Abstract
Purpose
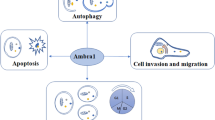
Autophagy, as a highly conserved cellular degradation and recycling process, plays an important part in maintaining cellular homeostasis. PKC signaling is involved in multiple pathways including cell cycle progression, tumorigenesis, migration and autophagy.
Methods
Literatures about PKC and autophagy from PubMed databases were reviewed in this study.
Results
Studies regarding the association of PKC and autophagy remain debatable. Different duration of the stimulation of autophagy and distinct cell contexts result in different function of PKC in regulating autophagy. The subcellular localization of PKCs and their downstream regulators may influence the autophagy regulation as well. As important intracellular components, the mitochondria play an important role in regulating autophagy, by metabolic modulation and structural derangement.
Conclusion
Phase II studies regarding PKC-β inhibitor, enzastaurin, showed promising results in MCL, DLBCL and recurrent high-grade gliomas. However, the detailed mechanism is still in need. The mechanism of PKC-β in mediating autophagy in lymphoma and high-grade gliomas remains elusive as well. Moreover, several studies were in agreement that rottlerin enhanced autophagy in breast cancer cells, which warrants further clinical studies to verify PKC-δ as a therapeutic target. Thus, identifying the function of PKC in modulating autophagy and conducting related clinical studies help find novel target for chemotherapy.



Similar content being viewed by others
References
Akar U, Ozpolat B, Mehta K, Fok J, Kondo Y, Lopez-Berestein G (2007) Tissue transglutaminase inhibits autophagy in pancreatic cancer cells. Mol Cancer Res 5:241–249. https://doi.org/10.1158/1541-7786.MCR-06-0229
Al Rawi S et al (2011) Postfertilization autophagy of sperm organelles prevents paternal mitochondrial DNA transmission. Science 334:1144–1147. https://doi.org/10.1126/science.1211878
Almami I, Dickenson JM, Hargreaves AJ, Bonner PL (2014) Modulation of transglutaminase 2 activity in H9c2 cells by PKC and PKA signalling: a role for transglutaminase 2 in cytoprotection. Br J Pharmacol 171:3946–3960. https://doi.org/10.1111/bph.12756
Berardi DE et al (2016) PKCdelta inhibition impairs mammary cancer proliferative capacity but selects cancer stem cells involving autophagy. J Cell Biochem 117:730–740. https://doi.org/10.1002/jcb.25358
Chen JL, Lin HH, Kim KJ, Lin A, Forman HJ, Ann DK (2008) Novel roles for protein kinase Cdelta-dependent signaling pathways in acute hypoxic stress-induced autophagy. J Biol Chem 283:34432–34444. https://doi.org/10.1074/jbc.M804239200
Choi SW, Song JK, Yim YS, Yun HG, Chun KH (2015) Glucose deprivation triggers protein kinase C-dependent beta-catenin proteasomal degradation. J Biol Chem 290:9863–9873. https://doi.org/10.1074/jbc.M114.606756
Coward J et al (2009) Safingol (L-threo-sphinganine) induces autophagy in solid tumor cells through inhibition of PKC and the PI3-. kinase pathway. Autophagy 5:184–193
D’Eletto M et al (2009) Transglutaminase 2 is involved in autophagosome maturation. Autophagy 5:1145–1154
D’Eletto M et al (2012) Type 2 transglutaminase is involved in the autophagy-dependent clearance of ubiquitinated proteins. Cell Death Differ 19:1228–1238. https://doi.org/10.1038/cdd.2012.2
Facchiano F, Facchiano A, Facchiano AM (2006) The role of transglutaminase-2 and its substrates in human diseases. Front Biosci 11:1758–1773
Fields AP, Murray NR (2008) Protein kinase C isozymes as therapeutic targets for treatment of human cancers. Adv Enzyme Regul 48:166–178. https://doi.org/10.1016/j.advenzreg.2007.11.014
Fine HA et al (2005) Results from phase II trial of enzastaurin (LY317615) in patients with recurrent high grade gliomas. J Clin Oncol 23:115S
