Abstract
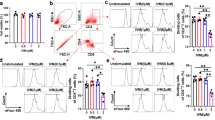
The immune responses of Angiostrongylus cantonensis infection are closely relevant to the host’s self-protection and the nematode’s pathogenesis. In the present study, BALB/c mice were randomly divided into uninfected control group, infection group 1, and infection group 2. The infection group 1 and infection group 2 were infected with 20 and 40 third-stage larvae of A. cantonensis per mouse, respectively. The splenocytes from the mice were collected and cultured on the 19th and 25th days post-infection; the subtypes of T cells in splenocytes were detected by flow cytometry with fluorescence staining method, and the cytokines in cultured supernatants of splenocytes were assayed by the method of ELISA. The specific IgG and IgE antibodies in sera of the mice were periodically detected by ELISA. The results showed that the percentages of CD4+ and CD4+ IL-4+ T cells in splenocytes of infected mice were much higher (P < 0.05) than those in control mice; however, the percentages of CD4+ IL-17+ and CD4+ IFN-γ+ T cell were much lower(P < 0.01) after the infection. The levels of CD8+ T cells in infected mice also rose, but differences between control mice and infected mice were not significant. In comparison with control mice, the concentration of IL-4 in the cultured supernatants of splenocytes in infected mice increased significantly (P < 0.05), but that of IL-17 decreased significantly (P < 0.01). In addition, the number of larvae infected and days after infection may influence levels of the T cell subtypes and the cytokines in spleen, too (P > 0.05). On humoral immunity, the levels of specific IgG antibodies in sera rose a bit at the fifth day post-infection, and reached a peak at the 20th day post-infection; the specific IgE antibodies gradually heightened during first 10 days post-infection; then, it showed a downward trend during the 15th to 25th days post-infection. It is evident that the percentages of CD4+ T lymphocytes of spleen in the mice infected with A. cantonensis markedly increase and polarize to Th2 phenotypes, and the function of Th17 cells is inhibited. In addition, the elevation of specific IgG antibodies in sera of the infected mice is more significant than that of specific IgE antibodies.






Similar content being viewed by others
References
Abbas AK, Murphy KM, Sher A (1996) Functional diversity of helper T lymphocytes. Nature 383(6603):787–793
Anukumar B, Shahir P (2011) Immune regulation in Chandipura virus infection: characterization of CD4+ T regulatory cells from infected mice. Virol J 8(259):1–11
Aoki M, Sugaya H, Ishida K et al (1998) The role of CD4+ and CD8+ T-cells in host morbidity and innate resistance to Angiostrongylus cantonensis in the mouse. Parasitol Res 84(2):91–99
Chen DX, Zhang Y, Shen HX et al (2011) Epidemiological survey of Angiostrongylus cantonensis in the west-central region of Guangdong Province, China. Parasitol Res 109:305–314
Chen P, Wang WL (2009) Th1/Th2 Cytokines and worm infection. J of Pathog Biol 4(4):318–320
Chen ZL (1998) Parasite infection and cytokine. Foreign Med Sci Parasit Dis 25(5):223–227
Dorta-Contreras AJ, Noris-Garcia E, Escobar-Perez X et al (2005) IgG1, IgG2 and IgE intrathecal synthesis in Angiostrongylus cantonensis meningoencephalitis. Neurol Sci 238(1–2):65–70
Dunne DW, Butterworth AE, Fulford AJ et al (1992) Human IgE responses to Schistosoma mansoni and resistance to reinfection. Mem Inst Oswaldo Cruz 87:99–103
Gounni AS, Lamkhioued B, Ochiai K et al (1994) High-affinity IgE receptor on eosinophils is involved in defence against parasites. Nature 367(6459):183–186
Gurish MF, Bryce PJ, Tao H et al (2004) IgE enhances parasite clearance and regulates mast cell responses in mice infected with Trichinella spiralis. J Immunol 172(2):1139–1145
Harrington LE, Hatton RD, Mangan PR et al (2005) Interleukin 17-producing CD4+ effector T cells develop via a lineage distinct from the T helper type 1 and 2 lineages. Nat Immunol 6(11):1123–1132
Huang XH, Yang FZ, Zhang YZ et al (2001) Determination of antibodies in sera of rats infected with Angiostrongylus cantonensis. Strait J Prev Med 7(2):41–42
Hüttemann M, Schmahl G, Mehlhorn H (2007) Light and electron microscopic studies on two nematodes, Angiostrongylus cantonensis and Trichuris muris, differing in their mode of nutrition. Parasitol Res 101(Suppl 2):S225–S232
Jin H, He X, Liu DX et al (2006) Dynamic observation on the serum IgG level in rats infected with Angiostrongylus cantonensis. China Trop Med 6(1):21–22
