Abstract
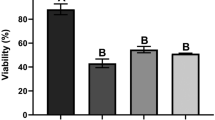
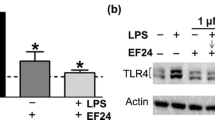
Sj16, a 16-kDa protein secreted from Schistosoma japonicum, has been demonstrated an anti-inflammatory effect in vitro and in vivo, but its mechanism is still not clear. In this study, microarray analysis was performed to investigate the effects of recombinant Sj16 (rSj16) on the gene expression of the lipopolysaccharide (LPS)-stimulated dendritic cells (DCs). Immature DCs were treated with LPS, LPS + recombinant Sj16 (rSj16), or rSj16 alone for 24 h, and the gene expression profiles were examined using complementary DNA (cDNA) microarrays. With the cutoff value of 2-fold change in the expression, 509 genes were affected, 226 genes upregulated, and 283 genes downregulated after adding rSj16. Analysis by functional annotation clustering tool showed that rSj16-affected genes mainly associated with inflammatory response, defense response, regulation of immune system process, apoptosis, and cell migration. The results revealed that rSj16 reduced the LPS-induced pro-inflammatory genes such as cytokines (e.g., IL6, IL18, IFN-γ, IL12a, IL1b), chemokines, and receptors (e.g., CXCL1, CXCL9, CCL5, CCR5, CCR1, CCR2, CXCR3) and increased the anti-inflammatory gene IL-10. Further data mining of these genes by pathway analysis showed that genes regulated by rSj16 were significantly involved in cytokine-cytokine receptor interaction, NOD-like receptor signaling pathway, Toll-like receptor signaling pathway, antigen processing and presentation, and Jak-STAT signaling pathway. In addition, quantitative real-time PCR (qRT-PCR) and Western blot analysis showed that rSj16 downregulated the expression of inhibitor of nuclear factor kappa-β kinase subunit beta (IKKβ) and nuclear factor-kappa β p65 (NF-κβ) messenger RNA (mRNA) and inhibited the phosphorylation of IKKβ and the NF-κB p65 protein, which implied that rSj16 exerting immunomodulatory effects by suppressing NF-κB signaling pathway. These results provide useful information in further understanding of the immunoregulation mechanisms of Sj16, and it is indicated that Sj16 could be as a potential molecule for the immunosuppressant drug development.




Similar content being viewed by others
References
Ahmad G et al (2009) Prime-boost and recombinant protein vaccination strategies using Sm-p80 protects against Schistosoma mansoni infection in the mouse model to levels previously attainable only by the irradiated cercarial vaccine. Parasitol Res 105(6):1767–1777. doi:10.1007/s00436-009-1646-z
Ben-Baruch A (2006) Inflammation-associated immune suppression in cancer: the roles played by cytokines, chemokines and additional mediators. Semin Cancer Biol 16(1):38–52. doi:10.1016/j.semcancer.2005.07.006
Faria MS, Reis FC, Lima AP (2012) Toll-like receptors in leishmania infections: guardians or promoters? J Parasitol Res 2012:930257 doi:10.1155/2012/930257
Franchi L et al (2008) Intracellular NOD-like receptors in innate immunity, infection and disease. Cell Microbiol 10(1):1–8. doi:10.1111/j.1462-5822.2007.01059.x
Gomez-Garcia L, Rivera-Montoya I, Rodriguez-Sosa M, Terrazas LI (2006) Carbohydrate components of Taenia crassiceps metacestodes display Th2-adjuvant and anti-inflammatory properties when co-injected with bystander antigen. Parasitol Res 99(4):440–448. doi:10.1007/s00436-006-0159-2
