Abstract
Purpose
Ovarian cancer has a high mortality rate and up to now no reliable molecular prognostic biomarkers have been established. During malignant progression, the cytoskeleton is strongly altered. Hence we analyzed if expression of certain cytoskeleton-associated proteins is correlated with clinical outcome of ovarian cancer patients.
Methods
First, in silico analysis was performed using the cancer genome atlas (TCGA), the human expression atlas and Pubmed. Selected candidates were validated on 270 ovarian cancer patients by qRT-PCR and/or by western blotting.
Results
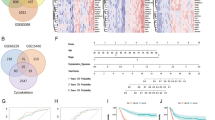
In silico analysis revealed that mRNAs of 214 cytoskeleton-associated proteins are detectable in ovarian cancer tissue. Among these, we selected 17 proteins that participate in cancer disease progression and cytoskeleton modulation: KIF14, KIF20A, KIF18A, ASPM, CEP55, DLGAP5, MAP9, EB1, KATNA1, DIAPH1, ANLN, SCIN, CCDC88A, FSCN1, GSN, VASP and CDC42. The first ten candidates interact with microtubules (MTs) and the others bind to actin filaments. Validation on clinical samples of ovarian cancer patients revealed that the expression levels of DIAPH1, EB1, KATNA1, KIF14 and KIF18A significantly correlated with clinical and histological parameters of ovarian cancer. High DIAPH1, EB1, KATNA1 and KIF14 protein levels were associated with increased overall survival (OAS) of ovarian cancer patients, while high DIAPH1 and EB1 protein levels were also associated with low differentiation of respective tumors (G2/3). Moreover, DIAPH1 was the only protein, whose expression significantly correlated with increased recurrence-free interval (RFI).
Conclusion
Mainly the expression levels of the MT-associated proteins analyzed in this study, correlated with prolonged survival of ovarian cancer patients. From > 200 genes initially considered, 17 cytoskeletal proteins are involved in cancer progression according to the literature. Among these, four proteins significantly correlated with improved survival of ovarian cancer patients.



Similar content being viewed by others
References
Akhmanova A, Steinmetz MO (2015) Control of microtubule organization and dynamics: two ends in the limelight. Nat Rev Mol Cell Biol 16:711–726
Almada E, Tonucci FM, Hidalgo F, Ferretti A, Ibarra S, Pariani A, Vena R, Favre C, Girardini J, Kierbel A, Larocca MC (2017) Akap350 Recruits Eb1 to The Spindle Poles, Ensuring Proper Spindle Orientation and Lumen Formation in 3d EpithelialCell Cultures. Sci Rep 7:14894
Arias-Romero LE, Chernoff J (2013) Targeting Cdc42 in cancer. Expert Opin Ther Targets 17:1263–1273
Brüning-Richardson A, Bond J, Alsiary R, Richardson J, Cairns DA, McCormack L, Hutson R, Burns P, Wilkinson N, Hall GD, Morrison EE (2011) Bell SM.ASPM and microcephalin expression in epithelial ovarian cancer correlates with tumour grade and survival. Br J Cancer 104:1602–1601
Chen J, Liu QJ, Wang D, Zhou XY, Xiong D, Li HJ, Li CL (2014) Hepatoma upregulated protein expression is involved in the pathogenesis of human breast carcinogenesis. Oncol Lett 8:2543–2548
Chen M, Yao S, Cao Q, Xia M (2017) Junxiu Liu, and Mian He The prognostic value of Ki67 in ovarian high-grade serous carcinoma: an 11-year cohort study of Chinese patients. Oncotarget 8:107877–107885
Choi JS, Kim KH, Oh E, Shin YK, Seo J, Kim SH, Park S, Choi YL (2017) Girdin protein expression is associated with poor prognosis in patients with invasive breast cancer. Pathology 49:618–626
Creekmore AL, Silkworth WT, Cimini D, Jensen RV, Roberts PC, Schmelz EM (2011) Changes in gene expression and cellular architecture in an ovarian cancer progression model. PLoS One 6:e17676
Dong X, Liu F, Sun L, Liu M, Li D, Su D, Zhu Z, Dong JT, Fu L, Zhou J (2010) Oncogenic function of microtubule end-binding protein 1 in breast cancer. J Pathol 220:361–369
Fu W, Wu H, Cheng Z, Huang S, Rao H (2018) The role of katanin p60 in breast cancer bone metastasis. Oncol Lett 15:4963–4969
Grabowski JP, Harter P, Heitz F, Pujade-Lauraine E, Reuss A, Kristensen G, Ray-Coquard I, Heitz, Pfisterer J, du Bois A (2016) Operability and chemotherapy responsiveness in advanced low-grade serous ovarian cancer. An analysis of the AGO Study Group metadatabase. Gynecol Oncol 140:457–462
Heinz LS, Muhs S, Schiewek J, Grüb S, Nalaskowski M, Lin YN, Wikman H, Oliveira-Ferrer L, Lange T, Wellbrock J, Konietzny A, Mikhaylova M, Windhorst S (2017) Strong fascin expression promotes metastasis independent of its F-actin bundling activity. Oncotarget 8:110077–110091
Idichi T, Seki N, Kurahara H, Yonemori K, Osako Y, Arai T, Okato A, Kita Y, Arigami T, Mataki Y, Kijima Y, Maemura K, Natsugoe S (2017) Regulation of actin-binding protein ANLN by antitumor miR-217 inhibits cancer cell aggressiveness in pancreatic ductal adenocarcinoma. Oncotarget 8:53180–53193
