Abstract
Objective
To determine if individual, instead of group, patient progression risk could be predicted using p53, Ki67 and CK20 biomarker percentage values at initial transurethral resection of bladder tumor specimens.
Methods
This was an observational study where biomarkers were measured with no knowledge of tumor outcome. Initial bladder tumor specimens were classified as non-invasive and invasive to sub-epithelium (pT1). Percentages of stained biomarker cells were tested as progression predictors from non-invasive to pT1 and pT1 to pT2. Progression probability was correlated with biomarker percentages resulting in a regression equation.
Results
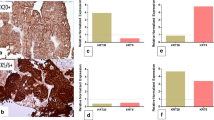
We studied 112 patients (median age = 67, range 37–91, males 83/112 (73%), with median follow-up of 39 months (range 1.7–140). Mean biomarker values were higher in stage pT1 than in non-invasive (all p < 0.001). Cut-off points separating progression from non-progression groups in stage pT1 were higher than in non-invasive for all biomarkers. Correlation R values for progression probability vs. biomarker percentages varied from 0.7 to 0.9 (all p < 0.001), regression slopes from 0.1 to 0.8 and intercepts from 11 to 35. A novel individual progression probability was calculated as the product of biomarker percentage of stained cells and slope, plus the prevalence-adjusted intercept.
Conclusions
Identification of individual risk of progression in patients with non-muscle-invasive bladder tumors was possible using p53- and Ki67-derived progression probability using a regression equation. Combining biomarker-derived progression probability to tumor stage pT1 improves progression to pT2 predictive accuracy.






Similar content being viewed by others
References
Agarwal P, Sen AK, Bhardwaj M, Dinand V, Ahuja A, Sood R (2018) Study of Proliferating cell nuclear antigen expression and Angiogenesis in Urothelial neoplasms: correlation with tumor grade and stage. Urol Ann 10:209–214
Ahmad I, Samsom OJ, Leung HY (2012) Exploring molecular genetics of bladeer cancer: lessons learned from mouse models. Dis Model Mech 5(3):323–332
Al-Hussain TO, Akhtar M (2013) Molecular basis of urinary bladder cancer. Adv Anat Pathol 20(1):53–60
Butterfield A, Gupta S (2017) Next-generation sequencing in non-muscle-invasive bladder cancer-a step towards personalized medicine for a superficial bladder tumor. Transl Androl Urol 6(6):1198–1202
D’Andrea D, Abufaraj M, Susani M et al (2018) Accurate prediction of progression to muscle-invasive disease in patients with pT1G3 bladder cancer: A clinical decision-making tool. Urol Oncol 36(5):239 (Abstract)
Fujii Y (2018) Prediction models for progression of non-muscle invasive bladder cancer: a review. Int J Urol 25:212–218
Goldstein I, Marcel V, Olivier M, Oren M, Rotter V, Hainaut P (2011) Understanding wild-type and mutant p53 activities in human cancer: new landmarks on the way to targeted therapies. Cancer Gene Ther 18(1):2–11
Grignon DJ (2014) Tumors of the urinary bladder. In: Amin MB, Eble J (eds) Urological pathology, 1st edn. Lippincott, Williams & Wilkins, Philadelphia, pp 394–397
Grignon DJ, Al-Ahmadie H, Agaba F (2016) Tumors of urinary tract. In: Moch H, Humphrey PA, Ulbright TM (eds) WHO classification of tumors of the urinary system and male genital organs, 4th edn. International Agency for Research on Cancer, Lyon, pp 78–107
Grossman HB, Liebert M, Antelo M et al (1998) p53 and RB expression predict progression in T1 bladder cancer. Clin Cancer Res 4(4):829–834
Hitchings AW, Kumar M, Jordan S, Nargund V, Martin J, Berney DM (2004) Prediction of progression in pTa and pT1 bladder carcinomas with p53, p16 and pRb. Br J Cancer 91:552–557
Humpfrey PA, Moch H, Cubilla AL, Ulbright TM, Reuter VE (2016) The 2016 WHO classification of tumors of the urinary system and male genital organs. Part B: prostate and bladder tumors. Eur Urol 70:106–119
Kluth LA, Black PC, Bochner BH et al (2015) Prognostic and Prediction Tools in Bladder Cancer: A Comprehensive Review of the Literature. Eur Urol 68:238–253
Kojima T, Kawai K, Miyazaki J, Nishiyama H (2017) Biomarkers for precision medicine in bladder cancer. Int J Clin Oncol 22(2):207–213
Kulkarni GS, Hakenberg OW, Gschwend JE et al (2010) An updated critical analysis of the treatment strategy for newly diagnosed high-grade T1 (previously T1G3) bladder cancer. Eur Urol 57:60–70
Kutwin P, Konecki T, Borkowska EM et al (2018) Urine miRNA as a potential biomarker for bladder cancer detection—a meta-analysis. Cent Eur J Urol 71(2):177–185
Lopez Beltran A, Montironi R (2004) Non invasive urothelial neoplasias WHO classification. Eur Urol 46:170–176
MacEneaney PM, Malone DE (2000) The meaning of diagnostic test results: a spreadsheet for swift data analysis. Radiology 55:227–235