Green MJ et al (2003) Serum MMP-3 and MMP-1 and progression of joint damage in early rheumatoid arthritis. Rheumatology 42(1):83–88. doi:10.1093/rheumatology/keg037
Hu S, Wu Z, Yang L, Fung MC (2009) Molecular cloning and expression of a functional anti-inflammatory protein, Sj16, of Schistosoma japonicum. Int J Parasitol 39(2):191–200. doi:10.1016/j.ijpara.2008.06.017
Hu S, Yang L, Wu Z, Wong CS, Fung MC (2012) Suppression of adaptive immunity to heterologous antigens by SJ16 of Schistosoma japonicum. J Parasitol 98(2):274–283. doi:10.1645/GE-2692.1
Jana Parohova SV, Barta A, Kovacsova M, Bartko D, Pechanova O (2009) The cross-talk of nuclear factor kappaB and nitric oxide in the brain. Activitas Nervosa Superior Rediviva 51:3–4
Kim JM, Oh YK, Kim YJ, Cho SJ, Ahn MH, Cho YJ (2001) Nuclear factor-kappa B plays a major role in the regulation of chemokine expression of HeLa cells in response to Toxoplasma gondii infection. Parasitol Res 87(9):758–763
Lee HH, Chou HL, Chen KM, Lai SC (2004) Association of matrix metalloproteinase-9 in eosinophilic meningitis of BALB/c mice caused by Angiostrongylus cantonensis. Parasitol Res 94(5):321–328. doi:10.1007/s00436-004-1196-3
Li ZWCW, Hu Y, Delhase M, Deerinck T, Ellisman M, Johnson R, Karin M (1999) The IKKβ subunit of IκB kinase (IKK) is essential for nuclear factor κB activation and prevention of apoptosis. J Exp Med 189(11):1839–1845
Liapis CD, Paraskevas KI (2003) The pivotal role of matrix metalloproteinases in the development of human abdominal aortic aneurysms. Vasc Med 8(4):267–271. doi:10.1191/1358863x03vm504ra
Luboshits G et al (1999) Elevated expression of the CC chemokine regulated on activation, normal T cell expressed and secreted (RANTES) in advanced breast carcinoma. Cancer Res 59(18):4681–4687
Maizels RM, Yazdanbakhsh M (2003) Immune regulation by helminth parasites: cellular and molecular mechanisms. Nat Rev Immunol 3(9):733–744. doi:10.1038/nri1183
Manicourt DH, Fujimoto N, Obata K, Thonar EJ (1995) Levels of circulating collagenase, stromelysin-1, and tissue inhibitor of matrix metalloproteinases 1 in patients with rheumatoid arthritis. Arthritis Rheum 38(8):1031–1039
McCormack G, Moriarty D, O'Donoghue DP, McCormick PA, Sheahan K, Baird AW (2001) Tissue cytokine and chemokine expression in inflammatory bowel disease. Inflamm Res 50(10):491–495
Nagase H, Visse R, Murphy G (2006) Structure and function of matrix metalloproteinases and TIMPs. Cardiovasc Res 69(3):562–573. doi:10.1016/j.cardiores.2005.12.002
Niwa Y, Akamatsu H, Niwa H, Sumi H, Ozaki Y, Abe A (2001) Correlation of tissue and plasma RANTES levels with disease course in patients with breast or cervical cancer. Clin Cancer Res 7(2):285–289
Ouyang WJ, Rutz S, Crellin NK, Valdez PA, Hymowitz SG (2011) Regulation and functions of the IL-10 family of cytokines in inflammation and disease. Annu Rev Immunol 29:71–109. doi:10.1146/annurev-immunol-031210-101312
Rodrigues MM, Oliveira AC, Bellio M (2012) The immune response to Trypanosoma cruzi: Role of Toll-like receptors and perspectives for vaccine development. J Parasitol Res 2012:507874. doi:10.1155/2012/507874
Sun X et al (2010) rSj16, a recombinant protein of Schistosoma japonicum-derived molecule, reduces severity of the complete Freund's adjuvant-induced adjuvant arthritis in rats' model. Parasite Immunol 32(11–12):739–748. doi:10.1111/j.1365-3024.2010.01240.x
