Abstract
Purpose
Malignant rhabdoid tumor (MRT) is a rare, highly aggressive sarcoma with an uncertain cell of origin. Despite the existing standard of intensive multimodal therapy, the prognosis of patients with MRT is very poor. Novel antitumor agents are needed for MRT patients. Forkhead box transcription factor 1 (FOXM1) is overexpressed and is correlated with the pathogenesis in several human malignancies. In this study, we identified the clinicopathological and prognostic values of the expression of FOXM1 and its roles in the progression of MRT.
Methods
We investigated the FOXM1 expression levels and their clinical significance in 23 MRT specimens using immunohistochemistry and performed clinicopathologic and prognostic analyses. We also demonstrated correlations between the downregulation of FOXM1 and oncological characteristics using small interfering RNA (siRNA) and FOXM1 inhibitor in MRT cell lines.
Results
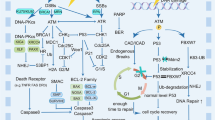
Histopathological analyses revealed that primary renal MRTs showed significantly low FOXM1 protein expression levels (p = 0.032); however, there were no significant differences in other clinicopathological characteristics or the survival rate. FOXM1 siRNA and FOXM1 inhibitor (thiostrepton) successfully downregulated the mRNA and protein expression of FOXM1 in vitro and the downregulation of FOXM1 inhibited cell proliferation, drug resistance to chemotherapeutic agents, migration, invasion, and caused the cell cycle arrest and apoptosis of MRT cell lines. A cDNA microarray analysis showed that FOXM1 regulated FANCD2 and NBS1, which are key genes for DNA damage repair.
Conclusion
This study demonstrates that FOXM1 may serve as a promising therapeutic target for MRT.








Similar content being viewed by others
Abbreviations
- MRT:
-
Malignant rhabdoid tumor
- MRTK:
-
MRT of the kidney
- EMRT:
-
Extra-renal non-cranial MRT
- AT/RT:
-
Atypical teratoid/rhabdoid tumor
- ATP:
-
Adenosine triphosphate
- CDK:
-
Cyclin-dependent kinase
- RB:
-
Retinoblastoma
- MYC:
-
C-avian myelocytomatosis viral oncogene homolog hemoglobin
- FOXM1:
-
Forkhead box protein M1
- DOX:
-
Doxorubicin
- FANCD2:
-
Fanconi anemia complementation group D2
- NBS1:
-
Nijmegen breakage syndrome 1
- ATM:
-
Ataxia-telangiectasia mutated
- IHC:
-
Immunohistochemical
- FBS:
-
Fetal bovine serum
- siRNA:
-
Small interfering RNA
- qRT-PCR:
-
Quantitative real-time polymerase chain reaction
- ANOVA:
-
Analysis of variance
- PI:
-
Propidium iodide
- ATR:
-
ATM and RAD3-related
- IR:
-
Ionizing radiation
- H2AX:
-
H2A histone family member X
- FAN1:
-
Fanconi-associated nuclease 1
- Pol Eta:
-
DNA polymerase eta
- BRCA1:
-
Breast cancer susceptibility gene 1
- MRE11:
-
Meiotic recombination 11
- RAD50:
-
DNA repair protein RAD50
- DSB:
-
DNA double-strand break
- HRR:
-
Homologous recombination repair
- NHEJ:
-
Non-homologous end joining
References
Abuzeid WM, Jiang X, Shi G et al (2009) Molecular disruption of RAD50 sensitizes human tumor cells to cisplatin-based chemotherapy. J Clin Invest 119:1974–1985. https://doi.org/10.1172/JCI33816
Beckwith JB, Palmer NF (1978) Histopathology and prognosis of Wilms tumor results from the first National Wilms’ tumor study. Cancer 41:1937–1948. https://doi.org/10.1002/1097-0142(197805)41:5%3c1937::AID-CNCR2820410538%3e3.0.CO;2-U
Biegel JA, Zhou JY, Rorke LB et al (1999) Germ-line and acquired mutations of INI1 in atypical teratoid and rhabdoid tumors. Cancer Res 59:74–79
