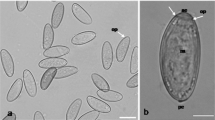
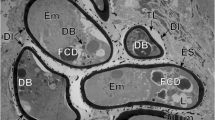
Abstract
Eggs of most species digenean flukes hatch in the external environment to liberate larvae that seek and penetrate a snail intermediate host. Those of the human liver flukes, Opisthorchis viverrini, hatch within the gastrointestinal canal of their snail hosts. While adult parasites are primarily responsible for the pathology in cases of human opisthorchiasis, their eggs also contribute by inducing granulomata and in serving as nidi for gallstone formation. In view of the peculiar biology of O. viverrini eggs and their contribution to pathology, we investigated embryogenesis in this species by light and transmission electron microscopy. Egg development was traced from earliest stages of coalescence in the ootype until full embryonation in the distal region of the uterus. Fully mature eggs were generally impermeable to resin and could not be examined by conventional electron microscopy methods. However, the use of high-pressure freezing and freeze-substitution fixation of previously fixed eggs enabled the internal structure of mature eggs, particularly the subshell envelopes, to be elucidated. Fertilization occurs in the ootype, and the large zygote is seen therein with a single spermatozoon wrapped around its plasma membrane. As the zygote begins to divide, the spent vitellocytes are pushed to the periphery of the eggs, where they progressively degrade. The early eggshell is formed in the ootype by coalescing eggshell precursor material released by approximately six vitelline cells. The early eggs have a thinner eggshell and are larger than, but lack the characteristic shape of, mature eggs. Characteristic shell ornamentation, the “muskmelon” appearance of eggs, appears after eggshell polymerization in the ootype. Pores are not present in the shell of O. viverrini eggs. The inner and outer envelopes are poorly formed in this species, with the outer envelope evident beneath the eggshell at the opercular pole of the mature egg. The miracidium has a conical anterior end that lacks the distinctive lamellar appearance of the terebratorium of other digeneans, such as the schistosomes. The miracidium is richly glandular, containing an apical gland in the anterior end, large cephalic gland, and posterior secretory glands. Each gland contains a secretory product with different structure. The paucity of vitelline cells associating with eggs, the reduced size of eggs, and reduced complexity of the extraembryonic envelopes are interpreted as adaptations to the peculiar hatching biology of the miracidia.







Similar content being viewed by others

References
Ashton PD, Harrop R, Shah B, Wilson RA (2001) The schistosome egg: development and secretions. Parasitology 122:329–338
Beer SA, German SM, Ditrich O, Giboda M (1991) The morphofunctional characteristics of the ultrastructure of the outer membrane of opisthorchid eggs. 1. The hatching of miracidia. Med Parazitol (Mosk) 2:18–22
Cai G, Bae Y, Zhang Y, He Y, Jiang M, He L (2008) Expression and characterization of two tyrosinases from the trematode Schistosoma japonicum. Parasitol Res 104:601–609
Colhoun LM, Fairweather I, Brennan GP (1998) Observations on the mechanism of eggshell formation in the liver fluke, Fasciola hepatica. Parasitology 116:555–567
Cribb TH, Bray RA, Olson PD, Littlewood DT (2003) Life cycle evolution in the digenea: a new perspective from phylogeny. Adv Parasitol 54:197–254
Ditrich O, Giboda M, Scholz T, Beer SA (1992) Comparative morphology of eggs of the Haplorchiinae (Trematoda: Heterophyidae) and some other medically important heterophyid and opisthorchiid flukes. Folia Parasitol (Praha) 39:123–132
Ebersberger I, Knobloch J, Kunz W (2005) Cracks in the shell-zooming in on eggshell formation in the human parasite Schistosoma mansoni. Dev Genes Evol 215:261–267
