Abstract
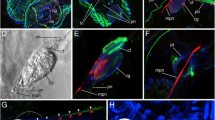
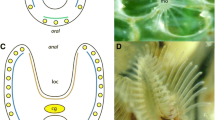
Thaliacea are marine planktonic animals within the taxon Tunicata. The species-poor taxon is characterized by diverse life cycles, with sexually (blastozooid) and asexually (oozooid) reproducing generations, that usually evolved polymorph phenotypes. While recent molecular phylogenetic studies indicate that tunicates might be closest living relatives to free-living craniates, additional insights into the evolution of the nervous systems of planktonic tunicates are overdue. To test the hypothesis that polymorphism in the different stages correlates with different distribution patterns of neurotransmitters, we comparatively conducted immunostaining experiments utilizing antibodies against serotonin and tyrosinated-α-tubulin, and confocal laser scanning microscopy in planktonic Thaliacea. We considerably increase the available data on serotonin-like immunoreactive (serotonin-lir) in Thaliacea and found that the pattern of distribution of serotonin-lir cells clearly differs between oozooid and blastozooid stages. Thereby, we reveal that the distribution of serotonin-lir cells in different tissues of thaliaceans can be considered as independent characters. Further, we test the potential phylogenetic signal present in serotonin-lir in thaliaceans, propose a phylogenetic mapping of conceptualized characters, and analyze the evolution of serotonin-lir cells. Comparing our results with data from previous studies indicated that it was necessary to reevaluate already described distribution patterns of serotonin-lir. Due to the complex life cycles of Salpida and Doliolida, erroneous descriptions of the localization of serotonin-lir occur in thaliacean literature. To facilitate the evaluation of data on serotonin-lir in thaliaceans for future readers, we suggest depicting whole-mount stainings of complete animals.





Similar content being viewed by others
References
Berrill NJ (1936) II—Studies in tunicate development. Part V—the evolution and classification of ascidians. Philos Trans R Soc B 226:43–70. https://doi.org/10.1098/rstb.1936.0002
Bone Q (1998) The biology of pelagic tunicates. OUP, Oxford
Braun K, Stach T (2016) Comparative study of serotonin-like immunoreactivity in the branchial basket, digestive tract, and nervous system in tunicates. Zoomorphology 135:351–366. https://doi.org/10.1007/s00435-016-0317-8
Braun K, Stach T (2017) Structure and ultrastructure of eyes and brains of Thalia democratica (Thaliacea, Tunicata, Chordata). J Morphol 278:1421–1437. https://doi.org/10.1002/jmor.20722
Braun K, Stach T (2018) Morphology and evolution of the central nervous system in adult tunicates. J Zool Syst Evol Res (accepted)
Brenneis G, Scholtz G (2015) Serotonin-immunoreactivity in the ventral nerve cord of Pycnogonida—support for individually identifiable neurons as ancestral feature of the arthropod nervous system. BMC Evol Biol 15:136. https://doi.org/10.1186/s12862-015-0422-1
Candiani S, Augello A, Oliveri D, Passalacqua M, Pennati R, De Bernardi F, Pestarino M (2001) Immunocytochemical localization of serotonin in embryos, larvae and adults of the lancelet, Branchiostoma floridae. Histochem J 33:413–420. https://doi.org/10.1023/A:1013775927978
Chamisso AV (1819) De animalibus quibusdam e classe vermium Linnaeana in circumnavigatione terrae auspicante Comite N. Romanzoff duce Ottone de Kotzebue annis 1815. 1816. 1817. 1818. peracta observatis. Fasciculus primus. De Salpa. Dümmler Verlag, Berlin
Chiba A (1998) Ontogeny of serotonin-immunoreactive cells in the gut epithelium of the cloudy dogfish, Scyliorhinus torazame, with reference to coexistence of serotonin and neuropeptide Y. Gen Comp Endocrinol 111:290–298. https://doi.org/10.1006/gcen.1998.7104
Delage Y, Hérouard E (1898) Les procordés. Schleicher Frères (eds.) Traité de Zoologie Concrète. Masson, Paris T.8.