Carr JR, Park HJ, Wang Z et al (2010) FoxM1 mediates resistance to herceptin and paclitaxel. Cancer Res 70:5054–5063. https://doi.org/10.1158/0008-5472.CAN-10-0545
Chi SN, Zimmerman MA, Yao X et al (2009) Intensive multimodality treatment for children with newly diagnosed CNS atypical teratoid rhabdoid tumor. J Clin Oncol 27:385–389. https://doi.org/10.1200/JCO.2008.18.7724
Chirnomas D, Taniguchi T, De La Vega M et al (2006) Chemosensitization to cisplatin by inhibitors of the Fanconi anemia/BRCA pathway. Mol Cancer Ther 5:952–961. https://doi.org/10.1158/1535-7163.MCT-05-0493
Chiu WT, Huang YF, Tsai HY et al (2015) FOXM1 confers to epithelial-mesenchymal transition, stemness and chemoresistance in epithelial ovarian carcinoma cells. Oncotarget 6:2349–2365. https://doi.org/10.18632/oncotarget.2957
Christensen L, Joo J, Lee S et al (2013) FOXM1 is an oncogenic mediator in Ewing sarcoma. PLoS ONE 8:1–7. https://doi.org/10.1371/journal.pone.0054556
de Moraes GN, Delbue D, Silva KL et al (2015) FOXM1 targets XIAP and Survivin to modulate breast cancer survival and chemoresistance. Cell Signal 27:2496–2505. https://doi.org/10.1016/j.cellsig.2015.09.013
Engeland K (2018) Cell cycle arrest through indirect transcriptional repression by p53: i have a DREAM. Cell Death Differ 25:114–132. https://doi.org/10.1038/cdd.2017.172
Gao R, Singh R, Kaul Z et al (2015) Targeting of DNA damage signaling pathway induced senescence and reduced migration of cancer cells. J Gerontol Ser A Biol Sci Med Sci 70:701–713. https://doi.org/10.1093/gerona/glu019
Gartel AL (2008) FoxM1 inhibitors as potential anticancer drugs. Expert Opin Ther Targets 12:663–665
Geller JI, Roth JJ, Biegel JA (2015) Biology and treatment of rhabdoid tumor. Crit Rev Oncog 20:199–216. https://doi.org/10.1615/CritRevOncog.2015013566
Ginn KF, Gajjar A (2012) Atypical teratoid rhabdoid tumor: current therapy and future directions. Front Oncol 2:1–13. https://doi.org/10.3389/fonc.2012.00114
Grant GD, Brooks L, Zhang X et al (2013) Identification of cell cycle-regulated genes periodically expressed in U2OS cells and their regulation by FOXM1 and E2F transcription factors. Mol Biol Cell 24:3634–3650. https://doi.org/10.1091/mbc.E13-05-0264
Grompe M (2001) Fanconi anemia and DNA repair. Hum Mol Genet 10:2253–2259. https://doi.org/10.1093/hmg/10.20.2253
Gu C, Jing X, Holman C et al (2019) Upregulation of FOXM1 leads to diminished drug sensitivity in myeloma. BMC Cancer 19:1238. https://doi.org/10.1186/s12885-019-6443-1
Halasi M, Gartel AL (2009) A novel mode of FoxM1 regulation: positive auto-regulatory loop. Cell Cycle 8:1966–1967. https://doi.org/10.4161/cc.8.12.8708
Hasselblatt M, Gesk S, Oyen F et al (2011) Nonsense mutation and inactivation of SMARCA4 (BRG1) in an atypical teratoid/rhabdoid tumor showing retained SMARCB1 (INI1) expression. Am J Surg Pathol 35:933–935. https://doi.org/10.1097/PAS.0b013e3182196a39
Helleday T, Lo J, van Gent DC, Engelward BP (2007) DNA double-strand break repair: from mechanistic understanding to cancer treatment. DNA Repair (Amst) 6:923–935. https://doi.org/10.1016/j.dnarep.2007.02.006
Ho V, Chung L, Singh A et al (2018) Overexpression of the MRE11-RAD50-NBS1 (MRN) complex in rectal cancer correlates with poor response to neoadjuvant radiotherapy and prognosis. BMC Cancer 18:1–11. https://doi.org/10.1186/s12885-018-4776-9
Hou Y, Zhu Q, Li Z et al (2017) The FOXM1-ABCC5 axis contributes to paclitaxel resistance in nasopharyngeal carcinoma cells. Cell Death Dis 8:e2659–e2711. https://doi.org/10.1038/cddis.2017.53
