Abstract
Purpose
Tumor explant culture systems can mimic the in vivo tumor microenvironment, proposing as a substitute for preclinical studies for prediction of individual treatment response. Therefore, our study evaluated the potential usefulness of ex vivo tumor explants culture assembled into the cell sheets by anticancer drug screening in patients with head and neck squamous cell carcinoma (HNSCC).
Methods
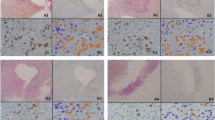
Our model included tumor explants incorporated into cell sheet composing of epithelium and subepithelial stroma using tumor and mucosal samples obtained from the HNSCC patients who underwent surgery. Cell growth, viability, and hypoxia were measured by cell counting kit-8, live/dead assay, propidium iodide, and LOX-1 staining, and were compared among the different treatment groups with vehicle, cisplatin or docetaxel.
Results
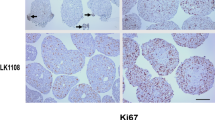
Tumor explants stably survived in the cell sheet over 10 days after explantation, whereas most of the explants in non-matrix culture became nonviable within 5–8 days with the significant daily decrease of viability. The live tissue areas of tumor explants in the cell sheet maintained over 30 days without significant changes although hypoxic cell areas gradually increased up to 5 days. Tissue viability and live cancer tissue areas significantly decreased after the treatment of cisplatin or docetaxel in the dose and time-dependent manners.
Conclusion
Our cell sheet-based tumor explants model might be applied to the reliable ex vivo screening for anticancer chemotherapeutics for HNSCC.





Similar content being viewed by others

References
Argiris A, Karamouzis MV, Raben D, Ferris RL (2008) Head and neck cancer. Lancet 371(9625):1695–1709
Argiris A, Harrington KJ, Tahara M, Schulten J, Chomette P, Ferreira Castro A, Licitra L (2017) Evidence-based treatment options in recurrent and/or metastatic squamous cell carcinoma of the head and neck. Front Oncol 7:72
Bazou D, Maimon N, Gruionu G, Grahovac J, Seano G, Liu H, Evans CL, Munn LL (2018) Vascular beds maintain pancreatic tumour explants for ex vivo drug screening. J Tissue Eng Regen Med 12(1):e318–e322
Choong N, Vokes E (2008) Expanding role of the medical oncologist in the management of head and neck cancer. CA Cancer J Clin 58(1):32–53
Costa EC, Moreira AF, de Melo-Diogo D, Gaspar VM, Carvalho MP, Correia IJ (2016) 3D tumor spheroids: an overview on the tools and techniques used for their analysis. Biotechnol Adv 34(8):1427–1441
Cukierman E, Pankov R, Stevens DR, Yamada KM (2001) Taking cell-matrix adhesions to the third dimension. Science 294(5547):1708–1712
Denaro N, Merlano MC (2018) Immunotherapy in head and neck squamous cell cancer. Clin Exp Otorhinolaryngol 11(4):217–223
Figiel S, Pasqualin C, Bery F, Maupoil V, Vandier C, Potier-Cartereau M, Domingo I, Guibon R, Bruyere F, Maheo K, Fromont G (2019) Functional organotypic cultures of prostate tissues: a relevant preclinical model that preserves hypoxia sensitivity and calcium signaling. Am J Pathol 189(6):1268–1275
Gerlach MM, Merz F, Wichmann G, Kubick C, Wittekind C, Lordick F, Dietz A, Bechmann I (2014) Slice cultures from head and neck squamous cell carcinoma: a novel test system for drug susceptibility and mechanisms of resistance. Br J Cancer 110(2):479–488
Haddad RI, Shin DM (2008) Recent advances in head and neck cancer. N Engl J Med 359(11):1143–1154
Hammond EM, Asselin MC, Forster D, O’Connor JP, Senra JM, Williams KJ (2014) The meaning, measurement and modification of hypoxia in the laboratory and the clinic. Clin Oncol (R Coll Radiol) 26(5):277–288