Inoda S, Hirohashi Y, Torigoe T, Nakatsugawa M, Kiriyama K, Nakazawa E, Harada K, Takasu H, Tamura Y, Kamiguchi K, Asanuma H, Tsuruma T, Terui T, Ishitani K, Ohmura T, Wang Q, Greene MI, Hasegawa T, Hirata K, Sato N (2009) Cep55/c10orf3, a tumor antigen derived from a centrosome residing protein in breast carcinoma. J Immunother 32:474–485
Jiang K, Rezabkova L, Hua S, Liu Q, Capitani G, Altelaar AFM, Heck AJR, Kammerer RA, Steinmetz MO, Akhmanova A (2017) Microtubule minus-end regulation at spindle poles by an ASPM-katanin complex. Nat Cell Biol 19:480–492
Kondoh E, Mori S, Yamaguchi K, Baba T, Matsumura N, Cory Barnett J, Whitaker RS, Konishi I, Fujii S, Berchuck A, Murphy SK (2010) Targeting slow-proliferating ovarian cancer cells. Int J Cancer 126:2448–2456
Lin YN, Bhuwania R, Gromova K, Failla AV, Lange T, Riecken K, Linder S, Kneussel M, Izbicki JR, Windhorst S (2015) Drosophila homologue of Diaphanous 1 (DIAPH1) controls the metastatic potential of colon cancer cells by regulating microtubule-dependent adhesion. Oncotarget 6:18577–18589
Liu JJ, Liu JY, Chen J, Wu YX, Yan P, Ji CD, Wang YX, Xiang DF, Zhang X, Zhang P, Cui YH, Wang JM, Bian XW, Qian F (2016) Scinderin promotes the invasion and metastasis of gastric cancer cells and predicts the outcome of patients. Cancer Lett 376:110–117
Matsuo M, Shimodaira T, Kasama T, Hata Y, Echigo A, Okabe M, Arai K, Makino Y, Niwa S, Saya H, Kishimoto T (2013) Katanin p60 contributes to microtubule instability around the midbody and facilitates cytokinesis in rat cells. PLoS One 8:e80392
Nehlig A, Molina A, Rodrigues-Ferreira S, Honoré S, Nahmias C (2017) Regulation of end-binding protein EB1 in the control of microtubule dynamics. Cell Mol Life Sci 74:2381–2393
Orr GA, Verdier-Pinard P, McDaid H, Horwitz SB (2003) Mechanisms of Taxol resistance related to microtubules. Oncogene 22:7280–7295
Poincloux R, Romao M, Montagnac G, Le Dez G, Bonne I, Rigaill G, Raposo G, Chavrier P (2009) Diaphanous-related formins are required for invadopodia formation and invasion of breast tumor cells. Cancer Res 69:2792–2800
Pula G, Krause M (2008) Role of Ena/VASP proteins in homeostasis and disease. Handb Exp Pharmacol 186:39–65
Rath O, Kozielski F (2012) Kinesins and cancer. Nat Rev Cancer 12:527–539
Rouquier S, Pillaire MJ, Cazaux C, Giorgi D (2014) Expression of the microtubule-associated protein MAP9/ASAP and its partners AURKA and PLK1 in colorectal and breast cancers. Dis Markers 798170
She ZY, Yang WX (2017) Molecular mechanisms of kinesin-14 motors in spindle assembly and chromosome segregation. J Cell Sci 130:2097–2110
Stangel D, Erkan M, Buchholz M, Gress T, Michalski C, Raulefs S, Friess H, Kleeff J (2015) Kif20a inhibition reduces migration and invasion of pancreatic cancer cells. J Surg Res 197:91–100
Tan DS, Agarwal R, Kaye SB (2006) Mechanisms of transcoelomic metastasis in ovarian cancer. Lancet Oncol 7:925–934
Thériault BL, Pajovic S, Bernardini MQ, Shaw PA, Gallie BL (2012) Kinesin family member 14: an independent prognostic marker and potential therapeutic target for ovarian cancer. Int J Cancer 130:1844–1854
Wang W, Shi Y, Li J, Cui W, Yang B. Up-regulation of KIF14 is a predictor of poor survival and a novel prognostic biomarker of chemoresistance to paclitaxel treatment in cervical cancer. Biosci Rep. 2016;36(2)
Wen Y, Eng CH, Schmoranzer J, Cabrera-Poch N, Morris EJ, Chen M, Wallar BJ, Alberts AS, Gundersen GG (2004) EB1 and APC bind to mDia to stabilize microtubules downstream of Rho and promote cell migration. Nat Cell Biol 6:820–830
Xu W, Mezencev R, Kim B, Wang L, McDonald J, Sulchek T (2012) Cell stiffness is a biomarker of the metastatic potential of ovarian cancer cells. PLoS One 7:e46609
Zhang C, Zhu C, Chen H, Li L, Guo L, Jiang W, Lu SH (2010) Kif18A is involved in human breast carcinogenesis. Carcinogenesis 31:1676–1684
Zhang Y, Wang F, Niu YJ, Liu HL, Rui R, Cui XS, Kim NH, Sun SC (2015) Formin mDia1, a downstream molecule of FMNL1, regulates Profilin1 for actin assembly and spindle organization during mouse oocyte meiosis. Biochim Biophys Acta 1853:317–327
Funding
This project was financially supported by initial financing of the UKE.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that there is no competing interest.
Ethical standards
Patients gave their written approvals for examining tissue samples and reviewing their medical records according to our investigational review board and ethics committee guidelines (#190504).
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Schiewek, J., Schumacher, U., Lange, T. et al. Clinical relevance of cytoskeleton associated proteins for ovarian cancer. J Cancer Res Clin Oncol 144, 2195–2205 (2018). https://doi.org/10.1007/s00432-018-2710-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-018-2710-9