Ito T, Kohashi K, Yamada Y et al (2016a) Prognostic significance of Forkhead box M1 (FOXM1) expression and antitumor effect of FOXM1 inhibition in angiosarcoma. J Cancer 7:823–830. https://doi.org/10.7150/jca.14461
Ito T, Kohashi K, Yamada Y et al (2016b) Prognostic significance of forkhead box M1 (FoxM1) expression and antitumour effect of FoxM1 inhibition in melanoma. Histopathology 69:63–71. https://doi.org/10.1111/his.12909
Kalin TV, Wang I-C, Ackerson TJ, Major ML, Detrisac CJ, Kalinichenko VV, Alexander LRHC (2006) Increased levels of the FoxM1 transcription factor accelerate development and progression of prostate carcinomas in both TRAMP and LADY transgenic mice. Cancer Res 66:1712–1720
Kalinichenko VV, Major ML, Wang X et al (2004) Foxm1b transcription factor is essential for development of hepatocellular carcinomas and is negatively regulated by the p19ARF tumor suppressor. Genes Dev 18:830–850. https://doi.org/10.1101/gad.1200704
Khan I, Halasi M, Patel A et al (2018) FOXM1 contributes to treatment failure in acute myeloid leukemia. JCI insight 3:1–17. https://doi.org/10.1172/jci.insight.121583
Khongkow P, Karunarathna U, Khongkow M et al (2014) FOXM1 targets NBS1 to regulate DNA damage-induced senescence and epirubicin resistance. Oncogene 33:4144–4155. https://doi.org/10.1038/onc.2013.457
Kim KH, Roberts CWM (2014) Mechanisms by which SMARCB1 loss drives rhabdoid tumor growth. Cancer Genet 207:365–372. https://doi.org/10.1016/j.cancergen.2014.04.004
Kim IM, Ackerson T, Ramakrishna S et al (2006) The Forkhead Box m1 transcription factor stimulates the proliferation of tumor cells during development of lung cancer. Cancer Res 66:2153–2161. https://doi.org/10.1158/0008-5472.CAN-05-3003
Komatsu H, Masuda T, Iguchi T et al (2017) Clinical significance of fancd2 gene expression and its association with tumor progression in hepatocellular carcinoma. Anticancer Res 37:1083–1090. https://doi.org/10.21873/anticanres.11420
Kuda M, Kohashi K, Yamada Y et al (2016) FOXM1 expression in rhabdomyosarcoma: a novel prognostic factor and therapeutic target. Tumour Biol 37:5213–5223. https://doi.org/10.1007/s13277-015-4351-9
Kuo K-T, Chou T-Y, Hsu H-S et al (2012) Prognostic significance of NBS1 and snail expression in esophageal squamous cell carcinoma. Ann Surg Oncol 19:549–557. https://doi.org/10.1245/s10434-011-2043-2
Kwok JMM, Myatt SS, Marson CM et al (2008) Thiostrepton selectively targets breast cancer cells through inhibition of forkhead box M1 expression. Mol Cancer Ther 7:2022–2032. https://doi.org/10.1158/1535-7163.MCT-08-0188
Kwok JMM, Peck B, Monteiro LJ et al (2010) FOXM1 confers acquired cisplatin resistance in breast cancer cells. Mol Cancer Res 8:24–34. https://doi.org/10.1158/1541-7786.MCR-09-0432
Laoukili J, Kooistra MRH, Brás A et al (2005) FoxM1 is required for execution of the mitotic programme and chromosome stability. Nat Cell Biol 7:126–136. https://doi.org/10.1038/ncb1217
Leon-Galicia I, Diaz-Chavez J, Albino-Sanchez ME et al (2018) Resveratrol decreases Rad51 expression and sensitizes cisplatin-resistant MCF-7 breast cancer cells. Oncol Rep 39:3025–3033. https://doi.org/10.3892/or.2018.6336
Li D, Ye L, Lei Y et al (2019) Downregulation of FoxM1 sensitizes nasopharyngeal carcinoma cells to cisplatin via inhibition of MRN-ATM-mediated DNA repair. BMB Rep 52:208–213. https://doi.org/10.5483/BMBRep.2019.52.3.249
