Abstract
Purpose
To investigate the clinical characteristics and prognostic factors of natural killer/T-cell lymphoma (NKTCL).
Methods
We retrospectively reviewed 410 NKTCL patients admitted to our lymphoma center from 2000 to 2019. Overall survival (OS) and progression-free survival (PFS) were estimated with the Kaplan–Meier method, and the differences between the study groups were compared by the log-rank test.
Results
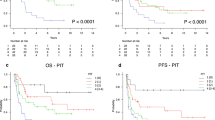
The median age of the 410 patients was 44 (range 8–84), and the 5-year OS and PFS were 61.2% and 38.4%, respectively. For patients with stage I/II, the 5-year PFS rate was 57.5%, and the 5-year OS rate was 77.2%. For patients with stage III/IV, the 5-year PFS rate was 17.4%, and the 5-year OS rate was 43.7%. Compared to the patients who received radiotherapy alone or chemotherapy alone as their initial treatment, the patients who received combined chemoradiotherapy had longer PFS (P = 0.013). Independent prognostic factors for OS were stage III/IV (P = 0.001), elevated IPI/aaIPI score (P = 0.019), elevated PINK score (P < 0.001) and elevated plasma EBV-DNA (P = 0.003). An elevated PINK score (P < 0.001) was an independent prognostic factor for PFS.
Conclusion
Stage III/IV, elevated IPI/aaIPI score, elevated PINK score and elevated plasma EBV-DNA were independent prognostic factors for OS. Elevated PINK score was an independent prognostic factor for PFS. In stage III/IV patients, the patients who received combined chemoradiotherapy had significantly longer PFS.


Similar content being viewed by others
Data availability statement
Only publicly available data were used in this study, and data sources and handling of these data are described in the Materials and Methods. Further information is available from the corresponding author upon request.
References
Aeppli S, Driessen C, Graf L, Hitz F (2018) Systemic treatment of a patient with relapsed and refractory extranodal NK/T-cell lymphoma (ENKL) and meningeosis leukemica with daratumumab. Hematol Oncol 36(4):713–714. https://doi.org/10.1002/hon.2533
Asif S, Begemann M, Bennett J, Fatima R, Masood A, Raza S (2019) Pembrolizumab in newly diagnosed EBV-negative extranodal natural killer/T-cell lymphoma: A case report. Mol Clin Oncol 10(3):397–400. https://doi.org/10.3892/mco.2019.1805
Au WY, Pang A, Choy C, Chim CS, Kwong YL (2004) Quantification of circulating Epstein-Barr virus (EBV) DNA in the diagnosis and monitoring of natural killer cell and EBV-positive lymphomas in immunocompetent patients. Blood 104(1):243–249. https://doi.org/10.1182/blood-2003-12-4197
Bi XW, Li YX, Fang H et al (2013) High-dose and extended-field intensity modulated radiation therapy for early-stage NK/T-cell lymphoma of Waldeyer’s ring: dosimetric analysis and clinical outcome[J]. Int J Radiat Oncol Biol Phys 87(5):1086–1093
Chan KC, Zhang J, Chan AT, Lei KI, Leung SF, Chan LY, Chow KC, Lo YM (2003) Molecular characterization of circulating EBV DNA in the plasma of nasopharyngeal carcinoma and lymphoma patients. Cancer Res 63(9):2028–2032