Joo YH, Cho JK, Koo BS, Kwon M, Kwon SK, Kwon SY, Kim MS, Kim JK, Kim H, Nam I, Roh JL, Park YM, Park IS, Park JJ, Shin SC, Ahn SH, Won S, Ryu CH, Yoon TM, Lee G, Lee DY, Lee MC, Lee JK, Lee JC, Lim JY, Chang JW, Jang JY, Chung MK, Jung YS, Cho JG, Choi YS, Choi JS, Lee GH, Chung PS (2019) Guidelines for the surgical management of oral cancer: Korean Society of Thyroid-Head and Neck Surgery. Clin Exp Otorhinolaryngol 12(2):107–144
Karekla E, Liao WJ, Sharp B, Pugh J, Reid H, Quesne JL, Moore D, Pritchard C, MacFarlane M, Pringle JH (2017) Ex vivo explant cultures of non-small cell lung carcinoma enable evaluation of primary tumor responses to anticancer therapy. Cancer Res 77(8):2029–2039
Kim BH, Park SJ, Jeong WJ, Ahn SH (2018) Comparison of treatment outcomes for T3 glottic squamous cell carcinoma: a meta-analysis. Clin Exp Otorhinolaryngol 11(1):1–8
Lee J, Shin D, Roh JL (2018a) Development of an in vitro cell-sheet cancer model for chemotherapeutic screening. Theranostics 8(14):3964–3973
Lee J, Shin D, Roh JL (2018b) Use of a pre-vascularised oral mucosal cell sheet for promoting cutaneous burn wound healing. Theranostics 8(20):5703–5712
Ma J, Zhang X, Liu Y, Yu H, Liu L, Shi Y, Li Y, Qin J (2016) Patterning hypoxic multicellular spheroids in a 3D matrix—a promising method for anti-tumor drug screening. Biotechnol J 11(1):127–134
Martinez-Santamaria L, Conti CJ, Llames S, Garcia E, Retamosa L, Holguin A, Illera N, Duarte B, Camblor L, Llaneza JM, Jorcano JL, Larcher F, Meana A, Escamez MJ, Del Rio M (2013) The regenerative potential of fibroblasts in a new diabetes-induced delayed humanised wound healing model. Exp Dermatol 22(3):195–201
McLean IC, Schwerdtfeger LA, Tobet SA, Henry CS (2018) Powering ex vivo tissue models in microfluidic systems. Lab Chip 18(10):1399–1410
Meijer TG, Naipal KA, Jager A, van Gent DC (2017) Ex vivo tumor culture systems for functional drug testing and therapy response prediction. Future Sci OA 3(2):190
Murata T, Mizushima H, Chinen I, Moribe H, Yagi S, Hoffman RM, Kimura T, Yoshino K, Ueda Y, Enomoto T, Mekada E (2011) HB-EGF and PDGF mediate reciprocal interactions of carcinoma cells with cancer-associated fibroblasts to support progression of uterine cervical cancers. Cancer Res 71(21):6633–6642
Naipal KA, Verkaik NS, Ameziane N, van Deurzen CH, Ter Brugge P, Meijers M, Sieuwerts AM, Martens JW, O’Connor MJ, Vrieling H, Hoeijmakers JH, Jonkers J, Kanaar R, de Winter JP, Vreeswijk MP, Jager A, van Gent DC (2014) Functional ex vivo assay to select homologous recombination-deficient breast tumors for PARP inhibitor treatment. Clin Cancer Res 20(18):4816–4826
Naipal KA, Verkaik NS, Sanchez H, van Deurzen CH, den Bakker MA, Hoeijmakers JH, Kanaar R, Vreeswijk MP, Jager A, van Gent DC (2016) Tumor slice culture system to assess drug response of primary breast cancer. BMC Cancer 16:78
Nelson CM, Bissell MJ (2005) Modeling dynamic reciprocity: engineering three-dimensional culture models of breast architecture, function, and neoplastic transformation. Semin Cancer Biol 15(5):342–352
Pal M, Chen H, Lee BH, Lee JYH, Yip YS, Tan NS, Tan LP (2019) Epithelial-mesenchymal transition of cancer cells using bioengineered hybrid scaffold composed of hydrogel/3D-fibrous framework. Sci Rep 9(1):8997
Psaila B, Lyden D (2009) The metastatic niche: adapting the foreign soil. Nat Rev Cancer 9(4):285–293