Margulis V, Lotan Y, Karakiewicz PI (2009) Multi-institutional validation of the predictive value of Ki-67 labeling index in patients with urinary bladder cancer. J Natl Cancer Inst 101(2):114–119
May M, Helke C, Nitzke T, Vogler H, Hoschke B (2004) Survival rates after radical cystectomy according to tumor stage of bladder carcinoma at first presentation. Urol Int 72(2):103–111
Mumtaz S, Hashmi AA, Hasan SH, Edhi MM, Khan M (2014) Diagnostic utility of p53 and CK20 immunohistochemical expression grading urothelial malignancies. Int Arch Med 7:36
Paner GP, Montironi R, Amin MB (2017) Challenges in pathologic staging of bladder cancer: proposals for fresh approaches of assessing pathologic stage in ligh of recent studies and observations pertaining to bladder histoanatomic variances. Adv Anat Pathol 24(3):113–127
Patterson K, Arya L, Bottomley S et al (2016) Altered RECQL5 expression in urothelial bladder carcinoma increases cellular proliferation and makes RECQL5 helicase activity a novel target for chemotherapy. Oncotarget 7(46):76140–76150
Pietzak EJ, Bagrodia A, Cha EK et al (2017) Next-generation sequencing of nonmuscle invasive bladder cancer reveals potential biomarkers and rational therapeutic targets. Eur Urol 72(6):952–959
Poletajew S, Wolinska E, Wasiutynski A, Dybowski B, Radziszewski P, Górnicka B (2017) Immunohistochemical differentiation between muscularis propria for improving the staging of bladder cancer undergoing transurethral resection of bladder tumors. Pol J Pathol 68(3):218–220
Quintero A, Alvarez-Kindelan J, Luque RJ et al (2006) Ki67 MIB 1 labelling index and the prognosis of primary TaT1 urothelial carcinoma of the bladder. J Clin Pathol 59:83–88
Ravvaz K, Walz ME, Weissert JA, Downs TM (2017) Predicting nonmuscle invasive bladder cancer recurrence and progression in a United States population. J Urol 198(4):824–831
Sanli O, Lotan Y (2018) Current approaches for identifying high-risk non-muscle invasive bladder cancer. Expert Rev Anticancer Ther 18(3):223–235
Seo KW, Kim BH, Park CH, Kim CI, Chang HS (2010) The efficacy of the EORTC scoring system and risk tables for the prediction of recurrence and progression of non-muscle-invasive bladder cancer after intravesical bacillus calmette-guerin instillation. Korean J Urol 51(3):165–170
Serth J, Kuczyk MA, Bokemeyer C et al (1995) p53 immunohistochemistry as an independent prognostic factor for superficial transitional cell carcinoma of the bladder. Br J Cancer 71:201–205
Shariat SF, Ashfaq R, Sagalowsky AL et al (2007) Predictive value of cell cycle biomarkers in non muscle invasive bladder transitional cell carcinoma. J Urol 177:481–487
Shariat SF, Margulis V, Lotan Y, Montorsi F, Karakiewicz PI (2008) Nomograms for bladder cancer. Eur Urol 54:41–53
Shariat SF, Bolenz C, Godoy G et al (2009) Predictive value of combined immunohistochemical markers in patients with pT1 urothelial carcinoma at radical cystectomy. J Urol 182:78–84
Spiess PE, Agarwal N, Bangs R et al (2017) Clinical practice guidelines. J Natl Compr Cancer Netw 15:1240
Sylvester RJ, van der Meijden AP, Oosterlinck W et al (2006) Predicting recurrence and progression in individual patients with stage Ta T1 bladder cancer using EORTC risk tables: a combined analysis of 2596 patients from seven EORTC trials. Eur Urol 49:466–477
van Kessel KEM, van der Keur KA, Dyrskjøt L et al (2018) Molecular markers increase precision of the European Association of Urology non-muscle-invasive bladder Cancer Progression Risk Groups. Clin Cancer Res 24(7):1586–1593
Vetterlein MW, Roschinski J, Gild P et al (2017) Impact of the Ki-67 labeling index and p53 expression status on disease-free survival in pT1 urothelial carcinoma of the bladder. Transl Androl Urol 6(6):1018–1026
Xylinas E, Kent M, Kluth L et al (2013) Accuracy of the EORTC risk tables and of the CUETO scoring model to predict outcomes in non-muscle-invasive urothelial carcinoma of the bladder. Br J Cancer 109(6):1460–1466
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Authors reported no conflicts of interest.
Ethical approval
This study complies with WHO ethical standards. Patients were not subject to intervention or exposed to manipulation, intervention, or other interaction with investigators either directly or through alteration of their environment; patients in this study cannot be individually identified through investigator’s collection, preparation, or use of biological material or medical or other records.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chirife, A.M., Villasante, N., Rojas Bilbao, É. et al. Individual patient risk of progression of urinary bladder papillary tumors estimated from biomarkers at initial transurethral resection of bladder tumor. J Cancer Res Clin Oncol 145, 1709–1718 (2019). https://doi.org/10.1007/s00432-019-02923-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-019-02923-1