Abstract
Purpose
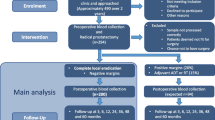
Prostate cancer (PCa) is one of the most common malignancies in men and one of the leading causes of cancer-related deaths; circulating tumor cells (CTC) are malignant cells that have broken off from original tumor or metastatic sites and extravasated into the blood vessels either naturally or maybe as a consequence of surgical procedures. This study aims to explore the feasibility of liquid biopsy technique to diagnose prostate cancer.
Method
We constructed an assay platform integrating magnetic separation and fluorescence in situ hybridization (FISH) to effectively capture prostate cancer CTCs and evaluate the distribution between healthy volunteers and prostate cancer patients, respectively.
Results
There was a significant difference in the number of CTCs between the healthy population and prostate cancer patients (P < 0.001). The results of the study showed that the CTCs capture identification system has good sensitivity and specificity in identifying prostate cancer patients.
Conclusion
The CTCs test allows us to accurately identify patients who are at high risk for prostate cancer, allowing for early intervention and treating patients effectively.






Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Adams DL, Martin SS, Alpaugh RK, Charpentier M, Tsai S, Bergan RC, Ogden IM, Catalona W, Chumsri S, Tang CM, Cristofanilli M (2014) Circulating giant macrophages as a potential biomarker of solid tumors. Proc Natl Acad Sci USA 111(9):3514–3519
Alix-Panabières C, Pantel K (2014) Challenges in circulating tumour cell research. Nat Rev Cancer 14(9):623–631
Allard WJ, Matera J, Miller MC, Repollet M, Connelly MC, Rao C, Tibbe AGJ, Uhr JW, Terstappen LWMM (2004) Tumor cells circulate in the peripheral blood of all major carcinomas but not in healthy subjects or patients with nonmalignant diseases. Clin Cancer Res 10(20):6897–6904
Antonarakis ES et al (2014) AR-V7 and resistance to enzalutamide and abiraterone in prostate cancer. N Engl J Med 371:1028–1038
Davis JW, Nakanishi H, Kumar VS, Bhadkamkar VA, McCormack R, Fritsche HA, Handy B, Gornet T, Babaian RJ (2008) Circulating tumor cells in peripheral blood samples from patients with increased serum prostate specific antigen: initial results in early prostate cancer. J Urol 179(6):2187–2191
Eisenwort G, Jurkin J, Yasmin N, Bauer T, Gesslbauer B, Strobl H (2011) Identification of TROP2 (TACSTD2), an EpCAM-like molecule, as a specific marker for TGF-β1-dependent human epidermal Langerhans cells. J Invest Dermatol 131(10):2049–2057
Gao Y et al (2016) Clinical significance of pancreatic circulating tumor cells using combined negative enrichment and immunostaining-fluorescence in situ hybridization. J Exp Clin Cancer Res 35:66
Habermann JK, Doering J, Hautaniemi S, Roblick UJ, Bündgen NK, Nicorici D, Kronenwett U, Rathnagiriswaran S, Mettu RKR, Ma Y, Krüger S, Bruch HP, Auer G, Guo NL, Ried T (2009) The gene expression signature of genomic instability in breast cancer is an independent predictor of clinical outcome. Int J Cancer 124(7):1552–1564
Han Y, Luo Y, Wang Y, Chen Y, Li M, Jiang Y (2016) Hepatocyte growth factor increases the invasive potential of PC-3 human prostate cancer cells via an ERK/MAPK and Zeb-1 signaling pathway. Oncol Lett 11(1):753–759
Helo P, Cronin AM, Danila DC, Wenske S, Gonzalez-Espinoza R, Anand A, Koscuiszka M, Väänänen RM, Pettersson K, Chun FKH, Steuber T, Huland H, Guillonneau BD, Eastham JA, Scardino PT, Fleisher M, Scher HI, Lilja H (2009) Circulating prostate tumor cells detected by reverse transcription-PCR in men with localized or castration-refractory prostate cancer: concordance with cell search assay and association with bone metastases and with survival. Clin Chem 55(4):765–773
Hristozova T et al (2011) The presence of circulating tumor cells (CTCs) correlates with lymph node metastasis in nonresectable squamous cell carcinoma of the head and neck region (SCCHN). Ann Oncol 22:1878–1885
Huang XY, Li F, Li TT, Zhang JT, Shi XJ, Huang XY, Zhou J, Tang ZY, Huang ZL (2023) A clinically feasible circulating tumor cell sorting system for monitoring the progression of advanced hepatocellular carcinoma. J Nanobiotechnology 21(1):25
Kiciński M, Vangronsveld J, Nawrot TS (2011) An epidemiological reappraisal of the familial aggregation of prostate cancer: a meta-analysis. PLoS One 6(10):e27130
Kops GJPL, Weaver BAA, Cleveland DW (2005) On the road to cancer: aneuploidy and the mitotic checkpoint. Nat Rev Cancer 5(10):773–785
Lee AJX, Endesfelder D, Rowan AJ, Walther A, Birkbak NJ, Futreal PA, Downward J, Szallasi Z, Tomlinson IPM, Howell M, Kschischo M, Swanton C (2011) Chromosomal instability confers intrinsic multidrug resistance. Cancer Res 71(5):1858–1870
Lin PP (2015) Integrated EpCAM-independent subtraction enrichment and iFISH strategies to detect and classify disseminated and circulating tumors cells. Clin Transl Med 4:38
