aguila C (2010) Phylogenetic approach to the variability of the microsporidian Enterocytozoon bieneusi and its implications for inter- and intrahost transmission. Appl Environ Microbiol 76:3333–3342
Karim MR, Wang R, Dong H, Zhang L, Li J, Zhang S, Rume FI, Qi M, Jian F, Sun M, Yang G, Zou F, Ning C, Xiao L (2014a) Genetic polymorphism and zoonotic potential of Enterocytozoon bieneusi from nonhuman primates in China. Appl Environ Microbiol 80:1893–1898
Karim MR, Yu F, Li J, Li J, Zhang L, Wang R, Rume FI, Jian F, Zhang S, Ning C (2014b) First molecular characterization of enteric protozoa and the human pathogenic microsporidian,Enterocytozoon bieneusi, in captive snakes in China. Parasitol Res 2014 Jun 7. [Epub ahead of print]
Keeling P (2009) Five questions about microsporidia. PLoS Pathog 5:e1000489
Lee JH (2007) Prevalence and molecular characteristics of Enterocytozoon bieneusi in cattle in Korea. Parasitol Res 101:391–396
Li N, Xiao L, Wang L, Zhao S, Zhao X, Duan L, Guo M, Liu L, Feng Y (2012a) Molecular surveillance of Cryptosporidium spp., Giardia duodenalis, and Enterocytozoon bieneusi by genotyping and subtyping parasites in wastewater. PLoS Negl Trop Dis 6:e1809
Li W, Cama V, Feng Y, Gilman RH, Bern C, Zhang X, Xiao L (2012b) Population genetic analysis of Enterocytozoon bieneusi in humans. Int J Parasitol 42:287–293
Li W, Li Y, Li W, Yang J, Song M, Diao R, Jia H, Yixin L, Zheng J, Zhang X, Xiao L (2014) Genotypes of Enterocytozoon bieneusi in livestock in China: high prevalence and zoonotic potential. PLoS One 9:e97623
Matos O, Lobo ML, Xiao L (2012) Epidemiology of Enterocytozoon bieneusi infection in humans. J Parasitol Res 2012:981424
Pirestani M, Sadraei J, Forouzandeh M (2013) Molecular characterization and genotyping of human related microsporidia in free-ranging and captive pigeons of Tehran, Iran. Infect Genet Evol 20:495–499
Reetz J, Rinder H, Thomschke A, Manke H, Schwebs M, Bruderek A (2002) First detection of the microsporidium Enterocytozoon bieneusi in non-mammalian hosts (chickens). Int J Parasitol 32:785–787
Santin M, Fayer R (2009a) Enterocytozoon bieneusi genotype nomenclature based on the internal transcribed spacer sequence: a consensus. J Eukaryot Microbiol 56:34–38
Santin M, Fayer R (2009b) A longitudinal study of Enterocytozoon bieneusi in dairy cattle. Parasitol Res 105:141–144
Santin M, Fayer R (2011) Microsporidiosis: Enterocytozoon bieneusi in domesticated and wild animals. Res Vet Sci 90:363–371
Santin M, Trout JM, Fayer R (2005) Enterocytozoon bieneusi genotypes in dairy cattle in the eastern United States. Parasitol Res 97:535–538
Santin M, Dargatz D, Fayer R (2012) Prevalence and genotypes of Enterocytozoon bieneusi in weaned beef calves on cow-calf operations in the USA. Parasitol Res 110:2033–2041
Thellier M, Breton J (2008) Enterocytozoon bieneusi in human and animals, focus on laboratory identification and molecular epidemiology. Parasite 15:349–358
Wang L, Xiao L, Duan L, Ye J, Guo Y, Guo M, Liu L, Feng Y (2013) Concurrent infections of Giardia duodenalis, Enterocytozoon bieneusi, and Clostridium difficile in children during a cryptosporidiosis outbreak in a pediatric hospital in China. PLoS Negl Trop Dis 7:e2437
Widmer G, Akiyoshi DE (2010) Host-specific segregation of ribosomal nucleotide sequence diversity in the microsporidian Enterocytozoon bieneusi. Infect Genet Evol 10:122–128
Zhang X, Wang Z, Su Y, Liang X, Sun X, Peng S, Lu H, Jiang N, Yin J, Xiang M, Chen Q (2011) Identification and genotyping of Enterocytozoon bieneusi in China. J Clin Microbiol 49:2006–2008
Zhao W, Zhang W, Yang F, Cao J, Liu H, Yang D, Shen Y, Liu A (2014) High prevalence of Enterocytozoon bieneusi in asymptomatic pigs and assessment of zoonotic risk at a genotype level. Appl Environ Microbiol 80:3699–3707
Acknowledgments
The study was supported partially by the State Key Program of the National Natural Science Foundation of China (grant no. 31330079) (Longxian Zhang) and the Heilongjiang Province Education Bureau of No. 12531266 (Aiqin Liu). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Wei Zhao and Weizhe Zhang contributed equally to this work.
Rights and permissions
About this article
Cite this article
Zhao, W., Zhang, W., Wang, R. et al. Enterocytozoon bieneusi in sika deer (Cervus nippon) and red deer (Cervus elaphus): deer specificity and zoonotic potential of ITS genotypes. Parasitol Res 113, 4243–4250 (2014). https://doi.org/10.1007/s00436-014-4100-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-014-4100-9