Abstract
Background
Emerging evidences show that G-protein-coupled estrogen receptor (GPER) can regulate the progression of various cancers, while its roles in the progression of osteosarcoma (OS) are not well illustrated.
Methods
The expression of GPER in OS cells and tissues were checked. Its roles in cell migration and expression of Snail was checked by use of its agonist G-1.
Results
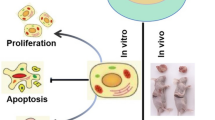
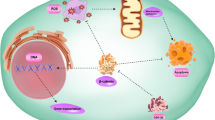
We found that the expression of GPER in OS cells and tissues were lower than that in their corresponding controls. OS patients with higher levels of GPER showed increased overall survival rate (OS) as compared with the lower ones. The activator of GPER (G-1) or overexpression of GPER can inhibit the migration and invasion of OS cells and downregulate mesenchymal markers. G-1 can rapidly decrease the expression of Snail, one powerful epithelial–mesenchymal transition transcription factor (EMT-TF). Overexpression of Snail can attenuate the suppression effects of G-1 on migration of OS cells, suggesting that Snail was involved in GPER-regulated migration of OS cells. Mechanically, G-1 rapidly decreased the protein of Snail but had no effect on its mRNA expression. This was because G-1 can decrease the protein stability of Snail. Further, G-1 increased the expression of FBXL5, which can trigger the proteasome-mediated degradation of Snail. Knockdown of FBXL5 can reverse G-1-induced downregulation of Snail in OS cells.
Conclusion
Activation of GPER can suppress the migration and invasion of OS cells via FBXL5-mediated post-translational down regulation of Snail. It suggested that targeted activation of GPER might be a potent potential therapy approach to overcome the metastasis of OS patients.






Similar content being viewed by others
References
Anderson ME (2016) Update on survival in osteosarcoma. Orthop Clin N Am 47:283–292
Azmi AS, Muqbil I, Wu J, Aboukameel A, Senapedis W, Baloglu E, Bollig-Fischer A, Dyson G, Kauffman M, Landesman Y et al (2015) Targeting the nuclear export protein XPO1/CRM1 reverses epithelial to mesenchymal transition. Sci Rep 5:16077
Brabletz T, Kalluri R, Nieto MA, Weinberg RA (2018) EMT in cancer. Nat Rev Cancer 18:128–128+
Bustos V, Nolan AM, Nijhuis A, Harvey H, Parker A, Poulsom R, McBryan J, Thomas W, Silver A, Harvey BJ (2017) GPER mediates differential effects of estrogen on colon cancer cell proliferation and migration under normoxic and hypoxic conditions. Oncotarget 8:84258–84275
Chen P, Wang H, Duan Z, Zou JX, Chen H, He W, Wang J (2014) Estrogen-related receptor alpha confers methotrexate resistance via attenuation of reactive oxygen species production and P53 mediated apoptosis in osteosarcoma cells. Biomed Res Int 2014:616025
Chen Y, Zhang K, Li Y, He Q (2017) Estrogen-related receptor alpha participates transforming growth factor-beta (TGF-beta) induced epithelial-mesenchymal transition of osteosarcoma cells. Cell Adhes Migr 11:338–346
Chou AJ, Geller DS, Gorlick R (2008) Therapy for osteosarcoma: where do we go from here? Paediatr Drugs 10:315–327
Daw NC, Chou AJ, Jaffe N, Rao BN, Billups CA, Rodriguez-Galindo C, Meyers PA, Huh WW (2015) Recurrent osteosarcoma with a single pulmonary metastasis: a multi-institutional review. Br J Cancer 112:278–282
Fang D, Yang H, Lin J, Teng Y, Jiang Y, Chen J, Li Y (2015) 17beta-estradiol regulates cell proliferation, colony formation, migration, invasion and promotes apoptosis by upregulating miR-9 and thus degrades MALAT-1 in osteosarcoma cell MG-63 in an estrogen receptor-independent manner. Biochem Biophys Res Commun 457:500–506
Fang S, Yu L, Mei H, Yang J, Gao T, Cheng A, Guo W, Xia K, Liu G (2016) Cisplatin promotes mesenchymal-like characteristics in osteosarcoma through Snail. Oncol Lett 12:5007–5014
Feldman RD, Ding Q, Hussain Y, Limbird LE, Pickering JG, Gros R (2016) Aldosterone mediates metastatic spread of renal cancer via the G protein-coupled estrogen receptor (GPER). FASEB J 30:2086–2096
Fic A, Mlakar SJ, Juvan P, Mlakar V, Marc J, Dolenc MS, Broberg K, Masic LP (2015) Genome-wide gene expression profiling of low-dose, long-term exposure of human osteosarcoma cells to bisphenol A and its analogs bisphenols AF and S. Toxicol In Vitro 29:1060–1069
Filardo EJ (2018) A role for G-protein coupled estrogen receptor (GPER) in estrogen-induced carcinogenesis: dysregulated glandular homeostasis, survival and metastasis. J Steroid Biochem Mol Biol 176:38–48
Filardo EJ, Quinn JA, Bland KI, Frackelton AR Jr (2000) Estrogen-induced activation of Erk-1 and Erk-2 requires the G protein-coupled receptor homolog, GPR30, and occurs via trans-activation of the epidermal growth factor receptor through release of HB-EGF. Mol Endocrinol (Baltim MD) 14:1649–1660
