Abstract
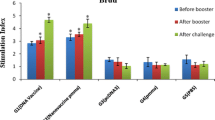
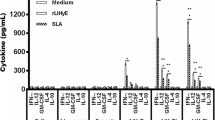
Various adjuvants and delivery systems have been evaluated for increasing the protective immune responses against leishmaniasis and mostly have been shown not to be effective enough. In this study, poly(d,l-lactide-co-glycolide) (PLGA) nanospheres as an antigen delivery system and CpG-ODN as an immunoadjuvant have been used for the first time to enhance the immune response against autoclaved Leishmania major (ALM). PLGA nanospheres were prepared by a double-emulsion (W/O/W) technique. Particulate characteristics were studied by scanning electron microscopy and particle size analysis. Mean diameter of ALM + CpG-ODN-loaded nanospheres was 300 ± 128 nm. BALB/c mice were immunized three times in 3-week intervals using ALM plus CpG-ODN-loaded nanospheres [(ALM + CpG-ODN)PLGA], ALM encapsulated PLGA nanospheres [(ALM)PLGA], (ALM)PLGA + CpG, ALM + CpG, ALM alone, or phosphate buffer solution (PBS). The intensity of infection induced by L. major challenge was assessed by measuring size of footpad swelling. The strongest protection, showed by significantly (P < 0.05) smaller footpad, was observed in mice immunized with (ALM + CpG-ODN)PLGA. The (ALM)PLGA, (ALM)PLGA + CpG, and ALM + CpG were also showed a significantly (P < 0.05) smaller footpad swelling compared to the groups received either PBS or ALM alone. The mice immunized with (ALM + CpG-ODN)PLGA, (ALM)PLGA + CpG, and ALM + CpG showed the highest IgG2a/IgG1 ratio, interferon-γ production, and lowest interleukin-4 production compared to the other groups. It is concluded that when both PLGA nanospheres and CpG-ODN adjuvants were used simultaneously, it induce stronger immune response and enhance protection rate against Leishmania infection.





Similar content being viewed by others
References
Ahmad-Nejad P, Hacker H, Rutz M, Bauer S, Vabulas RM, Wagner H (2002) Bacterial CPG-DNA and lipopolysaccharides activate toll-like receptors at distinct cellular compartments. Eur J Immunol 32(7):1958–1968
Alimohammadian MH, Khamesipour A, Darabi H, Firooz A, Malekzadeh S, Bahonar A, Dowlati Y, Modabber F (2002) The role of BCG in human immune responses induced by multiple injections of autoclaved Leishmania major as a candidate vaccine against leishmaniasis. Vaccine 21(3–4):174–180
Armijos RX, Weigel MM, Calvopina M, Hidalgo A, Cevallos W, Correa J (2004) Safety, immunogenecity, and efficacy of an autoclaved Leishmania amazonensis vaccine plus BCG adjuvant against new world cutaneous leishmaniasis. Vaccine 22(9–10):1320–1326
Badiee A, Jaafari MR, Khamesipour A (2007) Leishmania major: immune response in BALB/c mice immunized with stress-inducible protein 1 encapsulated in liposomes. Exp Parasitol 115(2):127–134
Bahar K, Dowlati Y, Shidani B, Alimohammadian MH, Khamesipour A, Ehsasi S, Hashemi-Fesharki R, Ale-Agha S, Modabber F (1996) Comparative safety and immunogenicity trial of two killed Leishmania major vaccines with or without BCG in human volunteers. Clin Dermatol 14(5):489–495
Barman SP, Lunsford L, Chambers P, Hedley ML (2000) Two methods for quantifying DNA extracted from poly(lactide-co-glycolide) microspheres. J Control Release 69(3):337–344
Bhowmick S, Ravindran R, Ali N (2007) Leishmanial antigens in liposomes promote protective immunity and provide immunotherapy against visceral leishmaniasis via polarized th1 response. Vaccine 25(35):6544–6556
