Abstract
Purpose
Multiple lung lesions found in a single patient at the time of diagnosis often pose a diagnostic dilemma: are these lesions independent primary tumors (IPT) or the result of intrapulmonary metastases (IPM)? While traditional pathological methods sometimes have difficulty distinguishing IPM from IPT, modern molecular profiling based on next-generation sequencing techniques may provide a new strategy.
Methods
Sixteen patients with multiple tumors were enrolled in this study. We performed targeted deep sequencing (~ 2000 × coverage) on a total of 40 tumors and matched blood samples. We compared mutational profiles between tumors within each patient and across patients to evaluate if they were genetically related. Computed tomographic images and histological staining were also used to validate tumor relationships.
Results
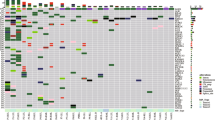
A total of 125 mutations were identified in 16 patients. Twelve out of fourteen patients whose histological diagnoses favored IPT did not have any shared mutations in their multiple tumors. The other two showed discrepancies: Pt01 had a shared EGFR exon19 deletion in the two lung tumors found, and Pt16 had one common mutation (BRAFD594G) in two out of five lung tumors. Pt14 with lung metastasis from salivary gland adenoid cystic carcinoma had shared mutations; and Pt15 with suspected intrapulmonary metastasis (IPM) had identical mutations between the two tumors. Visualized data can be readily accessed through the website: mlc.opengene.org.
Conclusion
Analysis of overlapping mutations among different tumors assists physicians in distinguishing IPM from IPT. Our findings demonstrate that DNA sequencing can provide additional evidence in clinical practice when pathology is inadequate to make a conclusive diagnosis.




Similar content being viewed by others
Availability of data and materials
To access the supplementary material accompanying this article, visit the online version of the journal at https://www.springer.com/journal/432/. Raw datasets analyzed during the current study are available from the corresponding author upon reasonable request.
Abbreviations
- IPT:
-
Independent primary tumor
- IPM:
-
Intrapulmonary metastasis
- NGS:
-
Next generation sequencing
- CT:
-
Computed tomography
- ctDNA:
-
Circulating tumor DNA
- VAF:
-
Variant allele frequency
- RUL:
-
Right upper lobe
- LUL:
-
Left upper lobe
- AIS:
-
Adenocarcinoma in situ
- MIA:
-
Minimally invasive adenocarcinoma
- ACC:
-
Adenoid cystic carcinoma
References
Aokage K, Ishii G, Nagai K et al (2007) Intrapulmonary metastasis in resected pathologic stage IIIB non-small cell lung cancer: possible contribution of aerogenous metastasis to the favorable outcome. J Thorac Cardiovasc Surg 134(2):386–391
Chen K, Chen W, Cai J et al (2018a) Favorable prognosis and high discrepancy of genetic features in surgical patients with multiple primary lung cancers. J Thorac Cardiovasc Surg 155(1):371–379
Chen S, Zhou Y, Chen Y et al (2019) Gencore: an efficient tool to generate consensus reads for error suppressing and duplicate removing of NGS data. BMC Bioinforma 20(Suppl 23):606
Chen S, Zhou Y, Chen Y, Gu J (2018c) Fastp: an ultra-fast all-in-one FASTQ preprocessor. Bioinformatics 34(17):i884–i890
Cheng DT, Mitchell TN, Zehir A et al (2015) Memorial sloan kettering-integrated mutation profiling of actionable cancer targets (MSK-IMPACT): a hybridization capture-based next-generation sequencing clinical assay for solid tumor molecular oncology. J Mol Diagn 17(3):251–264
Fabian T, Bryant AS, Mouhlas AL, Federico JA, Cerfolio RJ (2011) Survival after resection of synchronous non-small cell lung cancer. J Thorac Cardiovasc Surg 142(3):547–553
Gaikwad A, Souza CA, Inacio JR et al (2014) Aerogenous metastases: a potential game changer in the diagnosis and management of primary lung adenocarcinoma. Am J Roentgenol 203(6):W570–W582
Girard N, Pao W, Deshpande C et al (2009) Comprehensive histologic assessment helps to differentiate multiple lung primary nonsmall cell carcinomas from metastases. Am J Surg Pathol 33(12):1752–1764