Hattinger CM, Fanelli M, Tavanti E, Vella S, Ferrari S, Picci P, Serra M (2015) Advances in emerging drugs for osteosarcoma. Expert Opin Emerg Drugs 20:495–514
Kansara M, Teng MW, Smyth MJ, Thomas DM (2014) Translational biology of osteosarcoma. Nat Rev Cancer 14:722–735
Liang S, Chen Z, Jiang G, Zhou Y, Liu Q, Su Q, Wei W, Du J, Wang H (2017) Activation of GPER suppresses migration and angiogenesis of triple negative breast cancer via inhibition of NF-kappaB/IL-6 signals. Cancer Lett 386:12–23
Maggiolini M, Picard D (2010) The unfolding stories of GPR30, a new membrane-bound estrogen receptor. J Endocrinol 204:105–114
Marjon NA, Hu C, Hathaway HJ, Prossnitz ER (2014) G protein-coupled estrogen receptor regulates mammary tumorigenesis and metastasis. Mol Cancer Res 12:1644–1654
Meazza C, Scanagatta P (2016) Metastatic osteosarcoma: a challenging multidisciplinary treatment. Expert Rev Anticancer 16:543–556
Meng X, Zhu Y, Tao L, Zhao S, Qiu S (2018) miR-590-3p mediates melatonin-induced cell apoptosis by targeting septin 7 in the human osteoblast cell line hFOB 1.19. Mol Med Rep 17:7202–7208
Molina L, Figueroa CD, Bhoola KD, Ehrenfeld P (2017) GPER-1/GPR30 a novel estrogen receptor sited in the cell membrane: therapeutic coupling to breast cancer. Expert Opin Ther Targets 21:755–766
Nieto MA (2002) The snail superfamily of zinc-finger transcription factors. Nat Rev Mol Cell Biol 3:155–166
Revankar CM, Cimino DF, Sklar LA, Arterburn JB, Prossnitz ER (2005) A transmembrane intracellular estrogen receptor mediates rapid cell signaling. Science 307:1625–1630
Salvatori L, Caporuscio F, Coroniti G, Starace G, Frati L, Russo MA, Petrangeli E (2009) Down-regulation of epidermal growth factor receptor induced by estrogens and phytoestrogens promotes the differentiation of U2OS human osteosarcoma cells. J Cell Physiol 220:35–44
Svoboda M, Hamilton G, Thalhammer T (2010) Steroid hormone metabolizing enzymes in benign and malignant human bone tumors. Expert Opin Drug Metab Toxicol 6:427–437
Tang Z, Li C, Kang B, Gao G, Li C, Zhang Z (2017) GEPIA: a web server for cancer and normal gene expression profiling and interactive analyses. Nucleic Acids Res 45:W98–W102
Vinas-Castells R, Beltran M, Valls G, Gomez I, Garcia JM, Montserrat-Sentis B, Baulida J, Bonilla F, de Herreros AG, Diaz VM (2010) The hypoxia-controlled FBXL14 ubiquitin ligase targets SNAIL1 for proteasome degradation. J Biol Chem 285:3794–3805
Vinas-Castells R, Frias A, Robles-Lanuza E, Zhang K, Longmore GD, Garcia de Herreros A, Diaz VM (2014) Nuclear ubiquitination by FBXL5 modulates Snail1 DNA binding and stability. Nucleic Acids Res 42:1079–1094
Wei W, Chen ZJ, Zhang KS, Yang XL, Wu YM, Chen XH, Huang HB, Liu HL, Cai SH, Du J et al (2014) The activation of G protein-coupled receptor 30 (GPR30) inhibits proliferation of estrogen receptor-negative breast cancer cells in vitro and in vivo. Cell Death Dis 5:e1428
Yang M, Liu B, Jin L, Tao H, Yang Z (2017) Estrogen receptor beta exhibited anti-tumor effects on osteosarcoma cells by regulating integrin, IAP, NF-kB/BCL-2 and PI3K/Akt signal pathway. J Bone Oncol 9:15–20
Yen M-L, Chien C-C, Chiu I-M, Huang H-I, Chen Y-C, Hu H-I, Yen BL (2007) Multilineage differentiation and characterization of the human fetal osteoblastic 1.19 cell line: a possible in vitro model of human mesenchymal progenitors. Stem Cells (Dayt Ohio) 25:125–131
Zheng H, Shen M, Zha YL, Li W, Wei Y, Blanco MA, Ren G, Zhou T, Storz P, Wang HY et al (2014) PKD1 phosphorylation-dependent degradation of SNAIL by SCF-FBXO11 regulates epithelial-mesenchymal transition and metastasis. Cancer Cell 26:358–373
Zhou BP, Deng J, Xia W, Xu J, Li YM, Gunduz M, Hung MC (2004) Dual regulation of Snail by GSK-3beta-mediated phosphorylation in control of epithelial-mesenchymal transition. Nat Cell Biol 6:931–940
Zhu G, Huang Y, Wu C, Wei D, Shi Y (2016) Activation of G-protein-coupled estrogen receptor inhibits the migration of human nonsmall cell lung cancer cells via IKK-beta/NF-kappaB signals. DNA Cell Biol 35:434–442
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Research involving human participants and/or animals
No human or animal study.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Wang, Z., Chen, X., Zhao, Y. et al. G-protein-coupled estrogen receptor suppresses the migration of osteosarcoma cells via post-translational regulation of Snail. J Cancer Res Clin Oncol 145, 87–96 (2019). https://doi.org/10.1007/s00432-018-2768-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-018-2768-4