Sun R et al (2012a) Molecular cloning and characterization of a matrix metalloproteinase, from Caenorhabditis elegans: employed to identify homologous protein from Angiostrongylus cantonensis. Parasitol Res 110(5):2001–2012. doi:10.1007/s00436-011-2729-1
Sun X et al (2012b) Effects of a recombinant schistosomal-derived anti-inflammatory molecular (rSj16) on the lipopolysaccharide (LPS)-induced activated RAW264.7. Parasitol Res 110(6):2429–2437. doi:10.1007/s00436-011-2782-9
Sun XJ et al (2012c) Unique roles of Schistosoma japonicum protein Sj16 to induce IFN-gamma and IL-10 producing CD4(+)CD25(+) regulatory T cells in vitro and in vivo. Parasite Immunol 34(8–9):430–439. doi:10.1111/j.1365-3024.2012.01377.x
Talmor M, Mirza A, Turley S, Mellman I, Hoffman LA, Steinman RM (1998) Generation or large numbers of immature and mature dendritic cells from rat bone marrow cultures. Eur J Immunol 28(3):811–817. doi:10.1002/(SICI)1521-4141(199803)28:03<811::AID-IMMU811>3.0.CO;2-S
Tang CL et al (2011) Effect of CD4+ CD25+ regulatory T cells on the immune evasion of Schistosoma japonicum. Parasitol Res 108(2):477–480. doi:10.1007/s00436-010-2089-2
Thompson RW, Parks WC (1996) Role of matrix metalloproteinases in abdominal aortic aneurysms. Ann N Y Acad Sci 800:157–174
Wang SW et al (2012) CCL5 and CCR5 interaction promotes cell motility in human osteosarcoma. PLoS One 7(4):e35101. doi:10.1371/journal.pone.0035101
Yaal-Hahoshen N et al (2006) The chemokine CCL5 as a potential prognostic factor predicting disease progression in stage II breast cancer patients. Clin Cancer Res 12(15):4474–4480. doi:10.1158/1078-0432.Ccr-06-0074
Yang F et al (2013) A recombined protein (rSj16) derived from Schistosoma japonicum induces cell cycle arrest and apoptosis of murine myeloid leukemia cells. Parasitol Res 112(3):1261–1272. doi:10.1007/s00436-012-3260-8
Acknowledgments
This work was supported by grants from the National Natural Science Foundation of China (no. 30972574 and 81201309) and a grant from the China Postdoctoral Science Foundation (no. 2011 M501363), the Natural Science Foundation of Guangdong Province, China (no. 3S2012040007256), and the Foundation of Doctoral Program of Higher Education of China (no. 20120171120049).
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Table S1
Changes in gene expression of the 35 genes common among three group treatments (LPS, rSj16 + LPS, and rSj16) (DOCX 24 kb)
Table S2
Gene expression profiles that were upregulated when cells that were treated with rSj16 + LPS were compared with those that were treated with LPS alone for 24 h (XLSX 45 kb)
Table S3
Gene expression profiles that were reversed when cells that were treated with rSj16 + LPS were compared with those that were treated with LPS alone for 24 h (XLSX 43 kb)
Table S4
Functional categorization of downregulated genes induced by rSj16 + LPS versus LPS treatment (P < 0.05) (XLSX 21 kb)
Table S5
Functional categorization of upregulated genes induced by rSj16 + LPS versus LPS treatment (P < 0.05) (XLSX 16 kb)
Rights and permissions
About this article
Cite this article
Shen, J., Xu, L., Liu, Z. et al. Gene expression profile of LPS-stimulated dendritic cells induced by a recombinant Sj16 (rSj16) derived from Schistosoma japonicum . Parasitol Res 113, 3073–3083 (2014). https://doi.org/10.1007/s00436-014-3973-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-014-3973-y