Delsuc F, Brinkmann H, Chourrout D, Philippe H (2006) Tunicates and not cephalochordates are the closest living relatives of vertebrates. Nature 439:965–968. https://doi.org/10.1038/nature04336
Delsuc F, Philippe H, Tsagkogeorga G, Simion P, Tilak M-K, Turon X, López-Legentil S, Piette J, Lemaire P, Douzery EJP (2018) A phylogenomic framework and timescale for comparative studies of tunicates. bioRxiv. https://doi.org/10.1101/236448
Doran SA, Koss R, Tran CH, Christopher KJ, Gallin WJ, Goldberg JI (2004) Effect of serotonin on ciliary beating and intracellular calcium concentration in identified populations of embryonic ciliary cells. J Exp Biol 207:1415–1429. https://doi.org/10.1242/jeb.00924
Garstang W (1928) The morphology of the Tunicata, and its bearings on the phylogeny of the Chordata. Q J Microsc Sci 72:51–187
Georges D (1985) Presence of cells resembling serotonergic elements in four species of tunicates. Cell Tissue Res 242:341–348
Godeaux J (2003) History and revised classification of the order Cyclomyaria (Tunicata, Thaliacea, Doliolida). Bull Inst R Sci Nat Belgique Biol 73:191–222
Godeaux JEA, Bone Q, Braconnot JC (1998) Anatomy of Thaliacea. In: Bone Q (ed) The biology of pelagic tunicates. OUP, Oxford
Govindarajan AF, Bucklin A, Madin LP (2011) A molecular phylogeny of the Thaliacea. J Plankton Res 33:843–853. https://doi.org/10.1093/plankt/fbq157
Harris-Warrick RM, Cohen AH (1985) Serotonin modulates the central pattern generator for locomotion in the isolated lamprey spinal cord. J Exp Biol 116:27–46
Holland ND, Holland LZ (1993) Serotonin-containing cells in the nervous system and other tissues during ontogeny of a lancelet, Branchiostoma floridae. Acta Zool 74:195–204. https://doi.org/10.1111/j.1463-6395.1993.tb01234.x
Huxley TH (1860) XVI. On the anatomy and development of Pyrosoma. Trans Linn Soc Lond 23:193–250. https://doi.org/10.1111/j.1096-3642.1860.tb00130.x
Ihle JEW (1956) Dritte und letzte Ordnung der Thaliacea: Desmomyaria. In: Krumbach T (ed) Handbuch der Zoologie. Fünfter Band. Zweite Hälfte. Walter de Gruyter, Berlin, pp 401–544
Karnovsky MJ (1965) A formaldehyde–glutaraldehyde fixative of high osmolality for use in electron microscopy. J Cell Biol 27:137A
Kocot KM, Tassia MG, Halanych KM, Swalla BJ (2018) Phylogenomics offers resolution of major tunicate relationships. Mol Phylogenet Evol 121:166–173. https://doi.org/10.1016/j.ympev.2018.01.005
Kott P (2005) Catalogue of Tunicata in Australian waters. Australian Biological Resources Study, Canberra
Krohn A (1846) Observations sur la génération et le développement des Biphores (Salpa). Annales des Sciences naturelles. Troisième série Zoologie 6:110–132
Lacalli TC (1999) Tunicate tails, stolons, and the origin of the vertebrate trunk. Biol Rev 74:177–198
Lacalli TC, Holland LZ (1998) The developing dorsal ganglion of the salp Thalia democratica, and the nature of the ancestral chordate brain. Philos Trans R Soc B 353:1943–1967. https://doi.org/10.1098/rstb.1998.0347
Lewis SL, Lyons DE, Meekins TL, Newcomb JM (2011) Serotonin influences locomotion in the nudibranch mollusc Melibe leonina. Biol Bull 220:155–160. https://doi.org/10.1086/BBLv220n3p155
Moret F, Guilland J-C, Coudouel S, Rochette L, Vernier P (2004) Distribution of tyrosine hydroxylase, dopamine, and serotonin in the central nervous system of amphioxus (Branchiostoma lanceolatum): implications for the evolution of catecholamine systems in vertebrates. J Comp Neurol 468:135–150. https://doi.org/10.1002/cne.10965
Nusbaum MP, Kristan WB (1986) Swim initiation in the leech by serotonin-containing interneurons, cells 21 and 61. J Exp Biol 122:277–302
Pennati R, Groppelli S, Sotgia C, Candiani S, Pestarino M, De Bernardi F (2001) Serotonin localization in Phallusia mammillata larvae and effects of 5-HT antagonists during larval development. Dev Growth Differ 43:647–656. https://doi.org/10.1046/j.1440-169X.2001.00608.x
Pennati R, Dell´Anna A, Zega G, De Bernardi F (2012) Immunohistochemical study of the nervous system of the tunicate Thalia democratica (Forsskal, 1775). Eur J Histochem 56:e16. https://doi.org/10.4081/ejh.2012.16
Piette J, Lemaire P (2015) Thaliaceans, the neglected pelagic relatives of ascidians: a developmental and evolutionary enigma. Q Rev Biol 90:117–145. https://doi.org/10.1086/681440
Pogue MG, Mickevich MF (1990) Character definitions and character state delineation: the bete noire of phylogenetic inference. Cladistics 6:319–361. https://doi.org/10.1111/j.1096-0031.1990.tb00549.x
Richter S, Loesel R, Purschke G, Schmidt-Rhaesa A, Scholtz G, Stach T, Vogt L, Wanninger A, Brenneis G, Doring C, Faller S, Fritsch M, Grobe P, Heuer C, Kaul S, Moller O, Muller C, Rieger V, Rothe B, Stegner M, Harzsch S (2010) Invertebrate neurophylogeny: suggested terms and definitions for a neuroanatomical glossary. Front Zool 7:29. https://doi.org/10.1186/1742-9994-7-29