Liang CC, Li Z, Lopez-Martinez D et al (2016) The FANCD2-FANCI complex is recruited to DNA interstrand crosslinks before monoubiquitination of FANCD2. Nat Commun 7:1–10. https://doi.org/10.1038/ncomms12124
Liu M, Dai B, Kang SH et al (2006) FoxM1B is overexpressed in human glioblastomas and critically regulates the tumorigenicity of glioma cells. Cancer Res 66:3593–3602. https://doi.org/10.1158/0008-5472.CAN-05-2912
Liu Y, Chen X, Gu Y et al (2015) FOXM1 overexpression is associated with cisplatin resistance in non-small cell lung cancer and mediates sensitivity to cisplatin in A549 cells via the JNK/mitochondrial pathway. Neoplasma 62:61–71. https://doi.org/10.4149/neo_2015_008
Liu C, Shi J, Li Q et al (2019) STAT1-mediated inhibition of FOXM1 enhances gemcitabine sensitivity in pancreatic cancer. Clin Sci 133:645–663. https://doi.org/10.1042/CS20180816
Ma RYM, Tong THK, Leung WY, Yao KM (2010) Raf/MEK/MAPK signaling stimulates the nuclear translocation and transactivating activity of FOXM1. Methods Mol Biol 647:113–123. https://doi.org/10.1007/978-1-60761-738-9_6
Madureira PA, Varshochi R, Constantinidou D et al (2006) The forkhead box M1 protein regulates the transcription of the estrogen receptor α in breast cancer cells. J Biol Chem 281:25167–25176. https://doi.org/10.1074/jbc.M603906200
Maekawa A, Kohashi K, Kuda M et al (2016) Prognostic significance of FOXM1 expression and antitumor effect of FOXM1 inhibition in synovial sarcomas. BMC Cancer 16:1–12. https://doi.org/10.1186/s12885-016-2542-4
Maekawa A, Kohashi K, Setsu N et al (2016) Expression of Forkhead box M1 in soft tissue leiomyosarcoma: clinicopathologic and in vitro study using a newly established cell line. Cancer Sci 107:95–102. https://doi.org/10.1111/cas.12846
Major ML, Lepe R, Costa RH (2004) Forkhead Box M1B transcriptional activity requires binding of Cdk-Cyclin complexes for phosphorylation-dependent recruitment of p300/CBP coactivators. Mol Cell Biol 24:2649–2661. https://doi.org/10.1128/mcb.24.7.2649-2661.2004
Masliah-Planchon J, Bièche I, Guinebretière J-M et al (2015) SWI/SNF chromatin remodeling and human malignancies. Annu Rev Pathol Mech Dis 10:145–171
Metselaar DS, Meel MH, Benedict B et al (2019) Celastrol-induced degradation of FANCD2 sensitizes pediatric high-grade gliomas to the DNA-crosslinking agent carboplatin. EBioMedicine 50:81–92. https://doi.org/10.1016/j.ebiom.2019.10.062
Monteiro LJ, Khongkow P, Kongsema M et al (2013) The Forkhead Box M1 protein regulates BRIP1 expression and DNA damage repair in epirubicin treatment. Oncogene 32:4634–4645. https://doi.org/10.1038/onc.2012.491
Nakanishi K, Taniguchi T, Ranganathan V et al (2002) Interaction of FANCD2 and NBS1 in the DNA damage response. Nat Cell Biol 4:913–920. https://doi.org/10.1038/ncb879
Nakashima S, Kobayashi S, Nagano H et al (2015) BRCA/Fanconi anemia pathway implicates chemoresistance to gemcitabine in biliary tract cancer. Cancer Sci 106:584–591. https://doi.org/10.1111/cas.12652
Narita T, Taga T, Sugita K et al (2001) The autocrine loop of epidermal growth factor receptor-epidermal growth factor/transforming growth factor-α in malignant rhabdoid tumor cell lines: heterogeneity of autocrine mechanism in TTC549. Japanese J Cancer Res 92:269–278. https://doi.org/10.1111/j.1349-7006.2001.tb01091.x
Nepal M, Che R, Ma C et al (2017) FANCD2 and DNA damage. Int J Mol Sci 18:1–9. https://doi.org/10.3390/ijms18081804
Nicolaou KC, Zak M, Rahimipour S et al (2005) Discovery of a biologically active thiostrepton fragment. J Am Chem Soc 127:15042–15044. https://doi.org/10.1021/ja0552803