Garg R, Benedetti LG, Abera MB, Wang H, Abba M, Kazanietz MG (2014) Protein kinase C and cancer: what we know and what we do not. Oncogene 33:5225–5237. https://doi.org/10.1038/onc.2013.524
Gomel R, Xiang C, Finniss S, Lee HK, Lu W, Okhrimenko H, Brodie C (2007) The localization of protein kinase Cdelta in different subcellular sites affects its proapoptotic and antiapoptotic functions and the activation of distinct downstream signaling pathways. Mol Cancer Res 5:627–639. https://doi.org/10.1158/1541-7786.MCR-06-0255
Gomes LC, Di Benedetto G, Scorrano L (2011) During autophagy mitochondria elongate, are spared from degradation and sustain cell viability. Nat Cell Biol 13:589–598. https://doi.org/10.1038/ncb2220
Gonnella R, Granato M, Farina A, Santarelli R, Faggioni A, Cirone M (2015) PKC theta and p38 MAPK activate the EBV lytic cycle through autophagy induction. Biochim Biophys Acta 1853:1586–1595. https://doi.org/10.1016/j.bbamcr.2015.03.011
Gutierrez MG, Master SS, Singh SB, Taylor GA, Colombo MI, Deretic V (2004) Autophagy is a defense mechanism inhibiting BCG and Mycobacterium tuberculosis survival in infected macrophages. Cell 119:753–766. https://doi.org/10.1016/j.cell.2004.11.038
Heras-Sandoval D, Perez-Rojas JM, Hernandez-Damian J, Pedraza-Chaverri J (2014) The role of PI3K/AKT/mTOR pathway in the modulation of autophagy and the clearance of protein aggregates in neurodegeneration. Cell Signal 26:2694–2701. https://doi.org/10.1016/j.cellsig.2014.08.019
Hill BG, Benavides GA, Lancaster JR Jr, Ballinger S, Dell’Italia L, Jianhua Z, Darley-Usmar VM (2012) Integration of cellular bioenergetics with mitochondrial quality control and autophagy. Biol Chem 393:1485–1512. https://doi.org/10.1515/hsz-2012-0198
Huang C et al (2010) Autophagy and protein kinase C are required for cardioprotection by sulfaphenazole. Am J Physiol Heart Circ Physiol 298:H570–H579. https://doi.org/10.1152/ajpheart.00716.2009
Huang SP, Chien JY, Tsai RK (2015) Ethambutol induces impaired autophagic flux and apoptosis in the rat retina. Dis Model Mech 8:977–987. https://doi.org/10.1242/dmm.019737
Jaishy B et al (2015) Lipid-induced NOX2 activation inhibits autophagic flux by impairing lysosomal enzyme activity. J Lipid Res 56:546–561. https://doi.org/10.1194/jlr.M055152
Jiang H, Cheng D, Liu W, Peng J, Feng J (2010) Protein kinase C inhibits autophagy and phosphorylates LC3. Biochem Biophys Res Commun 395:471–476. https://doi.org/10.1016/j.bbrc.2010.04.030
Jin Y, Bai Y, Ni H, Qiang L, Ye L, Shan Y, Zhou M (2016) Activation of autophagy through calcium-dependent AMPK/mTOR and PKCtheta pathway causes activation of rat hepatic stellate cells under hypoxic stress. FEBS Lett 590:672–682. https://doi.org/10.1002/1873-3468.12090
Klionsky DJ (2008) Autophagy revisited: a conversation with Christian de Duve. Autophagy 4:740–743
Kumar D, Shankar S, Srivastava RK (2013) Rottlerin-induced autophagy leads to the apoptosis in breast cancer stem cells: molecular mechanisms. Mol Cancer 12:171. https://doi.org/10.1186/1476-4598-12-171
Kumar R et al (2016) MicroRNA 17-5p regulates autophagy in Mycobacterium tuberculosis-infected macrophages by targeting Mcl-1 and STAT3. Cell Microbiol 18:679–691. https://doi.org/10.1111/cmi.12540
Liang N et al (2013) ATM pathway is essential for ionizing radiation-induced autophagy. Cell Signal 25:2530–2539. https://doi.org/10.1016/j.cellsig.2013.08.010
Liesa M, Shirihai OS (2013) Mitochondrial dynamics in the regulation of nutrient utilization and energy expenditure. Cell Metab 17:491–506. https://doi.org/10.1016/j.cmet.2013.03.002
Madaro L, Marrocco V, Carnio S, Sandri M, Bouche M (2013) Intracellular signaling in ER stress-induced autophagy in skeletal muscle cells. FASEB J 27:1990–2000. https://doi.org/10.1096/fj.12-215475