Cabrera M, Blackwell JM, Castes M, Trujillo D, Convit J, Shaw MA (2000) Immunotherapy with live BCG plus heat killed Leishmania induces a T helper 1-like response in American cutaneous leishmaniasis patients. Parasite Immunol 22(2):73–79
Coelho EA, Tavares CA, Lima Kde M, Silva CL, Rodrigues JM Jr, Fernandes AP (2006) Mycobacterium HSP65 DNA entrapped into TDM-loaded PLGA microspheres induces protection in mice against Leishmania (leishmania) major infection. Parasitol Res 98(6):568–575
De Luca PM, Mayrink W, Alves CR, Coutinho SG, Oliveira MP, Bertho AL, Toledo VP, Costa CA, Genaro O, Mendonca SCF (1999) Evaluation of the stability and immunogenicity of autoclaved and nonautoclaved preparations of a vaccine against American tegumentary leishmaniasis. Vaccine 17(9–10):1179–1185
Diwan M, Tafaghodi M, Samuel J (2002) Enhancement of immune responses by co-delivery of a CPG oligodeoxynucleotide and tetanus toxoid in biodegradable nanospheres. J Control Release 85(1–3):247–262
Flynn B, Wang V, Sacks DL, Seder RA, Verthelyi D (2005) Prevention and treatment of cutaneous leishmaniasis in primates by using synthetic type D/A oligodeoxynucleotides expressing CPG motifs. Infect Immun 73(8):4948–4954
Follador I, Araujo C, Orge G, Cheng LH, de Carvalho LP, Bacellar O, Almeida RP, Carvalho EM (2002) Immune responses to an inactive vaccine against American cutaneous leishmaniasis together with granulocyte-macrophage colony-stimulating factor. Vaccine 20(9–10):1365–1368
Gupta RK, Siber GR (1995) Adjuvants for human vaccines—current status, problems and future prospects. Vaccine 13(14):1263–1276
Hemmi H, Takeuchi O, Kawai T, Kaisho T, Sato S, Sanjo H, Matsumoto M, Hoshino K, Wagner H, Takeda K et al (2000) A toll-like receptor recognizes bacterial DNA. Nature 408(6813):740–745
Iborra S, Carrion J, Anderson C, Alonso C, Sacks D, Soto M (2005) Vaccination with the Leishmania infantum acidic ribosomal p0 protein plus CPG oligodeoxynucleotides induces protection against cutaneous leishmaniasis in c57bl/6 mice but does not prevent progressive disease in BALB/c mice. Infect Immun 73(9):5842–5852
Iborra S, Parody N, Abánades DR, Bonay P, Prates D, Novais FO, Barral-Netto M, Alonso C, Soto M (2008) Vaccination with the Leishmania major ribosomal proteins plus CPG oligodeoxynucleotides induces protection against experimental cutaneous leishmaniasis in mice. Microbes Infect 10:1133–1141
Jaafari MR, Ghafarian A, Farrokh-Gisour A, Samiei A, Kheiri MT, Mahboudi F, Barkhordari F, Khamesipour A, McMaster WR (2006) Immune response and protection assay of recombinant major surface glycoprotein of Leishmania (rgp63) reconstituted with liposomes in BALB/c mice. Vaccine 24(29–30):5708–5717
Jaafari MR, Badiee A, Khamesipour A, Samiei A, Soroush D, Kheiri MT, Barkhordari F, McMaster WR, Mahboudi F (2007) The role of CpG ODN in enhancement of immune response and protection in BALB/c mice immunized with recombinant major surface glycoprotein of Leishmania (rgp63) encapsulated in cationic liposome. Vaccine 25(32):6107–6117
Javadian E, Nadim A, Tahvildare-Bidruni G, Assefi V (1976) Epidemiology of cutaneous leishmaniasis in Iran: B. Khorassan part V: report on a focus of zoonotic cutaneous leishmaniasis in Esferayen. Bull Soc Pathol Exot Fil 69(2):140–143
Johansen P, Men Y, Merkle HP, Gander B (2000) Revisiting PLA/PLGA microspheres: an analysis of their potential in parenteral vaccination. Eur J Pharm Biopharm 50(1):129–146
Kamil AA, Khalil EA, Musa AM, Modabber F, Mukhtar MM, Ibrahim ME, Zijlstra EE, Sacks D, Smith PG, Zicker F, El-Hassan AM (2003) Alum-precipitated autoclaved Leishmania major plus Bacille Calmette-Guerrin, a candidate vaccine for visceral leishmaniasis: safety, skin-delayed type hypersensitivity response and dose finding in healthy volunteers. Trans R Soc Trop Med Hyg 97(3):365–368
