Abstract
Background
Colorectal cancer (CRC) is now a major human cancer, and B-cell translocation gene 3 (BTG3) has been reported as a tumor-suppressor in CRC, but its upstream regulator has not been identified.
Methods
Endogenous expression levels of BTG3 were compared between normal colorectal cell line CCD-18Co and two CRC cell lines SW480 and HT29, as well as between CRC patient tumor and adjacent normal tissues. Analysis of BTG3 genomic region was performed which identified a putative hypoxia response element (HRE). Effects of hypoxia condition, BTG3 overexpression, and their combination on the radiation sensitivity of CRC cell lines were assessed.
Results
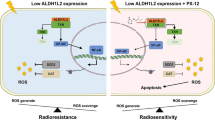
BTG3 was downregulated in CRC cell lines and patient tumor samples, via the HRE in its promoter region. Hypoxia and BTG3 overexpression could both induce radiation resistance in CRC cells. Combining hypoxia with BTG3 overexpression effectively rendered the resistance of CRC cells to radiation to a level lower than hypoxia alone and higher than normoxia alone, indicating the essential role of BTG3 in hypoxia-induced radiation resistance of CRC cells.
Conclusion
We therefore propose a novel signaling cascade involving hypoxia/BTG3 to be a potential risk factor for CRC patients undergoing radiation therapy, which could possibly serve as therapeutic targets among CRC patients with acquired radiotherapy resistance.







Similar content being viewed by others
References
Appelhoff RJ et al (2004) Differential function of the prolyl hydroxylases PHD1, PHD2, and PHD3 in the regulation of hypoxia-inducible factor. J Biol Chem 279:38458–38465. https://doi.org/10.1074/jbc.M406026200
Bertout JA et al (2009) HIF2alpha inhibition promotes p53 pathway activity, tumor cell death, and radiation responses. Proc Natl Acad Sci USA 106:14391–14396. https://doi.org/10.1073/pnas.0907357106
Cheng YC, Lin TY, Shieh SY (2013) Candidate tumor suppressor BTG3 maintains genomic stability by promoting Lys63-linked ubiquitination and activation of the checkpoint kinase CHK1. Proc Natl Acad Sci USA 110:5993–5998. https://doi.org/10.1073/pnas.1220635110
Cui H, Zhang S, Zhou H, Guo L (2017) Direct downregulation of B-cell translocation gene 3 by microRNA-93 is required for desensitizing esophageal cancer to radiotherapy. Dig Dis Sci 62:1995–2003. https://doi.org/10.1007/s10620-017-4579-x
Gou WF et al (2015) The roles of BTG3 expression in gastric cancer: a potential marker for carcinogenesis and a target molecule for gene therapy. Oncotarget 6:19841–19867. https://doi.org/10.18632/oncotarget.3734
Kuipers EJ et al (2015) Colorectal cancer. Nat Rev Dis Primers 1:15065. https://doi.org/10.1038/nrdp.2015.65
Lin TY, Cheng YC, Yang HC, Lin WC, Wang CC, Lai PL, Shieh SY (2012) Loss of the candidate tumor suppressor BTG3 triggers acute cellular senescence via the ERK-JMJD3-p16(INK4a) signaling axis. Oncogene 31:3287–3297. https://doi.org/10.1038/onc.2011.491
Liu ZG et al (2016) c-Fos over-expression promotes radioresistance and predicts poor prognosis in malignant glioma. Oncotarget 7:65946–65956. https://doi.org/10.18632/oncotarget.11779
Lv Z et al (2013) The suppressive role and aberrent promoter methylation of BTG3 in the progression of hepatocellular carcinoma. PLoS ONE 8:e77473. https://doi.org/10.1371/journal.pone.0077473
Lv C et al (2018) The function of BTG3 in colorectal cancer cells and its possible signaling pathway. J Cancer Res Clin Oncol 144:295–308. https://doi.org/10.1007/s00432-017-2561-9
Majid S et al (2010) Genistein reverses hypermethylation and induces active histone modifications in tumor suppressor gene B-Cell translocation gene 3 in prostate cancer. Cancer 116:66–76. https://doi.org/10.1002/cncr.24662
Mao D, Qiao L, Lu H, Feng Y (2016) B-cell translocation gene 3 overexpression inhibits proliferation and invasion of colorectal cancer SW480 cells via Wnt/beta-catenin signaling pathway. Neoplasma 63:705–716. https://doi.org/10.4149/neo_2016_507