Morschhauser F et al (2008) A phase II study of enzastaurin, a protein kinase C beta inhibitor, in patients with relapsed or refractory mantle cell lymphoma. Ann Oncol 19:247–253. https://doi.org/10.1093/annonc/mdm463
Murray NR, Kalari KR, Fields AP (2011) Protein kinase Ciota expression and oncogenic signaling mechanisms in cancer. J Cell Physiol 226:879–887. https://doi.org/10.1002/jcp.22463
Parzych KR, Klionsky DJ (2014) An overview of autophagy: morphology, mechanism, and regulation. Antioxid Redox Signal 20:460–473. https://doi.org/10.1089/ars.2013.5371
Patergnani S, Marchi S, Rimessi A, Bonora M, Giorgi C, Mehta KD, Pinton P (2013) PRKCB/protein kinase C, beta and the mitochondrial axis as key regulators of autophagy. Autophagy 9:1367–1385. https://doi.org/10.4161/auto.25239
Puigserver P, Spiegelman BM (2003) Peroxisome proliferator-activated receptor-gamma coactivator 1 alpha (PGC-1 alpha): transcriptional coactivator and metabolic regulator. Endocr Rev 24:78–90. https://doi.org/10.1210/er.2002-0012
Qu L, Li G, Xia D, Hongdu B, Xu C, Lin X, Chen Y (2016) PRKCI negatively regulates autophagy via PIK3CA/AKT-MTOR signaling. Biochem Biophys Res Commun 470:306–312. https://doi.org/10.1016/j.bbrc.2016.01.059
Rambold AS, Kostelecky B, Elia N, Lippincott-Schwartz J (2011) Tubular network formation protects mitochondria from autophagosomal degradation during nutrient starvation. Proc Natl Acad Sci USA 108:10190–10195. https://doi.org/10.1073/pnas.1107402108
Renna M et al (2013) IGF-1 receptor antagonism inhibits autophagy. Hum Mol Genet 22:4528–4544. https://doi.org/10.1093/hmg/ddt300
Robert G et al (2009) Acadesine kills chronic myelogenous leukemia (CML) cells through PKC-dependent induction of autophagic cell death. PLoS One 4:e7889. https://doi.org/10.1371/journal.pone.0007889
Robertson MJ et al (2007) Phase II study of enzastaurin, a protein kinase C beta inhibitor, in patients with relapsed or refractory diffuse large B-cell lymphoma. J Clin Oncol Off J Am Soc Clin Oncol 25:1741–1746. https://doi.org/10.1200/JCO.2006.09.3146
Sakaki K, Kaufman RJ (2008) Regulation of ER stress-induced macroautophagy by protein kinase C. Autophagy 4:841–843
Sakaki K, Wu J, Kaufman RJ (2008) Protein kinase Ctheta is required for autophagy in response to stress in the endoplasmic reticulum. J Biol Chem 283:15370–15380. https://doi.org/10.1074/jbc.M710209200
Sandoval H, Thiagarajan P, Dasgupta SK, Schumacher A, Prchal JT, Chen M, Wang J (2008) Essential role for Nix in autophagic maturation of erythroid cells. Nature 454:232–235. https://doi.org/10.1038/nature07006
Shahnazari S et al (2010) A diacylglycerol-dependent signaling pathway contributes to regulation of antibacterial autophagy. Cell Host Microb 8:137–146. https://doi.org/10.1016/j.chom.2010.07.002
Shintani T, Klionsky DJ (2004) Autophagy in health and disease: a double-edged sword. Science 306:990–995. https://doi.org/10.1126/science.1099993
Silva RD, Manon S, Goncalves J, Saraiva L, Corte-Real M (2011) Modulation of Bax mitochondrial insertion and induced cell death in yeast by mammalian protein kinase. Calpha Exp Cell Res 317:781–790. https://doi.org/10.1016/j.yexcr.2010.12.001
So KY, Oh SH (2016) Cadmium-induced heme-oxygenase-1 expression plays dual roles in autophagy and apoptosis and is regulated by both PKC-delta and PKB/Akt activation in NRK52E kidney cells. Toxicology 370:49–59. https://doi.org/10.1016/j.tox.2016.09.010
Tan SH, Shui G, Zhou J, Li JJ, Bay BH, Wenk MR, Shen HM (2012) Induction of autophagy by palmitic acid via protein kinase C-mediated signaling pathway independent of mTOR (mammalian target of rapamycin. J Biol Chem 287:14364–14376. https://doi.org/10.1074/jbc.M111.294157
Tanida I (2011) Autophagosome formation and molecular mechanism of autophagy. Antioxid Redox Signal 14:2201–2214. https://doi.org/10.1089/ars.2010.3482