Kedzierski L, Zhu Y, Handman E (2006) Leishmania vaccines: progress and problems. Parasitology 133(Suppl):S87–S112
Keshavarz Valian H, Khoshabe Abdollah Kenedy L, Nateghi Rostami M, Miramin Mohammadi A, Khamesipour A (2008) Role of Mycobacterium vaccae in the protection induced by first generation Leishmania vaccine against murine model of leishmaniasis. Parasitol Res 103:21–28
Khalil EA, El Hassan AM, Zijlstra EE, Mukhtar MM, Ghalib HW, Musa B, Ibrahim ME, Kamil AA, Elsheikh M, Babiker A et al (2000) Autoclaved Leishmania major vaccine for prevention of visceral leishmaniasis: a randomised, double-blind, BCG-controlled trial in Sudan. Lancet 356(9241):1565–1569
Khamesipour A, Dowlati Y, Asilian A, Hashemi-Fesharki R, Javadi A, Noazin S, Modabber F (2005) Leishmanization: use of an old method for evaluation of candidate vaccines against leishmaniasis. Vaccine 23(28):3642–3648
Khamesipour A, Rafati S, Davoudi N, Maboudi F, Modabber F (2006) Leishmaniasis vaccine candidates for development: a global overview. Indian J Med Res 123(3):423–438
Krieg AM (1999) Mechanisms and applications of immune stimulatory CPG oligodeoxynucleotides. Biochim Biophys Acta BBA Gene Struct Expr 1489(1):107–116
Krieg AM (2002) CPG motifs in bacterial DNA and their immune effects. Annu Rev Immunol 20:709–760
Mauel J (2002) Vaccination against Leishmania infections. Curr Drug Targets Immune Endocr Metabol Disord 2(3):201–226
McCluskie MJ, Davis HL (1998) CPG DNA is a potent enhancer of systemic and mucosal immune responses against hepatitis B surface antigen with intranasal administration to mice. J Immunol (1950) 161(9):4463–4466
Mendez S, Tabbara K, Belkaid Y, Bertholet S, Verthelyi D, Klinman D, Seder RA, Sacks DL (2003) Coinjection with CPG-containing immunostimulatory oligodeoxynucleotides reduces the pathogenicity of a live vaccine against cutaneous leishmaniasis but maintains its potency and durability. Infect Immun 71(9):5121–5129
Misra A, Dube A, Srivastava B, Sharma P, Srivastava JK, Katiyar JC, Naik S (2001) Successful vaccination against Leishmania donovani infection in Indian langur using alum-precipitated autoclaved Leishmania major with BCG. Vaccine 19(25–26):3485–3492
Modabber F (1995) Vaccines against leishmaniasis. Ann Trop Med Parasitol 89(Supplement 1):83–88
Mohebali M, Khamesipour A, Mobedi I, Zarei Z, Hashemi-Fesharki R (2004) Double-blind randomized efficacy field trial of alum precipitated autoclaved Leishmania major vaccine mixed with BCG against canine visceral leishmaniasis in Meshkin-Shahr District, I.R. Iran. Vaccine 22(29–30):4097–4100
Newman KD, Sosnowski DL, Kwon GS, Samuel J (1998) Delivery of muc1 mucin peptide by poly(d, l-lactic-co-glycolic acid) microspheres induces type 1 T helper immune responses. J Pharm Sci 87(11):1421–1427
Newman KD, Elamanchili P, Kwon GS, Samuel J (2002) Uptake of poly(d, l-lactic-co-glycolic acid) microspheres by antigen-presenting cells in vivo. J Biomed Mater Res 60(3):480–486
Noazin S, Modabber F, Khamesipour A, Smith PG, Moulton LH, Nasseri K, Sharifi I, Khalil EA, Bernal ID, Antunes CM, Kieny MP, Tanner M (2008) First generation leishmaniasis vaccines: a review of field efficacy trials. Vaccine 26(52):6759–6767
Rebelatto MC, Guimond P, Bowersock TL, HogenEsch H (2001) Induction of systemic and mucosal immune response in cattle by intranasal administration of pig serum albumin in alginate microparticles. Vet Immunol Immunopathol 83(1–2):93–105