Nagaraju GP, Bramhachari PV, Raghu G, El-Rayes BF (2015) Hypoxia inducible factor-1alpha: Its role in colorectal carcinogenesis and metastasis. Cancer Lett 366:11–18. https://doi.org/10.1016/j.canlet.2015.06.005
Ou YH, Chung PH, Hsu FF, Sun TP, Chang WY, Shieh SY (2007) The candidate tumor suppressor BTG3 is a transcriptional target of p53 that inhibits E2F1. EMBO J 26:3968–3980. https://doi.org/10.1038/sj.emboj.7601825
Papamichael D et al (2015) Treatment of colorectal cancer in older patients: International Society of Geriatric Oncology (SIOG) consensus recommendations 2013. Ann Oncol 26:463–476. https://doi.org/10.1093/annonc/mdu253
Perez-Carbonell L et al (2012) Comparison between universal molecular screening for Lynch syndrome and revised Bethesda guidelines in a large population-based cohort of patients with colorectal cancer. Gut 61:865–872. https://doi.org/10.1136/gutjnl-2011-300041
Rankin EB, Giaccia AJ (2008) The role of hypoxia-inducible factors in tumorigenesis. Cell Death Differ 15:678–685. https://doi.org/10.1038/cdd.2008.21
Ren XL et al (2015) Down-regulation of BTG3 promotes cell proliferation, migration and invasion and predicts survival in gastric cancer. J Cancer Res Clin Oncol 141:397–405. https://doi.org/10.1007/s00432-014-1826-9
Ren T et al (2019) The long non-coding RNA HOTAIRM1 suppresses cell progression via sponging endogenous miR-17–5p/ B-cell translocation gene 3 (BTG3) axis in 5-fluorouracil resistant colorectal cancer cells. Biomed Pharmacother 117:109171. https://doi.org/10.1016/j.biopha.2019.109171
Santoyo-Ramos P, Likhatcheva M, Garcia-Zepeda EA, Castaneda-Patlan MC, Robles-Flores M (2014) Hypoxia-inducible factors modulate the stemness and malignancy of colon cancer cells by playing opposite roles in canonical Wnt signaling. PLoS ONE 9:e112580. https://doi.org/10.1371/journal.pone.0112580
Semenza GL (2012) Hypoxia-inducible factors in physiology and medicine. Cell 148:399–408. https://doi.org/10.1016/j.cell.2012.01.021
Simon MC, Keith B (2008) The role of oxygen availability in embryonic development and stem cell function. Nat Rev Mol Cell Biol 9:285–296. https://doi.org/10.1038/nrm2354
Vasen HF, Tomlinson I, Castells A (2015) Clinical management of hereditary colorectal cancer syndromes. Nat Rev Gastroenterol Hepatol 12:88–97. https://doi.org/10.1038/nrgastro.2014.229
Wenger RH, Gassmann M (1997) Oxygen(es) and the hypoxia-inducible factor-1. Biol Chem 378:609–616
Wood SM, Gleadle JM, Pugh CW, Hankinson O, Ratcliffe PJ (1996) The role of the aryl hydrocarbon receptor nuclear translocator (ARNT) in hypoxic induction of gene expression. Studies in ARNT-deficient cells. J Biol Chem 271:15117–15123. https://doi.org/10.1074/jbc.271.25.15117
Wu L et al (2015) HIF-2alpha mediates hypoxia-induced LIF expression in human colorectal cancer cells. Oncotarget 6:4406–4417. https://doi.org/10.18632/oncotarget.3017
Funding
The study was supported by the Natural Science Foundation of Zhejiang Province (LY20H160004).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No conflicts of interest, financial or otherwise, are declared by the authors.
Research involving human participants and/or animals
The experimental protocols that involve patients in the current study obtained approval from the Ethical Committee of Eastern Hepatobiliary Surgery Hospital, Second Military Medical University.
Informed consent
All participants in this study were informed and gave a written consent.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ma, D., Gao, X., Tao, J. et al. Hypoxia-induced downregulation of B-cell translocation gene 3 confers resistance to radiation therapy of colorectal cancer. J Cancer Res Clin Oncol 146, 2509–2517 (2020). https://doi.org/10.1007/s00432-020-03307-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-020-03307-6