Cheson BD, Fisher RI, Barrington SF et al (2014) Recommendations for initial evaluation, staging, and response assessment of Hodgkin and non-Hodgkin lymphoma: the Lugano classification. J Clin Oncol 32(27):3059–3068. https://doi.org/10.1200/JCO.2013.54.8800
de Weers M, Tai YT, van der Veer MS, Bakker JM, Vink T, Jacobs DC, Oomen LA, Peipp M, Valerius T, Slootstra JW, Mutis T, Bleeker WK, Anderson KC, Lokhorst HM, van de Winkel JG, Parren PW (2011) Daratumumab, a novel therapeutic human CD38 monoclonal antibody, induces killing of multiple myeloma and other hematological tumors. J Immunol 186(3):1840–1848. https://doi.org/10.4049/jimmunol.1003032
Deutsch YE, Tadmor T, Podack ER, Rosenblatt JD (2011) CD30: an important new target in hematologic malignancies. Leuk Lymphoma 52(9):1641–1654. https://doi.org/10.3109/10428194.2011.574761
Gallagher A, Armstrong AA, MacKenzie J, Shield L, Khan G, Lake A, Proctor S, Taylor P, Clements GB, Jarrett RF (1999) Detection of Epstein-Barr virus (EBV) genomes in the serum of patients with EBV-associated Hodgkin’s disease. Int J Cancer 84(4):442–448. https://doi.org/10.1002/(sici)1097-0215(19990820)84:4%3c442::aid-ijc20%3e3.0.co;2-j
Gandhi MK, Lambley E, Burrows J, Dua U, Elliott S, Shaw PJ, Prince HM, Wolf M, Clarke K, Underhill C, Mills T, Mollee P, Gill D, Marlton P, Seymour JF, Khanna R (2006) Plasma Epstein-Barr virus (EBV) DNA is a biomarker for EBV-positive Hodgkin’s lymphoma. Clin Cancer Res 12(2):460–464. https://doi.org/10.1158/1078-0432.CCR-05-2008
Hari P, Raj RV, Olteanu H (2016) Targeting CD38 in refractory extranodal natural killer cell-T-cell lymphoma. N Engl J Med 375(15):1501–1502. https://doi.org/10.1056/NEJMc1605684
Hong H, Li Y, Lim ST et al (2020) A proposal for a new staging system for extranodal natural killer T-cell lymphoma: a multicenter study from China and Asia Lymphoma Study Group. Leukemia 34(8):2243–2248. https://doi.org/10.1038/s41375-020-0740-1
Horning SJ, Weller E, Kim K et al (2004) Chemotherapy with or without radiotherapy in limited-stage diffuse aggressive non-Hodgkin′s lymphoma: Eastern Cooperative Oncology Group study. J Clin Oncol 22(15):3032–3038. https://doi.org/10.1200/JCO.2004.06.088
Hu B, Oki Y (2018) Novel immunotherapy options for extranodal NK/T-cell lymphoma. Front Oncol 8:139. https://doi.org/10.3389/fonc.2018.00139
Kharfan-Dabaja MA, Kumar A, Ayala E et al (2017) Clinical practice recommendations on indication and timing of hematopoietic cell transplantation in mature T Cell and NK/T Cell lymphomas: an international collaborative effort on behalf of the guidelines committee of the american society for blood and marrow transplantation. Biol Blood Marrow Transplant 23(11):1826–1838. https://doi.org/10.1016/j.bbmt.2017.07.027
Kim HK, Moon SM, Moon JH, Park JE, Byeon S, Kim WS (2015) Complete remission in CD30-positive refractory extranodal NK/T-cell lymphoma with brentuximab vedotin. Blood Res 50(4):254–256. https://doi.org/10.5045/br.2015.50.4.254
Kim SJ, Yoon DH, Jaccard A, Chng WJ, Lim ST, Hong H, Park Y, Chang KM, Maeda Y, Ishida F, Shin DY, Kim JS, Jeong SH, Yang DH, Jo JC, Lee GW, Choi CW, Lee WS, Chen TY, Kim K, Jung SH, Murayama T, Oki Y, Advani R, d’Amore F, Schmitz N, Suh C, Suzuki R, Kwong YL, Lin TY, Kim WS (2016) A prognostic index for natural killer cell lymphoma after non-anthracycline-based treatment: a multicentre, retrospective analysis. Lancet Oncol 17(3):389–400. https://doi.org/10.1016/S1470-2045(15)00533-1