Roh JL, Jang H, Lee J, Kim EH, Shin D (2017) Promotion of oral surgical wound healing using autologous mucosal cell sheets. Oral Oncol 69:84–91
Roh JL, Lee J, Jang H, Kim EH, Shin D (2018) Use of oral mucosal cell sheets for accelerated oral surgical wound healing. Head Neck 40(2):394–401
Rohwer N, Cramer T (2011) Hypoxia-mediated drug resistance: novel insights on the functional interaction of HIFs and cell death pathways. Drug Resist Updat 14(3):191–201
Sato T, Stange DE, Ferrante M, Vries RG, Van Es JH, Van den Brink S, Van Houdt WJ, Pronk A, Van Gorp J, Siersema PD, Clevers H (2011) Long-term expansion of epithelial organoids from human colon, adenoma, adenocarcinoma, and Barrett’s epithelium. Gastroenterology 141(5):1762–1772
Sclafani AP, McCormick SA (2012) Induction of dermal collagenesis, angiogenesis, and adipogenesis in human skin by injection of platelet-rich fibrin matrix. Arch Facial Plast Surg 14(2):132–136
Serebriiskii I, Castello-Cros R, Lamb A, Golemis EA, Cukierman E (2008) Fibroblast-derived 3D matrix differentially regulates the growth and drug-responsiveness of human cancer cells. Matrix Biol 27(6):573–585
Siolas D, Hannon GJ (2013) Patient-derived tumor xenografts: transforming clinical samples into mouse models. Cancer Res 73(17):5315–5319
Sung KE, Beebe DJ (2014) Microfluidic 3D models of cancer. Adv Drug Deliv Rev 79–80:68–78
Tang XH, Scognamiglio T, Gudas LJ (2013) Basal stem cells contribute to squamous cell carcinomas in the oral cavity. Carcinogenesis 34(5):1158–1164
Unger FT, Witte I, David KA (2015) Prediction of individual response to anticancer therapy: historical and future perspectives. Cell Mol Life Sci 72(4):729–757
Vaira V, Fedele G, Pyne S, Fasoli E, Zadra G, Bailey D, Snyder E, Faversani A, Coggi G, Flavin R, Bosari S, Loda M (2010) Preclinical model of organotypic culture for pharmacodynamic profiling of human tumors. Proc Natl Acad Sci USA 107(18):8352–8356
van de Wetering M, Francies HE, Francis JM, Bounova G, Iorio F, Pronk A, van Houdt W, van Gorp J, Taylor-Weiner A, Kester L, McLaren-Douglas A, Blokker J, Jaksani S, Bartfeld S, Volckman R, van Sluis P, Li VS, Seepo S, Sekhar Pedamallu C, Cibulskis K, Carter SL, McKenna A, Lawrence MS, Lichtenstein L, Stewart C, Koster J, Versteeg R, van Oudenaarden A, Saez-Rodriguez J, Vries RG, Getz G, Wessels L, Stratton MR, McDermott U, Meyerson M, Garnett MJ, Clevers H (2015) Prospective derivation of a living organoid biobank of colorectal cancer patients. Cell 161(4):933–945
Zimmermann M, Lampe J, Lange S, Smirnow I, Konigsrainer A, Hann-von-Weyhern C, Fend F, Gregor M, Bitzer M, Lauer UM (2009) Improved reproducibility in preparing precision-cut liver tissue slices. Cytotechnology 61(3):145–152
Funding
This study was supported by the National Research Foundation of Korea (NRF) grant, funded by the Ministry of Science and ICT (MSIT), the Government of Korea (No. 2019R1A2C2002259).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest to disclose.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional research board and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. This article does not contain any studies with animals performed by any of the authors. Informed consent from all individual participants was obtained.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lee, J., You, J.H., Shin, D. et al. Ex vivo culture of head and neck cancer explants in cell sheet for testing chemotherapeutic sensitivity. J Cancer Res Clin Oncol 146, 2497–2507 (2020). https://doi.org/10.1007/s00432-020-03306-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-020-03306-7