Elkins DB, Haswell-Elkins MR, Mairiang E, Mairiang P, Sithithaworn P, Kaewkes S, Bhudhisawasdi V, Uttaravichien T (1990) A high frequency of hepatobiliary disease and suspected cholangiocarcinoma associated with heavy Opisthorchis viverrini infection in a small community in north-east Thailand. Trans R Soc Trop Med Hyg 84:715–719
El-Shoura SM, Ramadan NF, Hassouna O, Mahmoud HI, Shetty S, Westmuckett A (1995) Human bilharzial ureters: I. Fine structure of eggs deposited in the submucosa and muscularis. Afr J Med Med Sci 24:219–225
Fitzpatrick JM, Hirai Y, Hirai H, Hoffmann KF (2007) Schistosome egg production is dependent upon the activities of two developmentally regulated tyrosinases. FASEB J 21:823–835
Fujino T, Nakano T, Washioka H, Tonosaki A, Ichikawa H, Fried B (2000) Comparative ultrastructure of eggs in Echinostoma paraensei, E. caproni, and E. trivolvis (Trematoda: Echinostomatidae). Parasitol Res 86:427–430
Irwin SWB, Threadgold LT (1971) Electron microscope studied of Fasciola hepatica. X. Egg formation. Exp Parasitol 31:321–331
Jones MK, Ernst I, Whittington ID (1997) Variation in the egg cell forming region of Gyrodactylus kobayashii Hukuda, 1940 (Monogenea:Gyrodactylidae). Int J Parasitol 27:507–516
Jones MK, Bong SH, Green KM, Holmes P, Duke M, Loukas A, McManus DP (2008) Correlative and dynamic imaging of the hatching biology of Schistosoma japonicum from eggs prepared by high pressure freezing. PLoS Negl Trop Dis 2:e334
Kusel JR (1970) Studies on the structure and hatching of the eggs of Schistosoma mansoni. Parasitology 60:79–88
Moczon T, Swiderski Z, Huggel H (1992) Schistosoma mansoni: the chemical nature of the secretions produced by the Mehlis' gland and ootype as revealed by cytochemical studies. Int J Parasitol 22:65–73
Müller B, Schmidt J, Mehlhorn H (2007) PCR diagnosis of infections with different species of Opisthorchiidae using a rapid clean-up procedure for stool samples and specific primers. Parasitol Res 100:905–909
Neill PJ, Smith JH, Doughty BL, Kemp M (1988) The ultrastructure of the Schistosoma mansoni egg. Am J Trop Med Hyg 39:52–65
Orido Y (1991) Ultrastructure of Mehlis' gland in the lung fluke, Paragonimus ohirai (Trematoda: Troglotrematidae). J Morphol 207:9–16
Orido Y, Takamure A, Akamatsu T, Takashima Y (1994) Ultrastructure of the ootype of the lung fluke Paragonimus ohirai (Digenea, Troglotrematidae) in mated and single-infection worms. Parasitol Res 80:307–311
Pungpak S, Riganti M, Bunnag D, Harinasuta T (1985) Clinical features in severe opisthorchiasis viverrini. Southeast Asian J Trop Med Public Health 16:405–409
Riganti M, Pungpak S, Sachakul V, Bunnag D, Harinasuta T (1988) Opisthorchis viverrini eggs and adult flukes as nidus and composition of gallstones. Southeast Asian J Trop Med Public Health 19:633–636
Rim HJ, Chai JY, Min DY, Cho SY, Eom KS, Hong SJ, Sohn WM, Yong TS, Deodato G, Standgaard H, Phommasack B, Yun CH, Hoang EH (2003) Prevalence of intestinal parasite infections on a national scale among primary schoolchildren in Laos. Parasitol Res 91:267–272
Ruangsittichai J, Viyanant V, Vichasri-Grams S, Sobhon P, Tesana S, Upatham ES, Hofmann A, Korge G, Grams R (2006) Opisthorchis viverrini: identification of a glycine-tyrosine rich eggshell protein and its potential as a diagnostic tool for human opisthorchiasis. Int J Parasitol 36:1329–1339
Sadun EH (1955) Studies on Opisthorchis viverrini in Thailand. Am J Hyg 62:81–115
Salikhova ChR, Liubarskaia OD, Beer SA (1989) The outer membranes of the eggs of Opisthorchis felineus and Clonorchis sinensis. Med Parazitol (Mosk) 5:60–62
Schmidt J (1998) Glycan vesicle formation in vitellocytes and hatching vacuoles in eggs of Echinostoma caproni and Fasciola hepatica (Digenea). Tissue Cell 30:416–426
Sithithaworn P, Pipitgool V, Srisawangwong T, Elkins DB, Haswell-Elkins MR (1997) Seasonal variation of Opisthorchis viverrini infection in cyprinoid fish in north-east Thailand: implications for parasite control and food safety. Bull World Health Organ 75:125–131
