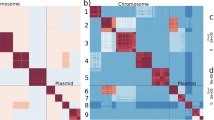
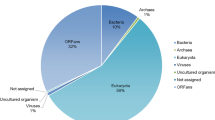
Abstract
Covalent modification of proteins by ubiquitin (Ub) and ubiquitin-like modifiers (Ubls) regulates many cellular functions in eukaryotes. These modifications are likely to be associated with pathogenesis, growth, and development of many protozoan parasites but molecular details about this pathway are unavailable for most protozoa. This study presents an analysis of the Ub pathway in three members of the Entamoeba species. Using bioinformatics tools we have identified all Ub and Ubl genes along with their corresponding activating, conjugating, and ligating enzymes (E1, E2s, and E3s) in three Entamoeba species, Entamoeba histolytica, Entamoeba dispar, and Entamoeba invadens. Phylogenetic trees were established for the identified E2s and RING finger E3s using maximum-likelihood method to infer the relationship among these proteins. In silico co-domain analysis of RING finger E3s implicates these proteins in a variety of functions. Several known and putative regulatory motifs were identified in the upstream regions of RING finger domain containing E3 genes. All E2 and E3 genes were analyzed in genomic context in E. histolytica and E. dispar. Most E2s and E3s were in syntenic positions in the two genomes. Association of these genes with transposable elements (TEs) was compared between E. histolytica and E. dispar. A closer association was found between RING finger E3s with TEs in E. histolytica. In summary, our analyses suggests that the complexity of the Ub pathway in Entamoeba species is close to that observed in higher eukaryotes. This study provides important data for further understanding the role of Ub pathway in the biology of these organisms.




Similar content being viewed by others
Abbreviations
- APC/C:
-
Anaphase promoting complex/cyclosome
- CRL:
-
Cullin-RING ligases
- HECT:
-
Homologous to E6-AP C-terminal
- RING:
-
Really interesting new gene
- PHD:
-
Plant homeo domain
- TE:
-
Transposable element
- UBC:
-
Ubiquitin conjugating
- Ubl:
-
Ubiquitin-like
- UPP:
-
Ubiquitin–proteasome pathway
References
Ali I, Ehrenkaufer G, Hackney J, Singh U (2007) Growth of the protozoan parasite Entamoeba histolytica in 5-azacytidine has limited effects on parasite gene expression. BMC Genomics 8:7
Alpi A, Pace P, Babu M, Patel K (2008) Mechanistic insight into site-restricted monoubiquitination of FANCD2 by Ube2t, FANCL, and FANCI. Mol Cell 32:767–777
Aravind L, Koonin E (2000) The U box is a modified RING finger—a common domain in ubiquitination. Curr Biol 10:R132–R134
Bailey T, Williams N, Misleh C, Li W (2006) MEME: Discoverng and analyzing DNA and protein sequence motifs. Nucleic Acids Res 34:W369–W373
Bays N, Hampton R (2002) Cdc48-Ufd1-Npl4: stuck in the middle with Ub. Curr Biol 12:R366–R371
Biederer T, Volkwein C, Sommer T (1997) Role of Cue1p in ubiquitination and degradation at the ER surface. Science 278:1806–1809
Burroughs A, Jaffee M, Iyer L, Aravind L (2008) Anatomy of the E2 ligase fold: implications for enzymology and evolution of ubiquitin/ub-like protein conjugation. J Struct Biol 162:205–218
Capili A, Schultz D, RauscherIII F, Borden K (2001) Solution structure of the PHD domain from the KAP-1 corepressor: structural determinants for PHD, RING and LIM zinc-binding domains. EMBO J 20:165–177
Carbia-Nagashima A, Gerez J, Perez-Castro C, Paez-Pereda M, Silberstein S, Stalla GK, Holsboer F, Arzt E (2007) RSUME, a small RWD-containing protein, enhances SUMO conjugation and stabilizes HIF-1alpha during hypoxia. Cell 131:309–323
Ciechanover A, Schwartz A (2004) The ubiquitin system: pathogenesis of human diseases and drug targeting. Biochim Biophys Acta 1695:3–17
Citarelli M, Teotia S, Lamb R (2010) Evolutionary history of the poly (ADP-ribose) polymerase gene family in eukaryotes. BMC Evol Biol 10:308