Liu Y, Li Q, Chen T, Shen T, Zhang X, Song P, Liu L, Liu J, Jiang T, Liang X (2021) Clinical verification of vimentin/EpCAM immunolipid magnetic sorting system in monitoring CTCs in arterial and venous blood of advanced tumor. J Nanobiotechnology 19(1):185
Liu C et al (2021) The value of circulating tumor cells with positive centromere probe 8 in the diagnosis of small pulmonary nodules. Transl Oncol 14:101052
Lojanapiwat B, Anutrakulchai W, Chongruksut W, Udomphot C (2014) Correlation and diagnostic performance of the prostate-specific antigen level with the diagnosis, aggressiveness, and bone metastasis of prostate cancer in clinical practice. Prostate Int 2(3):133–139
Lu X et al (2021) Evaluation of a new magnetic bead as an integrated platform for systematic CTC recognition, capture and clinical analysis. Colloids Surf B Biointerfaces 199:111542
McDaniel AS, Ferraldeschi R, Krupa R, Landers M, Graf R, Louw J, Jendrisak A, Bales N, Marrinucci D, Zafeiriou Z, Flohr P, Sideris S, Crespo M, Figueiredo I, Mateo J, de Bono JS, Dittamore R, Tomlins SA, Attard G (2017) Phenotypic diversity of circulating tumour cells in patients with metastatic castration-resistant prostate cancer. BJU Int 120(5B):E30-44
McGranahan N, Burrell RA, Endesfelder D, Novelli MR, Swanton C (2012) Cancer chromosomal instability: therapeutic and diagnostic challenges. EMBO Rep 13(6):528–538
Miyamoto DT, Lee RJ, Stott SL, Ting DT, Wittner BS, Ulman M, Smas ME, Lord JB, Brannigan BW, Trautwein J, Bander NH, Wu CL, Sequist LV, Smith MR, Ramaswamy S, Toner M, Maheswaran S, Haber DA (2012) Androgen receptor signaling in circulating tumor cells as a marker of hormonally responsive prostate cancer. Cancer Discov 2(11):995–1003
O’Rourke ME (2011) The prostate-specific antigen screening conundrum: examining the evidence. Semin Oncol Nurs 27(4):251–259
Passerini V, Ozeri-Galai E, de Pagter MS, Donnelly N, Schmalbrock S, Kloosterman WP, Kerem B, Storchová Z (2016) The presence of extra chromosomes leads to genomic instability. Nat Commun 7:10754
Pisapia P, Malapelle U, Troncone G (2019) Liquid biopsy and lung cancer. Acta Cytol 63:489–496
Potosky AL, Miller BA, Albertsen PC, Kramer BS (1995) The role of increasing detection in the rising incidence of prostate cancer. JAMA 273(7):548–552
Saini S (2016) PSA and beyond: alternative prostate cancer biomarkers. Cell Oncol (Dordr) 39(2):97–106
Scher HI, Morris MJ, Larson S, Heller G (2013) Validation and clinical utility of prostate cancer biomarkers. Nat Rev Clin Oncol 10(4):225–234
Scher HI, Lu D, Schreiber NA, Louw J, Graf RP, Vargas HA, Johnson A, Jendrisak A, Bambury R, Danila D, McLaughlin B, Wahl J, Greene SB, Heller G, Marrinucci D, Fleisher M, Dittamore R (2016) Association of AR-V7 on circulating tumor cells as a treatment-specific biomarker with outcomes and survival in castration-resistant prostate cancer. JAMA Oncol 2(11):1441–1449
Sharma S, Zapatero-Rodríguez J, O’Kennedy R (2017) Prostate cancer diagnostics: clinical challenges and the ongoing need for disruptive and effective diagnostic tools. Biotechnol Adv 35(2):135–149
Sharma S, Zhuang R, Long M, Pavlovic M, Kang Y, Ilyas A, Asghar W (2018) Circulating tumor cell isolation, culture, and downstream molecular analysis. Biotechnol Adv 36(4):1063–1078
Shen MM, Abate-Shen C (2010) Molecular genetics of prostate cancer: new prospects for old challenges. Genes Dev 24(18):1967–2000
Shetye JD, Liljefors ML, Emdin SO, Frödin JE, Strigård K, Mellstedt HT, Porwit A (2004) Spectrum of cytokeratin-positive cells in the bone marrows of colorectal carcinoma patients. Anticancer Res 24(4):2375–2383
Thompson IM, Goodman PJ, Tangen CM, Lucia MS, Miller GJ, Ford LG, Lieber MM, Cespedes RD, Atkins JN, Lippman SM, Carlin SM, Ryan A, Szczepanek CM, Crowley JJ, Coltman CA (2003) The influence of finasteride on the development of prostate cancer. N Engl J Med 349(3):215–224
Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A (2015) Global cancer statistics, 2012. CA Cancer J Clin 65(2):87–108
Toss A, Mu Z, Fernandez S, Cristofanilli M (2014) CTC enumeration and characterization: moving toward personalized medicine. Ann Transl Med 2:108
Wicha MS, Hayes DF (2011) Circulating tumor cells: not all detected cells are bad and not all bad cells are detected. J Clin Oncol 29:1508–1511
Yang Z, Bai H, Hu L, Kong D, Li G, Zhao C, Feng L, Cheng S, Shou J, Zhang W, Zhang K (2022) Improving the diagnosis of prostate cancer by telomerase-positive circulating tumor cells: a prospective pilot study. EClinicalMedicine 43:101161
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
JC, TX and JY wrote the manuscript, XL collected and analyzed the data, LH and SS were in charge of the methodology and conception, JD administrated the work. All authors reviewed and approved the submitted version of the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chen, J., Xie, T., Yang, J. et al. Feasibility study of expressing epcam + /vimentin + CTC in prostate cancer diagnosis. J Cancer Res Clin Oncol 149, 8699–8709 (2023). https://doi.org/10.1007/s00432-023-04819-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-023-04819-7