Govindan R, Ding L, Griffith M et al (2012) Genomic landscape of non-small cell lung cancer in smokers and never-smokers. Cell 150:1121–1134
Koboldt DC, Zhang Q, Larson DE et al (2012) VarScan 2: somatic mutation and copy number alteration discovery in cancer by exome sequencing. Genome Res 22(3):568–576
Lawrence MS, Stojanov P, Polak P et al (2013) Mutational heterogeneity in cancer and the search for new cancer-associated genes. Nature 499:214
Li H, Durbin R (2010) Fast and accurate long-read alignment with Burrows–Wheeler transform. Bioinformatics 26(5):589–595
Li H, Handsaker B, Wysoker A et al (2009) The sequence alignment/map format and SAMtools. Bioinformatics 25(16):2078–2079
Li R, Li X, Xue R et al (2018) Early metastasis detected in patients with multifocal pulmonary ground-glass opacities (GGOs). Thorax 73(3):290–292
Liu Y, Zhang J, Li L et al (2016) Genomic heterogeneity of multiple synchronous lung cancer. Nat Commun. 7:1–8
Macmahon H, Naidich DP, Goo JM et al (2017) Guidelines for management of incidental pulmonary nodules MacMahon et al. Radiol Radiol 284:228–243
Martini N, Melamed MR (1975) Multiple primary lung cancers. J Thorac Cardiovasc Surg 70(4):606–612
Murphy SJ, Aubry MC, Harris FR et al (2014) Identification of independent primary tumors and intrapulmonary metastases using DNA rearrangements in non-small-cell lung cancer. J Clin Oncol 32(36):4050–4058
Pomplun S (2006) Pathology of lung cancer. Lung Cancer 32(4):12–26
Sweeney SM, Cerami E, Baras A et al (2017) AACR project genie: powering precision medicine through an international consortium. Cancer Discov 7(8):818–831
Team TNLSTR (2011) Reduced lung-cancer mortality with low-dose computed tomographic screening. N Engl J Med. 365(5):395–409
Wu C, Zhao C, Yang Y et al (2015) High discrepancy of driver mutations in patients with NSCLC and synchronous multiple lung ground-glass nodules. J Thorac Oncol. 10(5):778–783
Yatabe Y, Kerr KM, Utomo A et al (2015) EGFR mutation testing practices within the Asia pacific region: results of a multicenter diagnostic survey. J Thorac Oncol 10(3):438–445
Zehir A, Benayed R, Shah RH et al (2017) Mutational landscape of metastatic cancer revealed from prospective clinical sequencing of 10,000 patients. Nat Med 23(6):703–713
Acknowledgements
We acknowledge the generous gift of clinical specimens from our patients, to whom we dedicate our work. We thank Jinhua Zhong for clinical sample coordination.
Funding
We thank grants from the Natural Science Foundation of Guangdong Province of China (2017A030310641), the Medical Scientific Research Foundation of Guangdong Province of China (A2017327) for study design and data collection, the Science and Technology Innovation Committee of Shenzhen Municipality (JCYJ20180228175531145) for data collection and analysis, the Shenzhen Strategic Emerging Industry Development Special Fund (20170922151538732) and the PUHSC-UMHS Joint Institute Project (2019020(PUSH)-r1) for interpretation and writing.
Author information
Authors and Affiliations
Contributions
JL, YL, SC and DW designed the study; LT performed the histological examination; JL, GM, XP, JW, XL and RL analyzed and interpreted the patient clinical data; MX and TH carried out the sequencing experiment and collected data; YL, WW, JZ and SC analyzed the bioinformatic data; YL, JL, XL and SC were major contributors in writing manuscript; SC and DW supervised the study.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Ethics approval and consent to participate
Clinical samples were obtained through the Peking University Shenzhen Hospital Institutional Review Board-approved informed consent process. No procedures were conducted for the exclusive purpose of research.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Liu, J., Mao, G., Li, Y. et al. Targeted deep sequencing helps distinguish independent primary tumors from intrapulmonary metastasis for lung cancer diagnosis. J Cancer Res Clin Oncol 146, 2359–2367 (2020). https://doi.org/10.1007/s00432-020-03227-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-020-03227-5