Abstract
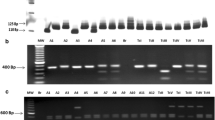
Different molecular markers have been employed for typing Trypanosoma cruzi strains from endemic areas of Chagas disease. The low-stringency single specific primer–polymerase chain reaction (LSSP–PCR) has been a sensitive and informative technique that uses the variable region of kinetoplast DNA minicircles as a genetic marker, allowing detection of DNA sequence variation. In the present study, we analyzed the intra-lineage genetic variability of the T. cruzi strains obtained from triatomine feces collected on filter paper FTA card by LSSP–PCR. The hybridization of the PCR products with a probe for the subgenus Schizotrypanum and a clone-specific probe from Dm28c confirmed the subgenus as T. (S.) cruzi and respective lineages as T. cruzi I. Phenetic analysis showed the presence of three clusters that diverged by different coefficients of similarity. Thirteen T. cruzi I genotypes were observed circulating among Triatoma pseudomaculata and Rhodnius nasutus from peridomiciliary and natural environments in five peri-urban and urban localities of Jaguaruana, Ceará, Brazil. These data indicate the importance of the circulation of T. cruzi I genotypes among T. pseudomaculata and R. nasutus in different environments and the possible risk of Chagas disease domestic transmission.



Similar content being viewed by others
References
Almeida CE, Vinhaes MC, Almeida JR, Silveira AC, Costa J (2000) Monitoring the domiciliary and peridomiciliary invasion process of Triatoma rubrovaria in the state of Rio Grande do Sul, Brazil. Mem Inst Oswaldo Cruz 95:761–768
Assis GFM, Azeredo BVM, La Fuente ALC, Diotaiuti L, Lana M (2007) Domiciliation of Triatoma pseudomaculata (Corrêa e Espínola 1964) in the Jequitinhonha valley, state of Minas Gerais. Rev Soc Bras Med Trop 40:391–396
Breniere SF, Bosseno MF, Revollo S, Rivera MT, Carlier Y, Tibayrenc M (1992) Direct identification of Trypanosoma cruzi natural clones in vectors and mammalian hosts by polymerase chain reaction amplification. Am J Trop Med Hyg 46:335–341
Breniere SF, Bosseno MF, Telleria J, Carrasco R, Vargas F, Yaksic N, Noireau F (1995) Field application of polymerase chain reaction diagnosis and strain typing of Trypanosoma cruzi in Bolivian triatomines. Am J Trop Med Hyg 53:179–184
Brisse S, Barnabé C, Tibayrenc M (2000) Identification of six Trypanosoma cruzi phylogenetic lineages by random amplified polymorphic DNA and multilocus enzyme electrophoresis. Int J Parasitol 30:35–44
Brito CMM, Sarquis O, Pires MQ, Lima MM, Pacheco RS (2005) Uso da PCR na detecção de DNA de Trypanosoma cruzi em fezes de triatomíneos naturalmente infectados. Rev Soc Bras Med Trop 38(Supl 1):320
Frota FCC, Lima JWO, Braga VSS (1999) Infecção humana pelo Trypanosoma cruzi, num foco urbano de Triatoma pseudomaculata, na cidade de Sobral. Norte do Ceará. Rev Soc Bras Med Trop 32(Supl I):85–86
Gaunt M, Miles M (2000) The ecotopes and evolution of triatomine bugs (Triatominae) and their associated trypanosomes. Mem Inst Oswaldo Cruz 95:557–565
Hamano S, Horio M, Miura S, Higo H, Iihoshi N, Noda K, Tada I, Takeuchi T (2001) Detection of kinetoplast DNA of Trypanosoma cruzi from dried feces of triatomine bugs by PCR. Parasitol International 50:135–138
Lima MM, Sarquis O (2008) Is Rhodnius nasutus (Hemíptera; Reduviidae) changing its habitat as a consequence of human activity? Parasitol Res 102:797–800
Madeira MF, Schubach A, Schubach TMP, Pacheco RS, Oliveira FS, Pereira AS, Figueiredo FB, Baptista C, Marzochi MCA (2006) Mixed infection with Leishmania (Viannia) braziliensis and Leishmania (Leishmania) chagasi in a naturally infected dog from Rio de Janeiro, Brazil. Trans R Soc Trop Med Hyg 100:442–445