Okada K, Fujiwara Y, Takahashi T et al (2013) Overexpression of forkhead box M1 transcription factor (FOXM1) is a potential prognostic marker and enhances chemoresistance for docetaxel in gastric cancer. Ann Surg Oncol 20:1035–1043. https://doi.org/10.1245/s10434-012-2680-0
Palle K, Vaziri C (2011) Rad18 E3 ubiquitin ligase activity mediates Fanconi anemia pathway activation and cell survival following DNA topoisomerase 1 inhibition. Cell Cycle 10:1625–1638. https://doi.org/10.4161/cc.10.10.15617
Park HJ, Gusarova G, Wang Z et al (2011) Deregulation of FoxM1b leads to tumour metastasis. EMBO Mol Med 3:21–34. https://doi.org/10.1002/emmm.201000107
Park YY, Jung SY, Jennings NB et al (2012) FOXM1 mediates Dox resistance in breast cancer by enhancing DNA repair. Carcinogenesis 33:1843–1853. https://doi.org/10.1093/carcin/bgs167
Peng WX, Han X, Zhang CL et al (2017) FoxM1-mediated RFC5 expression promotes temozolomide resistance. Cell Biol Toxicol 33:527–537. https://doi.org/10.1007/s10565-017-9381-1
Pilarsky C, Wenzig M, Specht T et al (2004) Identification and validation of commonly overexpressed genes in solid tumors by comparison of microarray data. Neoplasia 6:744–750. https://doi.org/10.1593/neo.04277
Raychaudhuri P, Park HJ (2011) FoxM1: a master regulator of tumor metastasis. Cancer Res 71:4329–4333. https://doi.org/10.1158/0008-5472.CAN-11-0640
Ren L, Chen L, Wu W et al (2017) Potential biomarkers of DNA replication stress in cancer. Oncotarget 8:36996–37008. https://doi.org/10.18632/oncotarget.16940
Roberts CWM, Orkin SH (2004) The SWI/SNF complex—chromatin and cancer. Nat Rev Cancer 4:133–142. https://doi.org/10.1038/nrc1273
Roh YG, Mun MH, Jeong MS et al (2018) Drug resistance of bladder cancer cells through activation of ABCG2 by FOXM1. BMB Rep 51:98–103. https://doi.org/10.5483/BMBRep.2018.51.2.222
Roh YG, Mun JY, Kim SK et al (2020) Fanconi anemia pathway activation by foxm1 is critical to bladder cancer recurrence and anticancer drug resistance. Cancers (Basel) 12:1–20. https://doi.org/10.3390/cancers12061417
Schneppenheim R, Frühwald MC, Gesk S et al (2010) Germline nonsense mutation and somatic inactivation of SMARCA4/BRG1 in a family with rhabdoid tumor predisposition syndrome. Am J Hum Genet 86:279–284. https://doi.org/10.1016/j.ajhg.2010.01.013
Smirnov A, Panatta E, Lena AM et al (2016) FOXM1 regulates proliferation, senescence and oxidative stress in keratinocytes and cancer cells. Aging (Albany NY) 8:1384–1397. https://doi.org/10.18632/aging.100988
Su WP, Ho YC, Wu CK et al (2017) Chronic treatment with cisplatin induces chemoresistance through the TIP60-mediated Fanconi anemia and homologous recombination repair pathways. Sci Rep 7:1–15. https://doi.org/10.1038/s41598-017-04223-5
Sultan I, Qaddoumi I, Rodriguez-Galindo C et al (2010) Age, stage, and radiotherapy, but not primary tumor site, affects the outcome of patients with malignant rhabdoid tumors. Pediatr Blood Cancer 54:35–40. https://doi.org/10.1002/pbc.22285
Tan Y, Raychaudhuri P, Costa RH (2007) Chk2 mediates stabilization of the FoxM1 transcription factor to stimulate expression of DNA repair genes. Mol Cell Biol 27:1007–1016. https://doi.org/10.1128/mcb.01068-06
Taniguchi T, Garcia-Higuera I, Xu B et al (2002) Convergence of the Fanconi anemia and ataxia telangiectasia signaling pathways. Cell 109:459–472. https://doi.org/10.1016/S0092-8674(02)00747-X
