Abstract
Purpose
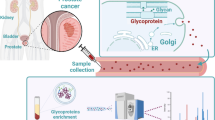
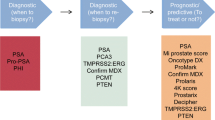
Markers are needed to increase the diagnostic accuracy of prostate-specific antigen (PSA) in prostate cancer (PCa) screening. Mounting evidence has shown that plasma proteins can be hopeful biomarkers for cancer diagnosis.
Methods
Tandem mass tag (TMT)-based proteomics and parallel reaction monitoring (PRM) analysis were used to screen the differential proteins and further validated in other independent studies (n = 539). Receiver-operating characteristic (ROC), decision curves and nomograms were applied to assess the diagnostic accuracy of biomarkers.
Results
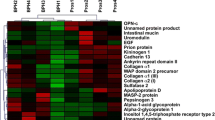
Three candidate proteins (DBP, LCAT and ORM2) were preliminarily screened. Subsequent validation studies revealed significant upregulation of ORM2 in PCa patients across other independent cohorts. ORM2 yielded excellent discriminative power for PCa from benign prostatic hyperplasia (BPH) patients (AUC = 0.861 and 0.814 in validation phases 2a and 2b, respectively). Importantly, the combination of ORM2 and PSA gave better predictive accuracy than PSA alone. We incorporated age, PSA and ORM2 into a nomogram, which yielded C-index of 0.883 in validation phase 2a. A similar C-index of 0.879 was obtained in external validation phase 2b.
Conclusions
In summary, our study suggests that ORM2 could be treated as a complementary biomarker for PSA in distinguishing PCa from BPH.





Similar content being viewed by others
Data availability statement
The data used and/or analyzed in this study are available on request from the corresponding author.
Abbreviations
- PSA:
-
Prostate-specific antigen
- PCa:
-
Prostate cancer
- TMT:
-
Tandem mass tag
- PRM:
-
Parallel reaction monitoring
- ROC:
-
Receiver-operating characteristic
- BPH:
-
Benign prostatic hyperplasia
- ELISA:
-
Enzyme-linked immunosorbent assay
- TRUS:
-
Transrectal ultrasound
- DRE:
-
Digital rectal examination
- MRI:
-
Magnetic resonance imaging
- TTAH:
-
The Third Affiliated Hospital of Soochow University
- YPH:
-
Yixing People’s Hospital
- AGC:
-
Automatic gain control
- HCD:
-
Higher energy collisional dissociation
- AUC:
-
The area under the curve
- 95% CI:
-
95% Confidence interval
- DCA:
-
Decision curve analysis
- PCA:
-
Principal component analysis
- ORM2:
-
Orosomucoid 2
- PDAC:
-
Pancreatic ductal adenocarcinoma
- THBS2:
-
Plasma protein thrombospondin-2
- AFP:
-
Alpha-fetoprotein
References
Bachtiar I et al (2009) Combination of alpha-1-acid glycoprotein and alpha-fetoprotein as an improved diagnostic tool for hepatocellular carcinoma. Clin Chim Acta 399:97–101. https://doi.org/10.1016/j.cca.2008.09.024
Borrebaeck CA (2017) Precision diagnostics: moving towards protein biomarker signatures of clinical utility in cancer. Nat Rev Cancer 17:199–204. https://doi.org/10.1038/nrc.2016.153
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1006/abio.1976.9999
Cao WQ et al (2019) Straightforward and highly efficient strategy for hepatocellular carcinoma glycoprotein biomarker discovery using a nonglycopeptide-based mass spectrometry pipeline. Anal Chem 91:12435–12443. https://doi.org/10.1021/acs.analchem.9b03074
Ceciliani F, Lecchi C (2019) The immune functions of alpha1 acid glycoprotein. Curr Protein Pept Sci 20:505–524. https://doi.org/10.2174/1389203720666190405101138
Cortese R et al (2012) Epigenetic markers of prostate cancer in plasma circulating DNA. Hum Mol Genet 21:3619–3631. https://doi.org/10.1093/hmg/dds192
Cucchiara V, Cooperberg MR, Dall’Era M, Lin DW, Montorsi F, Schalken JA, Evans CP (2018) Genomic markers in prostate cancer decision making. Eur Urol 73:572–582. https://doi.org/10.1016/j.eururo.2017.10.036
Gao F, Zhang X, Whang S, Zheng C (2014) Prognostic impact of plasma ORM2 levels in patients with stage II colorectal cancer. Ann Clin Lab Sci 44:388–393
Gulati R, Morgan TM, A’Mar T, Psutka SP, Tosoian JJ, Etzioni R (2020) Overdiagnosis and lives saved by reflex testing men with intermediate prostate-specific antigen levels. J Natl Cancer Inst 112:384–390. https://doi.org/10.1093/jnci/djz127
Ilic D et al (2018) Prostate cancer screening with prostate-specific antigen (PSA) test: a systematic review and meta-analysis. BMJ 362:k3519. https://doi.org/10.1136/bmj.k3519
Jones AL et al (2021) Prostate cancer diagnosis in the clinic using an 8-protein biomarker panel. Anal Chem 93:1059–1067. https://doi.org/10.1021/acs.analchem.0c04034
Kim J et al (2017) Detection of early pancreatic ductal adenocarcinoma with thrombospondin-2 and CA19–9 blood markers. Sci Transl Med. https://doi.org/10.1126/scitranslmed.aah5583
Kohaar I, Petrovics G, Srivastava S (2019) A rich array of prostate cancer molecular biomarkers: opportunities and challenges. Int J Mol Sci. https://doi.org/10.3390/ijms20081813