Smyth JD, Halton DW (1983) The physiology of trematodes, 2nd edn. Cambridge University Press, Cambridge, p 446
Sriamporn S, Pisani P, Pipitgool V, Suwanrungruang K, Kamsa-ard S, Parkin DM (2004) Prevalence of Opisthorchis viverrini infection and incidence of cholangiocarcinoma in Khon Kaen, Northeast Thailand. Trop Med Int Health 9:588–594
Sri-Aroon P, Butraporn P, Limsomboon J, Kerdpuech Y, Kaewpoolsri M, Kiatsiri S (2005) Freshwater mollusks of medical importance in Kalasin Province, northeast Thailand. Southeast Asian J Trop Med Public Health 36:653–657
Sripa B, Kanla P, Sinawat P, Haswell-Elkins MR (2004) Opisthorchiasis-associated biliary stones: light and scanning electron microscopic study. World J Gastroenterol 10:3318–3321
Sripa B, Kaewkes S, Sithithaworn P, Mairiang E, Laha T, Smout M, Pairojkul C, Bhudhisawasdi V, Tesana S, Thinkamrop B, Bethony JM, Loukas A, Brindley PJ (2007) Liver fluke induces cholangiocarcinoma. PLoS Med 4:e201
Świderski Z (1994) Origin, differentiation and ultrastructure of egg envelopes surrounding the miracidia of Schistosoma mansoni. Acta Parasitol 39:64–72
Świderski Z, Bakhoum AJS, Młocicki D, Miquel J (2010) Ultrastructural studies on egg envelopes surrounding the miracidia of Mediogonimus jourdanei Mas-Coma et Rocamora, 1978 (Digenea, Microphalloidea, Prosthogonimidae). Acta Parasitol 55:245–253
Tesana S, Srisawangwonk T, Kaewkes S, Sithithaworn P, Kanla P, Arunyanart C (1991) Eggshell morphology of the small eggs of human trematodes in Thailand. Southeast Asian J Trop Med Public Health 22:631–636
Vatanasapt V, Tangvoraphonkchai V, Titapant V, Pipitgool V, Viriyapap D, Sriamporn S (1990) A high incidence of liver cancer in Khon Kaen Province, Thailand. Southeast Asian J Trop Med Public Health 21:489–494
Vichasri S, Viyanant V, Upatham ES (1982) Opisthorchis viverrini: intensity and rates of infection in cyprinoid fish from an endemic focus in Northeast Thailand. Southeast Asian J Trop Med Public Health 13:138–141
Waite JH, Rice-Ficht AC (1987) Presclerotized eggshell protein from the liver fluke Fasciola hepatica. Biochemistry 26:7819–7825
Waite JH, Rice-Ficht AC (1992) Eggshell precursor proteins of Fasciola hepatica. II. Microheterogeneity in vitelline protein B. Mol Biochem Parasitol 54:143–151
World Health Organization (2009) Review on the epidemiological profile of helminthiases and their control in the Western Pacific Region, 1997–2008
Xu YZ, Dresden MH (1990) The hatching of schistosome eggs. Exp Parasitol 70(2):236–240
Zurita M, Bieber D, Mansour TE (1989) Identification, expression and in situ hybridization of an eggshell protein gene from Fasciola hepatica. Mol Biochem Parasitol 37:11–17
Acknowledgment
We are grateful for the financial support of this work from the Thailand Research Fund through the Royal Golden Jubilee Ph.D. Program (Grant number PHD/0005/2550) to student Miss Panita Khampoosa and advisor Assoc. Prof. Dr. Smarn Tesana. We also acknowledge the support of the Higher Education Research Promotion and National Research University Project of Thailand, through the Health Cluster (SHeP-GMS), Office of the Higher Education Commission, Khon Kaen University. Malcolm Jones acknowledges the support from the Australian Research Council and National Health and Medical Research Council of Australia. We thank Dr. Kathryn Green for the assistance and advice with electron microscopy.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Video 1
Movement biology of O. viverrini miracidium. The video commences in mature egg incubated in DW for 20 days to prove maturation of miracidium within the eggshell (MPG 3,111 kb)
Rights and permissions
About this article
Cite this article
Khampoosa, P., Jones, M.K., Lovas, E.M. et al. Light and electron microscopy observations of embryogenesis and egg development in the human liver fluke, Opisthorchis viverrini (Platyhelminthes, Digenea). Parasitol Res 110, 799–808 (2012). https://doi.org/10.1007/s00436-011-2557-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-011-2557-3