Joseph M, Auriault C, Capron A et al (1983) A new function for platelets: IgE dependent killing of schistosomes. Nature 303(5920):810–812
Kolls JK, Linden A (2004) Interleukin-17 family members and inflammation. Immunity 21(4):467–476
Ko RC, Chan SW, Chan KW et al (1987) Four documented cases of eosinophilic meningoencephalitis due to Angiostrongylus cantonensis in Hong Kong. Trans R Soc Trop Med Hyg 81(5):807–810
Kushawaha PK, Gupta R, Sundar S et al (2011) Elongation factor-2, a Th1 stimulatory protein of Leishmania donovani, generates strong IFN-γ and IL-12 response in cured Leishmania-infected patients/hamsters and protects hamsters against Leishmania challenge. J Immunol 187(12):6417–6427
Langermans AM, Van der Hulst ME, Nibbering PH et al (1992) IFN-gamma-induced L-arginine-dependent toxoplasmastatic activity in murine peritoneal macrophages is mediated by endogenous tumor necrosis factor-alpha. J Immunol 148(2):568–574
Lee JD, Wang JJ, Chang JH et al (1996) Role of T cell subpopulations in mice infected with Angiostrongylus cantonensis. J Helminthol 70(3):211–214
Lindo JF, Waugh C, Hall J et al (2002) Enzootic Angiostrongylus cantonensis in rats and snails after an outbreak of human eosinophilic meningitis, Jamaica. Emerg Infect Dis 8(3):324–326
Mosmann TR, Cherwinski H, Bond MW et al (1986) Two types of murine helper T cell clone I. Definition according to profiles of lymphokine activities and secreted proteins. J Immunol 136(7):2348–2357
OuYang L, Wei J, Wu ZD et al (2012) Differences of larval development and pathological changes in permissive and nonpermissive rodent hosts for Angiostrongylus cantonensis infection. Parasitol Res 111(4):1547–1557
Padilla-Docal B, Dorta-Contreras AJ, Bu-Coifiu-Fanego R et al (2008) Intrathecal synthesis of IgE in children with eosinophilic meningoencephalitis caused by Angiostrongylus cantonensis. Cerebrospinal Fluid Res 5:18
Pan CW, Ling HB, Liang SH et al (2000) Detection of antibodies in rats infected with Angiostrongylus cantonensis by ELISA. Chin J Zoonoses 16(1):79–80
Perez O, Capron M, Lastre M et al (1989) Angiostrongylus cantonensis: role of eosinophils in the neurotoxic syndrome (Gordon-like phenomenon). Exp Parasitol 68(4):403–413
Pinot de Moira A, Fulford AJ, Kabatereine NB et al (2010) Analysis of complex patterns of human exposure and immunity to Schistosomiasis mansoni: the influence of age, sex, ethnicity and IgE. PLoS Negl Trop Dis 4(9):1–11
Qu ZY, Zhan XM, He A (2007) The analysis on epidemic situation of angiostrongyliasis in China. Int J Med Parasit Dis 34(5):279–283
Slom TJ, Cortese MM, Gerber SI et al (2002) An outbreak of eosinophilic meningitis caused by Angiostrongylus cantonensis in travelers returning from the Caribbean. N Engl J Med 346:668–675
Sugaya H, Aoki M, Abe T et al (1997) Cytokine responses in mice infected with Angiostrongylus cantonensis. Parasitol Res 83(1):10–15
Su Z, Stevenson MM (2000) Central role of endogenous gamma interferon in protective immunity against blood-stage Plasmodium chabaudi AS infection. Infect Immun 68(8):4399–4406
Wang QP, Lai DH, Zhu XQ et al (2008) Human angiostrongyliasis. Lancet Infect Dis 8(10):621–630
Watanabe N, Bruschi F, Korenaga M (2005) IgE: a question of protective immunity in Trichinella spiralis infection. Trends Parasito 21(4):175–178
Wei J, Wu F, Sun X, Zeng X, Liang JY, Zheng HQ, Yu XB, Zhang KX, Wu ZD (2012) Differences in microglia activation between rats-derived cell and mice-derived cell after stimulating by soluble antigen of IV larva from Angiostrongylus cantonensis in vitro. Parasitol Res [Epub ahead of print]
Yang BY, Shen EX, Wu CY (2006) Phenotype of mouse CD4+CD25+ regulatory T cells in mouse splenocytes. Curr Immunol 26(3):208–212
Acknowledgments
This work was supported by the grants from the National Natural Science Foundation of P.R. China (30972577) and Natural Science Foundation of Guangdong Province (s2011040003741).
Author information
Authors and Affiliations
Corresponding author
Additional information
Haijuan Liu and Xiaodong Luo contributed equally to this article.
Rights and permissions
About this article
Cite this article
Liu, H., Luo, X., Shen, E. et al. Alteration of T cell subtypes in spleen and antibodies of serum in mice infected with Angiostrongylus cantonensis . Parasitol Res 112, 1255–1260 (2013). https://doi.org/10.1007/s00436-012-3259-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-012-3259-1