Torricelli C et al (2015) Phosphorylation-independent mTORC1 inhibition by the autophagy inducer. Rottlerin Cancer Lett 360:17–27. https://doi.org/10.1016/j.canlet.2015.01.040
Toton E, Romaniuk A, Budzianowski J, Hofmann J, Rybczynska M (2016) Zapotin (5,6,2′,6′-tetramethoxyflavone) modulates the crosstalk between autophagy and apoptosis pathways in cancer cells with overexpressed constitutively active. PKC Nutr Cancer 68:290–304. https://doi.org/10.1080/01635581.2016.1134595
Uekita T, Fujii S, Miyazawa Y, Hashiguchi A, Abe H, Sakamoto M, Sakai R (2013) Suppression of autophagy by CUB domain-containing protein 1 signaling is essential for anchorage-independent survival of lung cancer cells. Cancer Sci 104:865–870. https://doi.org/10.1111/cas.12154
Wang CW, Klionsky DJ (2003) The molecular mechanism of autophagy. Mol Med 9:65–76
Wang BS et al (2013) PKCiota counteracts oxidative stress by regulating Hsc70 in an esophageal cancer cell line. Cell Stress Chaperones 18:359–366. https://doi.org/10.1007/s12192-012-0389-4
Wang BS et al (2014) Inhibition of atypical protein kinase Ciota induces apoptosis through autophagic degradation of beta-catenin in esophageal cancer cells. Mol Carcinog 53:514–525. https://doi.org/10.1002/mc.22003
Wang F et al (2017) Protein kinase C-alpha suppresses autophagy and induces neural tube defects via miR-129-2 in diabetic pregnancy. Nat Commun 8:15182. https://doi.org/10.1038/ncomms15182
Wei H et al (2016) cPKCgamma-modulated autophagy in neurons alleviates ischemic injury in brain of mice with ischemic stroke through Akt-mTOR. Pathw Transl Stroke Res 7:497–511. https://doi.org/10.1007/s12975-016-0484-4
Weng LQ et al (2014) Aliskiren ameliorates pressure overload-induced heart hypertrophy and fibrosis in mice. Acta Pharmacol Sin 35:1005–1014. https://doi.org/10.1038/aps.2014.45
Xie Y, Kang R, Sun X, Zhong M, Huang J, Klionsky DJ, Tang D (2015) Posttranslational modification of autophagy-related proteins. in macroautophagy. Autophagy 11:28–45. https://doi.org/10.4161/15548627.2014.984267
Yu J et al (2013) Pseudolaric acid B induced cell cycle arrest, autophagy and senescence in murine fibrosarcoma l929 cell. Int J Med Sci 10:707–718. https://doi.org/10.7150/ijms.5726
Zhang Y, Wu Y, Tashiro S, Onodera S, Ikejima T (2009) Involvement of PKC signal pathways in oridonin-induced autophagy in HeLa cells: a protective mechanism against apoptosis. Biochem Biophys Res Commun 378:273–278. https://doi.org/10.1016/j.bbrc.2008.11.038
Zhang D, Xu X, Dong Z (2017) PRKCD/PKCdelta contributes to nephrotoxicity during cisplatin chemotherapy by suppressing autophagy. Autophagy 13:631–632. https://doi.org/10.1080/15548627.2016.1269990
Zhao M, Xia L, Chen GQ (2012) Protein kinase cdelta in apoptosis: a brief overview. Arch Immunol Ther Exp (Warsz) 60:361–372. https://doi.org/10.1007/s00005-012-0188-8
Zhou YY, Li Y, Jiang WQ, Zhou LF (2015) MAPK/JNK signalling: a potential autophagy regulation pathway Biosci Rep. https://doi.org/10.1042/BSR20140141
Acknowledgements
We are grateful to the staff at the NHC Key Laboratory of Radiobiology (Ministry of Health).
Funding
This study was supported by National Natural Science Foundation of China (NSFC) grants [grant number 31500682].
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Tianyi Wang declares that he has no conflict of interest. Conghe Liu declares that she has no conflict of interest. Lili Jia declares that she has no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Rights and permissions
About this article
Cite this article
Wang, T., Liu, C. & Jia, L. The roles of PKCs in regulating autophagy. J Cancer Res Clin Oncol 144, 2303–2311 (2018). https://doi.org/10.1007/s00432-018-2731-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-018-2731-4