Kim TM, Lee SY, Jeon YK et al (2008) Clinical heterogeneity of extranodal NK/T-cell lymphoma, nasal type: a national survey of the Korean Cancer Study Group. Ann Oncol 19(8):1477–1484. https://doi.org/10.1093/annonc/mdn147
Lee J, Park YH, Kim WS, Lee SS, Ryoo BY, Yang SH, Park KW, Kang JH, Park JO, Lee SH, Kim K, Jung CW, Park YS, Im YH, Kang WK, Lee MH, Ko YH, Ahn YC, Park K (2005) Extranodal nasal type NK/T-cell lymphoma: elucidating clinical prognostic factors for risk-based stratification of therapy. Eur J Cancer 41(10):1402–1408. https://doi.org/10.1016/j.ejca.2005.03.010
Lee J, Suh C, Park YH et al (2006) Extranodal natural killer T-cell lymphoma, nasal-type: a prognostic model from a retrospective multicenter study. J Clin Oncol 24(4):612–618. https://doi.org/10.1200/JCO.2005.04.1384
Lei KI, Chan LY, Chan WY, Johnson PJ, Lo YM (2000) Quantitative analysis of circulating cell-free Epstein-Barr virus (EBV) DNA levels in patients with EBV-associated lymphoid malignancies. Br J Haematol 111(1):239–246. https://doi.org/10.1046/j.1365-2141.2000.02344.x
Lei KI, Chan LY, Chan WY, Johnson PJ, Lo YM (2002) Diagnostic and prognostic implications of circulating cell-free Epstein-Barr virus DNA in natural killer/T-cell lymphoma. Clin Cancer Res 8(1):29–34
Li L, Zhang C, Zhang L, Li X, Wu JJ, Sun ZC, Fu XR, Wang XH, Chang Y, Wang R, Qiu YJ, Zhang MZ (2014) Efficacy of a pegaspargase-based regimen in the treatment of newly-diagnosed extranodal natural killer/T-cell lymphoma. Neoplasma 61(2):225–232. https://doi.org/10.4149/neo_2014_029
Li X, Cui Y, Sun Z et al (2016a) DDGP versus SMILE in newly diagnosed advanced natural killer/T-cell lymphoma: a randomized controlled, multicenter, open-label study in China. Clin Cancer Res 22(21):5223–5228. https://doi.org/10.1158/1078-0432.CCR-16-0153
Li X, Cui Y, Sun Z, Zhang L, Li L, Wang X, Wu J, Fu X, Ma W, Zhang X, Chang Y, Nan F, Li W, Su L, Wang J, Xue H, Zhang M (2016b) DDGP versus SMILE in newly diagnosed advanced natural killer/T-cell lymphoma: a randomized controlled, multicenter, open-label Study in China. Clin Cancer Res 22(21):5223–5228. https://doi.org/10.1158/1078-0432.CCR-16-0153
Li X, Cheng Y, Zhang M, Yan J, Li L, Fu X, Zhang X, Chang Y, Sun Z, Yu H, Zhang L, Wang X, Wu J, Li Z, Nan F, Tian L, Li W, Young KH (2018) Activity of pembrolizumab in relapsed/refractory NK/T-cell lymphoma. J Hematol Oncol 11(1):15. https://doi.org/10.1186/s13045-018-0559-7
Lin NJ, Song YQ, Zheng W et al (2015) Non-upper aerodigestive tract NK/T-cell lymphoma: an analysis of clinical features and survival from a single center in China [J]. Chin J Hematol 36(1):29–33. https://doi.org/10.3760/cma.j.issn.0253-2727.2015.01.007
Liu XH, Qin Y, Xu XM et al (2020) Clinical features and prognostic analysis of extranodal nasal type NK/T cell lymphoma [J]. Leuk Lymphoma 29(05):265–270. https://doi.org/10.3760/cma.j.cn115356-20190924-00189