Landegren U, Hammond M (2020) Cancer diagnostics based on plasma protein biomarkers: hard times but great expectations. Mol Oncol. https://doi.org/10.1002/1878-0261.12809
Lin K, Lipsitz R, Miller T, Janakiraman S, Force USPST (2008) Benefits and harms of prostate-specific antigen screening for prostate cancer: an evidence update for the U.S. Preventive Services Task Force. Ann Intern Med 149:192–199. https://doi.org/10.7326/0003-4819-149-3-200808050-00009
Liu S et al (2020) Discovery of potential serum protein biomarkers in ankylosing spondylitis using tandem mass tag-based quantitative proteomics. J Proteome Res 19:864–872. https://doi.org/10.1021/acs.jproteome.9b00676
Luo Z, Lei H, Sun Y, Liu X, Su DF (2015) Orosomucoid, an acute response protein with multiple modulating activities. J Physiol Biochem 71:329–340. https://doi.org/10.1007/s13105-015-0389-9
Mann M, Kulak NA, Nagaraj N, Cox J (2013) The coming age of complete, accurate, and ubiquitous proteomes. Mol Cell 49:583–590. https://doi.org/10.1016/j.molcel.2013.01.029
Nagaraj N et al (2011) Deep proteome and transcriptome mapping of a human cancer cell line. Mol Syst Biol 7:548. https://doi.org/10.1038/msb.2011.81
Rucksaken R, Charoensuk L, Pinlaor P, Pairojkul C, Khuntikeo N, Pinlaor S (2017) Plasma orosomucoid 2 as a potential risk marker of cholangiocarcinoma. Cancer Biomark 18:27–34. https://doi.org/10.3233/CBM-160670
Saini S (2016) PSA and beyond: alternative prostate cancer biomarkers. Cell Oncol (dordr) 39:97–106. https://doi.org/10.1007/s13402-016-0268-6
Scher HI et al (2015) Circulating tumor cell biomarker panel as an individual-level surrogate for survival in metastatic castration-resistant prostate cancer. J Clin Oncol 33:1348–1355. https://doi.org/10.1200/JCO.2014.55.3487
Schroder FH et al (2014) Screening and prostate cancer mortality: results of the European Randomised Study of Screening for Prostate Cancer (ERSPC) at 13 years of follow-up. Lancet 384:2027–2035. https://doi.org/10.1016/S0140-6736(14)60525-0
Schwanhausser B et al (2011) Global quantification of mammalian gene expression control. Nature 473:337–342. https://doi.org/10.1038/nature10098
Schwenk JM et al (2017) The human plasma proteome draft of 2017: building on the human plasma PeptideAtlas from mass spectrometry and complementary assays. J Proteome Res 16:4299–4310. https://doi.org/10.1021/acs.jproteome.7b00467
Siegel R, Naishadham D, Jemal A (2013) Cancer statistics, 2013 CA Cancer. J Clin 63:11–30. https://doi.org/10.3322/caac.21166
Sorensson J, Matejka GL, Ohlson M, Haraldsson B (1999) Human endothelial cells produce orosomucoid, an important component of the capillary barrier. Am J Physiol 276:H530-534. https://doi.org/10.1152/ajpheart.1999.276.2.H530
Stastna M, Van Eyk JE (2012) Secreted proteins as a fundamental source for biomarker discovery. Proteomics 12:722–735. https://doi.org/10.1002/pmic.201100346
Vickers AJ (2017) Prostate cancer screening: time to question how to optimize the ratio of benefits and harms. Ann Intern Med 167:509–510. https://doi.org/10.7326/M17-2012
Zhang X et al (2012) The potential role of ORM2 in the development of colorectal cancer. PLoS ONE 7:e31868. https://doi.org/10.1371/journal.pone.0031868
Zhang B et al (2014) Proteogenomic characterization of human colon and rectal cancer. Nature 513:382–387. https://doi.org/10.1038/nature13438
Funding
This work was supported by the Natural Science Foundation of Jiangsu Province (BK20190600; BK20190555), National Natural Science Foundation of China (81903390; 81802880), Huai’an City Science and Technology Project (HAB202051), and the Youth Talent Science and Technology Project of Changzhou Health Commission (QN202110).
Author information
Authors and Affiliations
Contributions
Study conception and design: GM, XY, LC, QW, and YG. Acquisition of data: BX, HC, WJ, XL, YF, and QO. Analysis and interpretation of data: YG and QO. Drafting of the manuscript: YG. Critical revision for important intellectual content: QW, BX, GM, and XY. Obtained funding: GM, YG, QS, and QY. Administrative, technical or material support: QY and YS. Study supervision: GM. Final approval of the manuscript: all the authors.
Corresponding authors
Ethics declarations
Conflict of interest
The authors have declared that no competing interest exists.
Ethical approval
This study was approved by the Institutional Review Board of each participating institution. All the participants provided their informed consent to participate.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ge, Y., Xu, B., Cai, H. et al. Diagnostic role of plasma ORM2 in differentiating prostate cancer from benign prostatic hyperplasia. J Cancer Res Clin Oncol 149, 2301–2310 (2023). https://doi.org/10.1007/s00432-022-04380-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-022-04380-9