Coscoy L, Sanchez D, Ganem D (2001) A novel class of herpesvirus-encoded membrane-bound E3 ubiquitin ligases regulates endocytosis of proteins involved in immune recognition. J Cell Biol 155:1265–1273
Denuc A, Marfany G (2010) SUMO and ubiquitin paths converge. Biochem Soc Trans 38:34–39
Doerig C (2004) Protein kinases as targets for anti-parasitic chemotherapy. Biochim Biophys Acta 1697:155–168
Ehrenkaufer G, Haque R, Hackney J, Eichinger D, Singh U (2007) Identification of developmentally regulated genes in Entamoeba histolytica: insights into mechanisms of stage conversion in a protozoa parasite. Cell Microbiol 9:1426–1444
Eisenhaber B, Chumak N, Eisenhaber F, Hauser M (2007) The ring between ring fingers (RBR) protein family. Genome Biol 8:209–219
Furukawa K, Mizushima N, Noda T, Ohsumi Y (2000) A protein conjugation system in yeast with homology to biosynthetic enzyme reaction of prokaryotes. J Biol Chem 275:7462–7465
Geng J, Klionsky D (2008) The Atg8 and Atg12 ubiquitin-like conjugation systems in macroautophagy. ‘Protein modifications: beyond the usual suspects’ review series. EMBO Rep 9:859–864
Gómez C, Pérez D, López-Bayghen E, Orozco E (1998) Transcriptional analysis of the EhPgp1 promoter of Entamoeba histolytica multidrug-resistant mutant. J Biol Chem 273:7277–7284
Gonzalez J, Bai G, Frevert U, Corey E, Eichinger D (1999) Proteasome-dependent cyst formation and stage-specific ubiquitin mRNA accumulation in Entamoeba invadens. Eur J Biochem 264:897–904
Goodman C, McFadden G (2007) Fatty acid biosynthesis as a drug target in apicomplexan parasites. Curr Drug Targets 8:15–30
Hackney J, Ehrenkaufer G, Singh U (2007) Identification of putative transcriptional regulatory networks in Entamoeba histolytica using Bayesian inference. Nucleic Acids Res 35:2141–2152
Hatakeyama S, Yada M, Matsumoto M, Ishida N, Nakayama K (2001) U Box proteins as a new family of ubiquitin-protein ligases. J Biol Chem 276:33111–33120
Heck J, Cheung S, Hampton R (2010) Cytoplasmic protein quality control degradation mediated by parallel actions of the E3 ubiquitin ligases Ubr1 and San1. Proc Natl Acad Sci U S A 107:1106–11011
Hershko A (2005) The ubiquitin system for protein degradation and some of its roles in the control of the cell division cycle. Cell Death Differ 12:1191–1197
Hochstrasser M (2009) Origin and function of ubiquitin-like proteins. Nature 458:422–429
Horrocks P, Newbold C (2000) Intraerythrocytic polyubiquitin expression in Plasmodium falciparum is subjected to developmental and heat-shock control. Mol Biochem Parasitol 105:115–125
Jentsch S, Seufert W, Hauser H (1991) Genetic analysis of the ubiquitin system. Biochim Biophys Acta 1089:127–139
Jin L, Williamson A, Banerjee S, Philipp I, Rape M (2008) Mechanism of ubiquitin-chain formation by the human anaphase-promoting complex. Cell 133:653–665
Kerscher O, Felberbaum R, Hochstrasser M (2006) Modification of proteins by ubiquitin and ubiquitin-like proteins. Annu Rev Cell Dev Biol 22:159–180
Kosarev P, Mayer K, Hardtke C (2002) Evaluation and classification of RING-finger domains encoded by the Arabidopsis genome. Genome Biol 3:RESEARCH0016.0011–RESEARCH0016.0012
Kostova Z, Tsai Y, Weissman A (2007) Ubiquitin ligases, critical mediators of endoplasmic reticulum-associated degradation. Semin Cell Dev Biol 18:770–779
Kumari V, Sharma R, Yadav V, Gupta A, Bhattacharya A, Bhattacharya S (2011) Differential distribution of a SINE element in the Entamoeba histolytica and Entamoeba dispar genomes: role of the LINE encoded endonuclease. BMC Genomics 12:267
LaCount D, Vignali M, Chettier R, Phansalkar A, Bell R, Hesselberth J, Schoenfeld L, Ota I, Sahasrabudhe S, Kurschner C, Fields S, Hughes R (2005) A protein interaction network of the malaria parasite Plasmodium falciparum. Nature 438:103–107
