Abstract
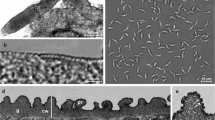
One type of sarcocyst was found in two of eight investigated jackdaws (Corvus monedula) and proposed as Sarcocystis corvusi sp. nov. By light microscope, cysts resembled a thick thread and were very long (the largest fragment found amounted to 6 mm) and relatively thin (up to 60 μm). The cyst wall measured <1 μm and seemed smooth. Using a computerized image analysis system, knolls, which resembled protrusions, were visible on the wall surface. Ultrastructurally, the cyst wall was wavy and reached up to 1.1 μm. The waves were of different heights and resembled low protrusions. The parasitophorous vacuolar membrane had many invaginations. Lancet- or orange segment-shaped cystozoites were 5.9–7.3 μm long. These sarcocysts had type-1 tissue cyst wall. According to 18S rRNA, 28S rRNA genes and ITS-1 region sequences, it was shown that S. corvusi is a genetically separate species. On the basis of these genetic markers, S. corvusi was most closely related to S. columbae, S. calchasi and S. wobeseri which parasitize birds and are characterized by the same type of sarcocyst wall.


Similar content being viewed by others
References
Burfield I, van Bommel F (2004) Birds in Europe: population estimates, trends, and conservation status, vol 12, BirdLife Conservation. BirdLife International, Cambridge
Černá Ž (1984) The role of birds as definitive hosts and intermediate hosts of heteroxenous coccidians. J Protozool 31:579–581
Drouin TE, Mahrt JL (1979) The prevalence of Sarcocystis Lankester, 1882, in some bird species in western Canada, with notes on its life cycle. Can J Zool 57:1915–1921
Drouin TE, Mahrt JL (1980) The morphology of cysts of Sarcocystis infecting birds in western Canada. Can J Zool 58:1477–1482
Dubey JP, Speer CA, Fayer R (1989) Sarcocystosis of animals and man. CRC Press, Boca Raton
Dubey JP, Odening K (2001) Toxoplasmosis and related infections. In: Samuel WM, Pybus MJ, Kocan AA (eds) Parasitic diseases of wild mammals. Iowa State University Press, Ames, pp 478–519
Edgar RC (2004) MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res 32:1792–1797
Gjerde B, Dahlgren SS (2010) Corvid birds (Corvidae) act as definitive hosts for Sarcocystis ovalis in moose (Alces alces). Parasitol Res 107:1445–1453
Holmdahl OJ, Morrision DA, Ellis JT, Huong LT (1999) Evolution of ruminant Sarcocystis (Sporozoa) parasites based on small subunit rDNA sequences. Mol Phylogenet Evol 11:27–37
Kutkienė L, Sruoga A, Butkauskas D (2006) Sarcocystis sp. from white-fronted goose (Anser albiforns): cyst morphology and life cycle studies. Parasitol Res 99:562–565
Kutkienė L, Sruoga A, Butkauskas D (2008) Sarcocystis sp. from the goldeneye (Bucephala clangula) and the mallard (Anas platyrhynchos): cyst morphology and ribosomal DNA analysis. Parasitol Res 102:691–696
Kutkienė L, Prakas P, Sruoga A, Butkauskas D (2009) Sarcocystis in the birds family Corvidae with description of Sarcocystis cornixi sp. nov. from the hooded crow (Corvus cornix). Parasitol Res 104:329–336
Kutkienė L, Prakas P, Sruoga A, Butkauskas D (2010) The mallard duck (Anas platyrhynchos) as intermediate host for Sarcocystis wobeseri sp. nov. from the barnacle goose (Branta leucopsis). Parasitol Res 107:879–888
Kutkienė L, Prakas P, Sruoga A, Butkauskas D (2012a) Description of Sarcocystis anasi sp. nov. and Sarcocystis albifronsi sp. nov. in birds of the order Anseriformes. Parasitol Res 110:1043–1046
Kutkienė L, Prakas P, Sruoga A, Butkauskas D (2012b) Description of Sarcocystis turdusi sp. nov. from the common blackbird (Turdus merula). Parasitology 139:1438–1443
Morrison DA, Bornstein S, Thebo P, Wernery U, Kinne J, Mattsson JG (2004) The current status of the small subunit rRNA phylogeny of the coccidia (Sporozoa). Int J Parasitol 34:501–514
Munday BL, Hartley WJ, Harrigan KE, Presidente PJA, Obendorf DL (1979) Sarcocystis and related organisms in Australian wildlife: II. Survey findings in birds, reptiles, amphibians and fish. J Wildl Dis 15:57–73
Olias P, Gruber AD, Hafez HM, Heydorn AO, Mehlhorn H, Lierz M (2010a) Sarcocystis calchasi sp. nov. of the domestic pigeon (Columba livia f. domestica) and the Northern goshawk (Accipiter gentilis): light and electron microscopical characteristics. Parasitol Res 106:577–585
Olias P, Olias L, Lierz M, Mehlhorn H, Gruber AD (2010b) Sarcocystis calchasi is distinct to Sarcocystis columbae sp. nov. from the wood pigeon (Columba palumbus) and Sarcocystis sp. from the sparrowhawk (Accipiter nisus). Vet Parasitol 171:7–14
Olias P, Olias L, Krücken J, Lierz M, Gruber AD (2011) High prevalence of Sarcocystis calchasi sporocysts in European Accipiter hawks. Vet Parasitol 175:230–236
Pak SM, Eshtokina NV (1984) Sarcosporidians of birds. In: Panin VJ (ed) Sarcosporidians of animals in Kazakhstan. Nauka, Alma-Ata, pp 150–168
Pinayeva LM, Pak CM, Kokhno LI (1998) Sarcocystis of the wild birds of Kazakhstan. Parasitol Int 47:143
Prakas P, Kutkienė L, Sruoga A, Butkauskas D (2011) Sarcocystis sp. from the herring gull (Larus argentatus) identity to Sarcocystis wobeseri based on cyst morphology and DNA results. Parasitol Res 109:1603–1608
Ronquist F, Huelsenbeck JP (2003) MrBayes 3: Bayesian phylogenetic inference under mixed models. Bioinformatics 19:1572–1574
Acknowledgments
We would like to express our gratitude to Mrs. I. Žalakevičienė from the Centre of Innovative Medicine, Department of Experimental and Clinical Medicine (Vilnius, Lithuania), for her help in carrying out investigations of electron microscopy.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Prakas, P., Kutkienė, L., Butkauskas, D. et al. Molecular and morphological investigations of Sarcocystis corvusi sp. nov. from the jackdaw (Corvus monedula). Parasitol Res 112, 1163–1167 (2013). https://doi.org/10.1007/s00436-012-3247-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-012-3247-5