Miles MA, Lanham SM, Souza A, Povoa M (1980) Further enzymic characters of Trypanosoma cruzi and their evalution for strain identification. Trans R Soc Trop Med Hyg 74:221–237
Oliveira AWS, Silva IG (2007) Geographical distribution and entomological indicators of synanthropic triatomines captured in the state of Goiás. Rev Soc Bras Med Trop 40:204–208
Oliveira FS, Pirmez C, Pires MQ, Brazil RP, Pacheco RS (2005) PCR-based diagnosis for detection of Leishmania in skin and blood of rodents from an endemic area of cutaneous and visceral leishmaniasis in Brazil. Vet Parasitol 129:219–227
Pacheco RS, Thomaz N, Brandão AA, Pires MQ, Momen H, Degrave W (1996) Synthetic oligonucleotide that discriminates between the subgenera Schizotrypanum and Megatrypanum. Parasite 3:297–299
Pacheco RS, Brito CMM, Sarquis O, Pires MQ, Borges-Pereira J, Lima MM (2005) Genetic heterogeneity in Trypanosoma cruzi strains from naturally infected triatomine vectors in northeastern Brazil: epidemiological implications. Biochemical Genetics 43:519–30
Romana CA, Pizarro JCN, Rodas E, Guilbert E (1999) Palm trees as ecological indicators of risk areas for Chagas disease. Trans R Soc Trop Med Hyg 93:594–595
Russomando G, Arias AR, Almiron M, Figueredo A, Ferreira ME, Morita K (1996) Trypanosoma cruzi: polymerase chain reaction-based detection in dried feces of Triatoma infestans. Exp Parasit 83:62–66
Salazar A, Schijman AG, Triana-Chávez O (2006) High variability of Colombian Trypanosoma cruzi lineage I stocks as revealed by low-stringency single primer-PCR minicircle signatures. Acta Trop 100:110–118
Sarquis O, Sposina R, Oliveira TG, MacCord JR, Cabello PH, Borges-Pereira J, Lima MM (2006) Aspects of peridomiciliary ecotopes in rural áreas of Northeastern Brazil associated to triatomine (Hemíptera, Reduviidae) infestation, vectors of Chagas disease. Mem Inst Oswaldo Cruz 101:143–147
Schmunis GA (2007) Epidemiology of Chagas disease in non-endemic countries: the role of international migration. Mem Inst Oswaldo Cruz 102:75–85
Silva ES, Gontijo CMF, Pacheco RS, Brazil RP (2004) Diagnosis of human visceral leishmaniasis by PCR using blood samples spotted on filter paper. Genet Mol Res 3:251–257
Silveira AC, Vinhaes MC (1998) Doença de Chagas: aspectos epidemiológicos e de controle. Rev Soc Bras Med Trop 31:15–60
Southern EM (1975) Detection of specific sequences among DNA fragments separated by gel electrophoresis. J Mol Biol 98:503–517
World Health Organization (2002) Control of Chagas disease. WHO Technical Report Series 811. Second report of the WHO Expert Committee, Geneva
World Health Organization (2007) New global effort to eliminate Chagas disease. http://www.who.int/mediacentre/news/releases/2007/pr36/en/index.html. Cited 10 August 2007
Acknowledgment
This work received financial support from Programa de Apoio a Pesquisa Estratégica (PAPES-III/FIOCRUZ) and Fundação de Apoio a Pesquisa do Estado do Rio de Janeiro (FAPERJ). The authors declare that the experiments comply with the current Brazilian laws.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Brito, C.M.M., Lima, M.M., Sarquis, O. et al. Genetic polymorphism in Trypanosoma cruzi I isolated from Brazilian Northeast triatomines revealed by low-stringency single specific primer–polymerase chain reaction. Parasitol Res 103, 1111–1117 (2008). https://doi.org/10.1007/s00436-008-1102-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-008-1102-5