Lv K, Li X, Yu H, Chen X, Zhang M, Wu X (2020) Selection of new immunotherapy targets for NK/T cell lymphoma. Am J Transl Res 12(11):7034–7047
Miyake MM, Oliveira MV, Miyake MM, Garcia JO, Granato L (2014) Clinical and otorhinolaryngological aspects of extranodal NK/T cell lymphoma, nasal type. Braz J Otorhinolaryngol 80(4):325–329. https://doi.org/10.1016/j.bjorl.2014.05.013
Müller P, Martin K, Theurich S, Schreiner J, Savic S, Terszowski G, Lardinois D, Heinzelmann-Schwarz VA, Schlaak M, Kvasnicka HM, Spagnoli G, Dirnhofer S, Speiser DE, von Bergwelt-Baildon M, Zippelius A (2014) Microtubule-depolymerizing agents used in antibody-drug conjugates induce antitumor immunity by stimulation of dendritic cells. Cancer Immunol Res 2(8):741–755. https://doi.org/10.1158/2326-6066.CIR-13-0198
Perry AM, Diebold J, Nathwani BN et al (2016) Non-Hodgkin lymphoma in the developing world: review of 4539 cases from the International Non-Hodgkin Lymphoma Classification Project. Haematologica 101(10):1244–1250. https://doi.org/10.3324/haematol.2016.148809
Poon LM, Kwong YL (2016) Complete remission of refractory disseminated NK/T cell lymphoma with brentuximab vedotin and bendamustine. Ann Hematol 95(5):847–849. https://doi.org/10.1007/s00277-016-2627-9
Su YJ, Wang PN, Chang H, Shih LY, Lin TL, Kuo MC, Chuang WY, Wu JH, Tang TC, Hung YS, Dunn P, Kao HW (2018) Extranodal NK/T-cell lymphoma, nasal type: Clinical features, outcome, and prognostic factors in 101 cases. Eur J Haematol 101(3):379–388. https://doi.org/10.1111/ejh.13126
Suzuki R, Yamaguchi M, Izutsu K, Yamamoto G, Takada K, Harabuchi Y, Isobe Y, Gomyo H, Koike T, Okamoto M, Hyo R, Suzumiya J, Nakamura S, Kawa K, Oshimi K; NK-cell Tumor Study Group. Prospective measurement of Epstein-Barr virus-DNA in plasma and peripheral blood mononuclear cells of extranodal NK/T-cell lymphoma, nasal type. Blood. 2011;118(23):6018–22. https://doi.org/10.1182/blood-2011-05-354142.
Tse E, Kwong YL (2017) The diagnosis and management of NK/T-cell lymphomas. J Hematol Oncol 10(1):85. https://doi.org/10.1186/s13045-017-0452-9
Tse E, Kwong YL (2016) Diagnosis and management of extranodal NK/T cell lymphoma nasal type. Expert Rev Hematol 9(9):861–871. https://doi.org/10.1080/17474086.2016.1206465
Wang B, Li XQ, Ma X, Hong X, Lu H, Guo Y (2008) Immunohistochemical expression and clinical significance of P-glycoprotein in previously untreated extranodal NK/T-cell lymphoma, nasal type. Am J Hematol 83(10):795–799. https://doi.org/10.1002/ajh.21256
Wang C, Li W, Liu CS et al (2015) Clinicopathological features and prognositic analysis of Epstein-Barr virus-positive diffuse large B-cell lymphoma in elderly: 7 cases report and literatures review [J]. Chin J Hematol 36(8):698–701. https://doi.org/10.3760/cma.j.issn.0253-2727.2015.08.016
Yamaguchi M, Suzuki R, Oguchi M (2018a) Advances in the treatment of extranodal NK/T-cell lymphoma, nasal type. Blood 131(23):2528–2540. https://doi.org/10.1182/blood-2017-12-791418
Yamaguchi M, Oguchi M, Suzuki R (2018b) Extranodal NK/T-cell lymphoma: Updates in biology and management strategies. Best Pract Res Clin Haematol 31(3):315–321. https://doi.org/10.1016/j.beha.2018.07.002