Lindenthal C, Weich N, Chia Y, Heussler V, Klinkert M (2005) The proteasome inhibitor MLN-273 blocks exoerythrocytic and erythrocytic development of Plasmodium parasites. Parasitology 131:37–44
Lorenzi H, Thiagarajan M, Haas B, Wortman J, Hall N, Caler E (2008) Genome wide survey, discovery and evolution of repetitive elements in three Entamoeba species. BMC Genomics 9:595
Lorenzi H, Puiu D, Miller J, Brinkac L, Amedeo P, Hall N, Caler E (2010) New assembly, reannotation and analysis of the Entamoeba histolytica genome reveal new genomic features and protein content information. PLoS Negl Trop Dis 4:e716
Madsen L, Schulze A, Seeger M, Hartmann-Petersen R (2007) Ubiquitin domain proteins in disease. BMC Biochem 8(Suppl 1):S1
Manning-Cela R, Jaishankar S, Swindle J (2006) Life-cycle and growth-phase-dependent regulation of the ubiquitin genes of Trypanosoma cruzi. Arch Med Res 37:593–601
Matsumoto M, Wickliffe K, Dong K, Yu C, Bosanac I, Bustos D, Phu L, Kirkpatrick D, Hymowitz S, Rape M, Kelly RF, Dixit VM (2010) K11-linked polyubiquitination in cell cycle control revealed by a K11 linkage-specific antibody. Mol Cell 39:477–484
Merlet J, Burger J, Gomes J, Pintard L (2009) Regulation of cullin-RING E3 ubiquitin-ligases by neddylation and dimerization. Cell Mol Life Sci 66:1924–1938
Michelle C, Vourch P, Mignon L, Andres C (2009) What was the set of ubiquitin and ubiquitin-like conjugating enzymes in the eukaryote common ancestor? J Mol Evol 68:616–628
Myler M (2008) Searching the Tritryp genomes for drug targets. Adv Exp Med Biol 625:133–140
Newton K, Vucic D (2007) Ubiquitin ligases in cancer: ushers for degradation. Cancer Invest 25:502–513
Noma A, Sakaguchi Y, Suzuki T (2009) Mechanistic characterization of the sulfur-relay system for eukaryotic 2-thiouridine biogenesis at tRNA wobble positions. Nucleic Acids Res 37:1335–1352
Parry G, Estelle M (2004) Regulation of cullin-based ubiquitin ligases by the Nedd8/RUB ubiquitin-like proteins. Semin Cell Dev Biol 15:221–229
Ponder E, Bogyo M (2007) Ubiquitin-like modifiers and their deconjugating enzymes in medically important parasitic protozoa. Eukaryot Cell 6:1943–1952
Ponts N, Yang J, Chung D, Prudhomme J, Girke T, Horrocks P, Roch KL (2008) Deciphering the ubiquitin-mediated pathway in apicomplexan parasites: a potential strategy to interfere with parasite virulence. PLoS One 3:e2386
Purdy J, Pho L, Mann B, Petri WJ (1996) Upstream regulatory elements controlling expression of the Entamoeba histolytica lectin. Mol Biochem Parasitol 34:7891–7903
Raasi S, Wolf D (2007) Ubiquitin receptors and ERAD: a network of pathways to the proteasome. Semin Cell Dev Biol 18:780–791
Rassi S, Schmidtke G, Groettrup M (2001) The ubiquitin-like protein FAT10 forms covalent conjugates and induces apoptosis. J Biol Chem 276:35334–35343
Rhodes D, Bd B, Trowsdale J (2005) Relationship between SPRY and B30.2 protein domains. Evolution of a component of immune defence? Immunology 116:411–417
Rice P, Longden I, Bleasby A (2000) EMBOSS: the European Molecular Biology Open Software Suite. Trends Genet 16:276–277
Ritchie K, Zhang D (2004) ISG15: the immunological kin of ubiquitin. Semin Cell Dev Biol 15:237–246
Rodrigo-Brenni M, Morgan D (2007) Sequential E2s drive polyubiquitin chain assembly on APC targets. Cell 130:127–139
Romero-Díaz M, Gómez C, López-Reyes I, Martínez M, Orozco E, Rodríguez M (2010) Structural and functional analysis of the Entamoeba histolytica EhrabB gene promoter. Gene 455:32–42
Salvat C, Wang G, Dastur A, Lyon N, Huibregtse J (2004) The -4 phenylalanine is required for substrate ubiquitination catalyzed by HECT ubiquitin ligases. J Biol Chem 279:18935–18943
Schlieker C, Veen AVd, Damon J, Spooner E, Ploegh H (2008) A functional proteomics approach links the ubiquitin-related modifier Urm1 to a tRNA modification pathway. Proc Natl Acad Sci U S A 105:18255–18260