Tassi RA, Todeschini P, Siegel ER et al (2017) FOXM1 expression is significantly associated with chemotherapy resistance and adverse prognosis in non-serous epithelial ovarian cancer patients. J Exp Clin Cancer Res 36:1–18. https://doi.org/10.1186/s13046-017-0536-y
Teh MT, Wong ST, Neill GW et al (2002) FOXM1 is a downstream target of Gli1 in basal cell carcinomas. Cancer Res 62:4773–4780
Tian L, Zhao Z, Xie L, Zhu JP (2018) MiR-361–5p suppresses chemoresistance of gastric cancer cells by targeting FOXM1 via the PI3K/Akt/mTOR pathway. Oncotarget 9:4886–4896. https://doi.org/10.18632/oncotarget.23513
Tomlinson GE, Breslow NE, Dome J et al (2005) Rhabdoid tumor of the kidney in the National Wilms’ Tumor Study: age at diagnosis as a prognostic factor. J Clin Oncol 23:7641–7645. https://doi.org/10.1200/JCO.2004.00.8110
Uddin S, Ahmed M, Hussain A et al (2011) Genome-wide expression analysis of Middle Eastern colorectal cancer reveals FOXM1 as a novel target for cancer therapy. Am J Pathol 178:537–547. https://doi.org/10.1016/j.ajpath.2010.10.020
van den Heuvel-Eibrink MM, van Tinteren H, Rehorst H et al (2011) Malignant rhabdoid tumours of the kidney (MRTKs), registered on recent SIOP protocols from 1993 to 2005: a report of the SIOP renal tumour study group. Pediatr Blood Cancer 56:733–737. https://doi.org/10.1002/pbc.22922
Versteege I, Sevenet N, Lange J, Rousseau-Merck M-F, Ambros P, Handgretinger Rt, Airias A, Delattre O (1998) Truncating mutations of hSNF5/INI1 in aggressive paedeatric cancer. Nature 394:203–206
Wan LY, Deng J, Xiang XJ et al (2015) miR-320 enhances the sensitivity of human colon cancer cells to chemoradiotherapy in vitro by targeting FOXM1. Biochem Biophys Res Commun 457:125–132. https://doi.org/10.1016/j.bbrc.2014.11.039
Wang X, Haswell JR, CMWR (2014) Molecular pathways: SWI/SNF(BAF) complexes are frequently mutated in cancer—mechanisms and potential therapeutic insights. Clin Cancer Res 20:21–27. https://doi.org/10.1158/1078-0432
Wang I-C, Chen Y-J, Hughes D et al (2005) Forkhead Box M1 regulates the transcriptional network of genes essential for mitotic progression and genes encoding the SCF (Skp2-Cks1) Ubiquitin Ligase. Mol Cell Biol 25:10875–10894. https://doi.org/10.1128/mcb.25.24.10875-10894.2005
Wang Y, Wiltshire T, Senft J et al (2006) Fanconi anemia D2 protein confers chemoresistance in response to the anticancer agent, irofulven. Mol Cancer Ther 5:3153–3161. https://doi.org/10.1158/1535-7163.MCT-06-0427
Wang Z, Banerjee S, Kong D et al (2007) Down-regulation of Forkhead Box M1 transcription factor leads to the inhibition of invasion and angiogenesis of pancreatic cancer cells. Cancer Res 67:8293–8300. https://doi.org/10.1158/0008-5472.CAN-07-1265
Wang I-C, Chen Y-J, Hughes DE et al (2008) FoxM1 regulates transcription of JNK1 to promote the G 1 /S transition and tumor cell invasiveness. J Biol Chem 283:20770–20778. https://doi.org/10.1074/jbc.M709892200
Weeks DA, Beckwith JB, Mierau GW, Luckey DW (1989) Rhabdoid tumor of kidney. A report of 111 cases from the National Wilms’ Tumor Study Pathology Center. Am J Surg Pathol 13:439–458
Westhoff GL, Chen Y, Teng NNH (2017) Targeting foxm1 improves cytotoxicity of paclitaxel and cisplatinum in platinum-resistant ovarian cancer. Int J Gynecol Cancer 27:887–894. https://doi.org/10.1097/IGC.0000000000000969
Wierstra I, Alves J (2007) FOXM1, a typical proliferation-associated transcription factor. Biol Chem 388:1257–1274. https://doi.org/10.1515/BC.2007.159