Yang JJ, Sang W et al (2019) Research progress on targeted therapies of Epstein-Barr virus-associated lymphomas[J]. Chin J Hematol 40(3):261–264. https://doi.org/10.3760/cma.j.issn.0253-2727.2019.03.021
Yao B, LiY X, Song YW et al (2006) Treatment option and outcome for patients with primary non-Hodgkin′s lymphoma of the nasal cavity. Chin J Oncol 28(1):58–61. https://doi.org/10.3760/j.issn:0253-3766.2006.01.016
Younes A, Gopal AK, Smith SE, Ansell SM, Rosenblatt JD, Savage KJ, Ramchandren R, Bartlett NL, Cheson BD, de Vos S, Forero-Torres A, Moskowitz CH, Connors JM, Engert A, Larsen EK, Kennedy DA, Sievers EL, Chen R (2012) Results of a pivotal phase II study of brentuximab vedotin for patients with relapsed or refractory Hodgkin’s lymphoma. J Clin Oncol 30(18):2183–2189. https://doi.org/10.1200/JCO.2011.38.0410
Yuan ZY, LiY X, Zhao LJ et al (2004) Clinical features, treatment and prognosis of 136 patients with primary non-Hodgkin′s lymphoma of the nasopharynx. Chin J Oncol 26(7):425–429. https://doi.org/10.3760/j.issn:0253-3766.2004.07.013
Zhang L, Li S, Jia S, Nan F, Li Z, Cao J, Fan S, Zhang C, Su L, Wang J, Xue H, Zhang M. The DDGP (cisplatin, dexamethasone, gemcitabine, and pegaspargase) regimen for treatment of extranodal natural killer (NK)/T-cell lymphoma, nasal type. Oncotarget. 2016;7(36):58396–58404. https://doi.org/10.18632/oncotarget.11135
Zhang L, Li S, Jia S, et al. The DDGP (cisplatin, dexamethasone, gemcitabine, and pegaspargase) regimen for treatment of extranodal natural killer (NK)/T-cell lymphoma, nasal type. Oncotarget. 2016;7(36):58396–58404. https://doi.org/10.18632/oncotarget.11135
Zhao Q, Fan S, Chang Y et al (2019) Clinical efficacy of cisplatin, dexamethasone, gemcitabine and pegaspargase (DDGP) in the initial treatment of advanced stage (stage III-IV) extranodal NK/T-cell lymphoma, and its correlation with Epstein-Barr virus. Cancer Manag Res. 11:3555–3564. https://doi.org/10.2147/CMAR.S191929
Zhou Z, Li X, Chen C, Li X, Zhang L, Li L, Wang X, Ma W, Fu X, Wu J, Sun Z, Zhang X, Li Z, Yan J, Chang Y, Lu L, Qin B, Li X, Wen J, Zhang M (2014) Effectiveness of gemcitabine, pegaspargase, cisplatin, and dexamethasone (DDGP) combination chemotherapy in the treatment of relapsed/refractory extranodal NK/T cell lymphoma: a retrospective study of 17 patients. Ann Hematol 93(11):1889–1894. https://doi.org/10.1007/s00277-014-2136-7
Zhou JF, Li CR et al (2014) Diagnosis and differential diagnosis of aggressive NK cell leukemia[J]. Chin J Hematol 35(04):278–279. https://doi.org/10.3760/cma.j.issn.0253-2727.2014.04.003
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this study was supported by National Science and Technology Major Project of China (Grant No. 2020ZX09201-009), the National Natural Science Foundation of China (Grant No. 81970184).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sun, Z., Wan, W., Zhang, X. et al. The clinical characteristics and prognostic factors of 410 patients with natural killer/T-cell lymphoma. J Cancer Res Clin Oncol 148, 3449–3459 (2022). https://doi.org/10.1007/s00432-022-04203-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-022-04203-x