Reiner SL, Locksley RM (1995) The regulation of immunity to Leishmania major. Annu Rev Immunol 13:151–177
Rhee EG, Mendez S, Shah JA, Wu CY, Kirman JR, Turon TN, Davey DF, Davis H, Klinman DM, Coler RN, Sacks DL, Seder RA (2002) Vaccination with heat-killed Leishmania antigen or recombinant leishmanial protein and CPG oligodeoxynucleotides induces long-term memory cd4+ and cd8+ T cell responses and protection against Leishmania major infection. J Exp Med 195(12):1565–1573
Sacks D, Noben-Trauth N (2002) The immunology of susceptibility and resistance to Leishmania major in mice. Nat Rev Immunol 2(11):845–858
Satti IN, Osman HY, Daifalla NS, Younis SA, Khalil EA, Zijlstra EE, El Hassan AM, Ghalib HW (2001) Immunogenicity and safety of autoclaved Leishmania major plus BCG vaccine in healthy Sudanese volunteers. Vaccine 19(15–16):2100–2106
Scott P, Pearce E, Natovitz P, Sher A (1987) Vaccination against cutaneous leishmaniasis in a murine model. I. Induction of protective immunity with a soluble extract of promastigotes. J Immunol 139(1):221–227
Shimizu Y, Yamakami K, Gomi T, Nakata M, Asanuma H, Tadakuma T, Kojima N (2003) Protection against Leishmania major infection by oligomannose-coated liposomes. Bioorg Med Chem 11(7):1191–1195
Shimizu Y, Takagi H, Nakayama T, Yamakami K, Tadakuma T, Yokoyama N, Kojima N (2007) Intraperitoneal immunization with oligomannose-coated liposome-entrapped soluble leishmanial antigen induces antigen-specific T-helper type immune response in BALB/c mice through uptake by peritoneal macrophages. Parasite Immunol 29(5):229–239
Sinha VR, Trehan A (2003) Biodegradable microspheres for protein delivery. J Control Release 90(3):261–280
Tafaghodi M, Sajadi Tabassi SA, Jaafari M-R, Zakavi SR, Momen Nejad M (2004) Evaluation of the clearance characteristics of various microspheres in the human nose by gamma-scintigraphy. Int J Pharm 280:125–135
Tewary P, Pandya J, Mehta J, Sukumaran B, Madhubala R (2004) Vaccination with Leishmania soluble antigen and immunostimulatory oligodeoxynucleotides induces specific immunity and protection against Leishmania donovani infection. FEMS Immunol Med Microbiol 42(2):241–248
Venkataprasad N, Coombes AGA, Singh M, Rohde M, Wilkinson K, Hudecz F, Davis SS, Vordermeier HM (1999) Induction of cellular immunity to a mycobacterial antigen adsorbed on lamellar particles of lactide polymers. Vaccine 17(15–16):1814–1819
Verthelyi D, Kenney RT, Seder RA, Gam AA, Friedag B, Klinman DM (2002) Cpg oligodeoxynucleotides as vaccine adjuvants in primates. J Immunol (1950) 168(4):1659–1663
Waterborg JH (2002) Quantitation of proteins. In: Walker J (ed) The protein protocols handbook, 2nd edn. Humana, New Jersey, pp 3–36
Wu W, Weigand L, Belkaid Y, Mendez S (2006) Immunomodulatory effects associated with a live vaccine against Leishmania major containing CPG oligodeoxynucleotides. Eur J Immunol 36(12):3238–3247
Acknowledgements
This study was part of a research project which was financially supported by the vice chancellor for research of Mashhad University of Medical Sciences. We acknowledge the excellent technical assistance of Afshin Samiei and Dina Soroush.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tafaghodi, M., Khamesipour, A. & Jaafari, M.R. Immunization against leishmaniasis by PLGA nanospheres encapsulated with autoclaved Leishmania major (ALM) and CpG-ODN. Parasitol Res 108, 1265–1273 (2011). https://doi.org/10.1007/s00436-010-2176-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-010-2176-4