Wonsey DR, Follettie MT (2005) Loss of the forkhead transcription factor FoxM1 causes centrosome amplification and mitotic catastrophe. Cancer Res 65:5181–5189. https://doi.org/10.1158/0008-5472.CAN-04-4059
Wu QF, Liu C, Tai MH et al (2010) Knockdown of FoxM1 by siRNA interference decreases cell proliferation, induces cell cycle arrest and inhibits cell invasion in MHCC-97H cells in vitro. Acta Pharmacol Sin 31:361–366. https://doi.org/10.1038/aps.2010.4
Xia L, Mo P, Huang W et al (2012) The TNF-α/ROS/HIF-1-induced upregulation of foxMI expression promotes HCC proliferation and resistance to apoptosis. Carcinogenesis 33:2250–2259. https://doi.org/10.1093/carcin/bgs249
Xie T, Geng J, Wang Y, et al (2017) FOXM1 evokes 5-fluorouracil resistance in colorectal cancer depending on ABCC10 8:8574–8589
Xu K, Liu X, Mao X et al (2015) MicroRNA-149 suppresses colorectal cancer cell migration and invasion by directly targeting forkhead box transcription factor FOXM1. Cell Physiol Biochem 35:499–515. https://doi.org/10.1159/000369715
Ye Q, Chen L, Yin X et al (2014) Development of serous ovarian cancer is associated with the expression of homologous recombination pathway proteins. Pathol Oncol Res 20:931–938. https://doi.org/10.1007/s12253-014-9776-8
Yu J, Deshmukh H, Payton JE et al (2011) Array-based comparative genomic hybridization identifies CDK4 and FOXM1 alterations as independent predictors of survival in malignant peripheral nerve sheath tumor. Clin Cancer Res 17:1924–1934. https://doi.org/10.1158/1078-0432.CCR-10-1551
Zhang N, Wu X, Yang L et al (2012) FoxM1 inhibition sensitizes resistant glioblastoma cells to temozolomide by downregulating the expression of DNA-repair gene Rad51. Clin Cancer Res 18:5961–5971. https://doi.org/10.1158/1078-0432.CCR-12-0039
Zhang JR, Lu F, Lu T et al (2014) Inactivation of FoxM1 transcription factor contributes to curcumin-induced inhibition of survival, angiogenesis, and chemosensitivity in acute myeloid leukemia cells. J Mol Med 92:1319–1330. https://doi.org/10.1007/s00109-014-1198-2
Zhang C, Wang Y, Feng YF et al (2016) Gli1 promotes colorectal cancer metastasis in a Foxm1-dependent manner by activating EMT and PI3k-AKT signaling. Oncotarget 7:86134–86147
Zhu X, Xue L, Yao Y et al (2018) The FoxM1-ABCC4 axis mediates carboplatin resistance in human retinoblastoma Y-79 cells. Acta Biochim Biophys Sin (Shanghai) 50:914–920. https://doi.org/10.1093/abbs/gmy080
Acknowledgements
The authors thank to Maiko Takahashi (Kamakura Techno-Science, Inc.) for her invaluable assistance with the 3D-Gene analysis and the technical assistance from The Research Support Center, Research Center for Human Disease Modeling, Kyushu University Graduate School of Medical Sciences.
Funding
This study was supported by the Japan Society for the Promotion of Science KAKENHI grants, (JP19H03444).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest in association with the present study.
Ethics approval
This study was approved by the institutional review board at Kyushu University (permission code 29-429,29-625).
Informed consent
Informed consent was obtained from all participants included in this study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Shibui, Y., Kohashi, K., Tamaki, A. et al. The forkhead box M1 (FOXM1) expression and antitumor effect of FOXM1 inhibition in malignant rhabdoid tumor. J Cancer Res Clin Oncol 147, 1499–1518 (2021). https://doi.org/10.1007/s00432-020-03438-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-020-03438-w