Schüle T, Rose M, Entian K, Thumm M, Wolf D (2000) Ubc8p functions in catabolite degradation of fructose-1, 6-bisphosphate in yeast. EMBO J 19:2161–2167
Shilova V, Garbuz D, Myasyankina E, Chen B, Evgen’ev M, Feder M, Zatsepina O (2006) Remarkable site specificity of local transposition into the Hsp70 promoter of Drosophila melanogaster. Genetics 173:809–820
Skaar J, Pagano M (2009) Control of cell growth by the SCF and APC/C ubiquitin ligases. Curr Opin Cell Biol 21:816–824
Stone S, Hauksdottir H, Troy A, Herschleb J, Kraft E, Callis J (2005) Functional analysis of the RING-type ubiquitin ligase family of Arabidopsis. Plant Physiol 137:13–30
Suzuki K, Nakamura M, Nariai Y, Dekio S, Tanigawa Y (1996) Monoclonal nonspecific suppressor beta (MNSF beta) inhibits the production of TNF-alpha by lipopolysaccharide-activated macrophages. Immunology 195:187–198
Varshavsky A (1997) The N-end rule pathway of protein degradation. Genes Cells 2:13–28
Whitney N, Pearson L, Lunsford R, McGill L, Gomer R, Lindsey D (2006) A putative Ariadne-like ubiquitin ligase is required for Dictyostelium discoideum development. Eukaryot Cell 5:1820–1825
Wilkinson C, Dittmar G, Ohi M, Uetz P, Jones N, Finley D (2004) Ubiquitin-like protein Hub1 is required for pre-mRNA splicing and localization of an essential splicing factor in fission yeast. Curr Biol 14:2283–2288
Wöstmann C, Tannich E, Grunwald T (1992) Ubiquitin of Entamoeba histolytica deviates in six amino acid residues from the consensus of all other known ubiquitins. FEBS 308:54–58
Wöstmann C, Liakopoulous D, Ciechanover A, Grunwald T (1996) Characterization of ubiquitin genes and transcripts and demonstration of an ubiquitin-conjugating system in Entamoeba histolytica. Mol Biochem Parasitol 82:81–92
Xu P, Duong D, Seyfried N, Cheng D, Xie Y, Robert J, Rush J, Hochstrasser M, Finley D, Peng J (2009) Quantitative proteomics reveals the function of unconventional ubiquitin chains in proteasomal degradation. Cell 137:133–145
Ye Y, Meyer H, Rapoport T (2001) The AAA ATPase Cdc48/p97 and its partners transport proteins from the ER into the cytosol. Nature 414:652–656
Ying M, Zhan Z, Wang W, Chen D (2009) Origin and evolution of ubiquitin-conjugating enzymes from Guillardia theta nucleomorph to hominoid. Gene 447:72–85
Zhao J (2007) Sumoylation regulates diverse biological processes. Cell Mol Life Sci 64:3017–3033
Acknowledgments
This work was supported by grants from the Department of Science and Technology to ST. SA was supported by fellowship from the University Grants Commission. PG was supported by the Defense Research and Development Organization grant to ST. We thank Dr. Sudha Bhattacharya for sharing the data on transposons. We are grateful to Dr Sudha and Alok Bhattacharya for critical reading of the manuscript and for many stimulating discussions.
Author information
Authors and Affiliations
Corresponding author
Additional information
Shweta Arya and Gaurav Sharma contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Online resource 1
(PDF 78 kb)
Online resource 2
(PDF 79 kb)
Online resource 3
(PDF 172 kb)
Online resource 4
(PDF 272 kb)
Online resource 5
(PDF 85 kb)
Online resource 6
(PDF 93 kb)
Online resource 7
(PDF 152 kb)
Online resource 8
(PDF 100 kb)
Online resource 9
(PDF 169 kb)
Online resource 10
(PDF 96 kb)
Online resource 11
(PDF 101 kb)
Online resource 12
(PDF 98 kb)
Online resource 13
(PDF 88 kb)
Online resource 14
(PDF 187 kb)
Rights and permissions
About this article
Cite this article
Arya, S., Sharma, G., Gupta, P. et al. In silico analysis of ubiquitin/ubiquitin-like modifiers and their conjugating enzymes in Entamoeba species. Parasitol Res 111, 37–51 (2012). https://doi.org/10.1007/s00436-011-2799-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-011-2799-0