Schindelin J, Arganda-Carreras I, Frise E, Kaynig V, Longair M, Pietzsch T, Preibisch S, Rueden C, Saalfeld S, Schmid B, Tinevez J-Y, White DJ, Hartenstein V, Eliceiri K, Tomancak P, Cardona A (2012) Fiji: an open-source platform for biological-image analysis. Nat Methods 9:676–682. https://doi.org/10.1038/nmeth.2019
Schmidt-Rhaesa A, Harzsch S, Purschke G (2015) Structure and evolution of invertebrate nervous systems. OUP, Oxford
Stach T (2005) Comparison of the serotonergic nervous system among Tunicata: implications for its evolution within Chordata. Org Divers Evol 5:15–24. https://doi.org/10.1016/j.ode.2004.05.004
Stach T, Turbeville JM (2002) Phylogeny of Tunicata inferred from molecular and morphological characters. Mol Phylogenet Evol 25:408–428. https://doi.org/10.1016/s1055-7903(02)00305-6
Stemme T, Stern M, Bicker G (2017) Serotonin-containing neurons in basal insects: in search of ground patterns among Tetraconata. J Comp Neurol 525:79–115. https://doi.org/10.1002/cne.24043
Trandaburu T, Trandaburu I (2007) Serotonin (5-hydroxytryptamine, 5-HT) immunoreactive endocrine and neural elements in the chromaffin enteropancreatic system of amphibians and reptiles. Acta Histochem 109:237–247. https://doi.org/10.1016/j.acthis.2006.10.005
Tsagkogeorga G, Turon X, Hopcroft RR, Tilak M-G, Feldstein T, Shenkar N, Loya Y, Huchon D, Douzery EJP, Delsuc F (2009) An updated 18S rRNA phylogeny of tunicates based on mixture and secondary structure models. BMC Evol Biol 9:187. https://doi.org/10.1186/1471-2148-9-187
Valero-Gracia A, Marino R, Crocetta F, Nittoli V, Tiozzo S, Sordino P (2016) Comparative localization of serotonin-like immunoreactive cells in Thaliacea informs tunicate phylogeny. Front Zool 13:45. https://doi.org/10.1186/s12983-016-0177-6
Wada Y, Mogami Y, Baba S (1997) Modification of ciliary beating in sea urchin larvae induced by neurotransmitters: beat-plane rotation and control of frequency fluctuation. J Exp Biol 200:9–18
Acknowledgements
The authors would like to thank Stefano Tiozzo for expert collection and fixation of specimens of Salpa fusiformis. We are thankful to the invaluable help of Woody Lee and Scott Jones from the Smithsonian Marine Station in Fort Pierce, Florida in securing specimens of Iasis cylindrica, Salpa fusiformis, Thalia democratica and Doliolum nationalis. We are also indebted to Prof. Valerie Paul and Prof. Mary Rice for generously providing access to the facilities of the Smithsonian Marine Station. We are much obliged to the scientific party and the crew of FS SONNE during the cruise SO258/1, especially to Dr. Nina Furchheim and Reinhard Werner for collecting and fixing specimens of Pyrostremma agassizi, Pyrosoma atlanticum, and Pyrosomella verticillata. We are grateful for the access to the Leica SPE CLSM granted by Dr. Carsten Lüter (Museum für Naturkunde Berlin). Funding by the Deutsche Forschungsgemeinschaft (DFG), the German Academic Exchange Service (DAAD), and the Elsa-Neumann-Stipendium des Landes Berlin is gratefully acknowledged.
Funding
This study was funded by the Deutsche Forschungsgemeinschaft (DFG) Grant STA655/4-1, the German Academic Exchange Service (DAAD), and the Elsa-Neumann-Stipendium des Landes Berlin.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest. This article does not contain any studies with human participants performed by any of the authors. All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Electronic supplementary material
Below is the link to the electronic supplementary material.
435_2018_416_MOESM1_ESM.pdf
Supplementary Fig.: Interactive 3d model of the internal anatomy of the blastozooid stage of D. nationalis and of P. atlanticum based on serial sections for light microscopy. A: Anatomy of D. nationalis, dorsal view anterior to the top. The ciliated funnel (cf) opens into the branchial basket (bb—not completely shown in the 3d reconstruction) in close spatial relation to the dorsal spiral of the peripharyngeal band (pb). B: Internal anatomy of P. atlanticum, view from the left side, anterior to the top. The pyloric gland (pg) is dendritically shaped. Originating from the stomach (st), the pyloric gland forms several branches that connect with the posterior intestinal wall. br: brain, en: endostyle, ep: epidermis, es: esophagus, ex: excurrent siphon, in: intestine, is: incurrent siphon, lo: light organ, ng: neural gland, ov: ovary, pe: pericardium, te: testis. (PDF 45876 KB)
Rights and permissions
About this article
Cite this article
Braun, K., Stach, T. Distribution and evolution of serotonin-like immunoreactive cells in Thaliacea (Tunicata). Zoomorphology 137, 565–578 (2018). https://doi.org/10.1007/s00435-018-0416-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00435-018-0416-9