Abstract
Purpose
To evaluate the prognostic value and pathobiological significance of Glasgow microenvironment score (GMS), a parameter based on tumor stroma percentage and inflammatory cell infiltration, in gastric cancer.
Methods
A total of 225 cases of gastric cancer were histologically reviewed, and GMS was evaluated for each case. The association between GMS and patients’ survival was investigated. Then the relationship between GMS and mismatch repair (MMR) status, Epstein–Barr virus (EBV) infection were determined using immunohistochemistry (IHC) and in situ hybridization, and the expression of PD1/PD-L1 was examined. Furthermore, the amount of cancer-associated fibroblasts (CAFs), the content and maturity of collagen components were detected using IHC, Picrosirius Red staining and second harmonic generation imaging.
Results
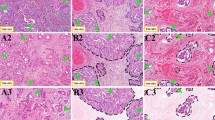
GMS was significantly associated with clinical outcomes of gastric cancer, and multivariate analysis indicated that GMS was an independent factor (HR 1.725, P = 0.002). Low GMS was a manifestation of better prognosis and inflammatory tumor microenvironment, which was related to MMR deficiency (P = 0.042) and EBV infection (P = 0.032), and within this microenvironment, expression of PD-L1 in carcinoma cells (P = 0.030) or in inflammatory cells (P = 0.029) was significantly higher. In contrast, high GMS linked to a poorer survival and desmoplastic stroma, in which there existed markedly increased CAFs and collagen deposition.
Conclusion
GMS can serve as a useful prognostic factor for gastric cancer, and according to GMS, the tumor microenvironment in this cancer type may be partially classified as inflammatory or desmoplastic microenvironment that possesses different pathobiological features.






Similar content being viewed by others
References
Alexander J, Cukierman E (2016) Stromal dynamic reciprocity in cancer: intricacies of fibroblastic-ECM interactions. Curr Opin Cell Biol 42:80–93
Alsina M, Moehler M, Hierro C, Guardeño R, Tabernero J (2016) Immunotherapy for gastric cancer: a focus on immune checkpoints. Target Oncol 11:469–477
Baldan V, Griffiths R, Hawkins RE, Gilham DE (2015) Efficient and reproducible generation of tumour-infiltrating lymphocytes for renal cell carcinoma. Br J Cancer 112:1510–1518
Berruti A, Piovesan A, Torta M, Raucci CA, Gorzegno G, Paccotti P, Dogliotti L, Angeli A (1996) Biochemical evaluation of bone turnover in cancer patients with bone metastases: relationship with radiograph appearances and disease extension. Br J Cancer 73:1581–1587
Calon A, Tauriello DV, Batlle E (2014) TGF-beta in CAF-mediated tumor growth and metastasis. Semin Cancer Biol 25:15–22
Chetty R (2012) Gastrointestinal cancers accompanied by a dense lymphoid component: an overview with special reference to gastric and colonic medullary and lymphoepithelioma-like carcinomas. J Clin Pathol 65:1062–1065
Conklin MW, Keely PJ (2012) Why the stroma matters in breast cancer: insights into breast cancer patient outcomes through the examination of stromal biomarkers. Cell Adh Migr 6:249–260
Dieci MV, Mathieu MC, Guarneri V, Conte P, Delaloge S, Andre F, Goubar A (2015) Prognostic and predictive value of tumor-infiltrating lymphocytes in two phase III randomized adjuvant breast cancer trials. Ann Oncol 26:1698–1704
Drifka CR, Tod J, Loeffler AG, Liu Y, Thomas GJ, Eliceiri KW, Kao WJ (2015) Periductal stromal collagen topology of pancreatic ductal adenocarcinoma differs from that of normal and chronic pancreatitis. Mod Pathol 28:1470–1480
Edge SB, Byrd DR, Compton CC, Fritz AG, Greene FL, Trotti A (2009) AJCC Cancer Staging Manual. Springer, New York
Egeblad M, Rasch M, Weaver VM (2010) Dynamic interplay between the collagen scaffold and tumor evolution. Curr Opin Cell Biol 22: 697–706
Fearon DT (2014) The carcinoma-associated fibroblast expressing fibroblast activation protein and escape from immune surveillance. Cancer. Immunol Res 2:187–193
Ferlay J, Soerjomataram I, Ervik M, Dikshit R, Eser S, Mathers C, Rebelo M, Parkin DM, Forman D, Bray F (2013) GLOBOCAN 2012 v1.0, Cancer Incidence and Mortality Worldwide: IARC CancerBase No. 11. Lyon, France: International Agency for Research on Cancer. http://globocan.iarc.fr. Accessed 20 Dec 2016
Freeman GJ, Long AJ, Iwai Y, Bourque K, Chernova T, Nishimura H, Fitz LJ, Malenkovich N, Okazaki T, Byrne MC, Horton HF, Fouser L, Carter L, Ling V, Bowman MR, Carreno BM, Collins M, Wood CR, Honjo T (2000) Engagement of the PD-1 immunoinhibitory receptor by a novel B7 family member leads to negative regulation of lymphocyte activation. J Exp Med 192:1027–1034
Grossman M, Benchetrit N, Zhuravlev A, Afik R, Bassat E, Solomonov I, Yarden Y, Sagi I (2016) Tumor cell invasion can be blocked by modulators of collagen fibril alignment that control assembly of the extracellular matrix. Cancer Res 76:4249–4258
Gujam FJ, Edwards J, Mohammed ZM, Going JJ, McMillan DC (2014) The relationship between the tumour stroma percentage, clinicopathological characteristics and outcome in patients with operable ductal breast cancer. Br J Cancer 111:157–165
Harper J, Sainson RC (2014) Regulation of the anti-tumour immune response by cancer-associated fibroblasts. Semin Cancer Biol 25:69–77
Hulmes DJ (2002) Building collagen molecules, fibrils, and suprafibrillar structures. J Struct Biol 137:2–10
Joyce JA, Pollard JW (2009) Microenvironmental regulation of metastasis. Nat Rev Cancer 9:239–252
Kang BW, Seo AN, Yoon S, Bae HI, Jeon SW, Kwon OK (2016) Prognostic value of tumor-infiltrating lymphocytes in Epstein-Barr virus-associated gastric cancer. Ann Oncol 27:494–501
Kauppila S, Stenback F, Risteli J, Jukkola A, Risteli L (1998) Aberrant type I and type III collagen gene expression in human breast cancer in vivo. J Pathol 186:262–268
Keikhosravi A, Bredfeldt JS, Sagar AK, Eliceiri KW (2014) Second harmonic generation imaging of cancer. Methods Cell Biol 123:531–546
Kim SY, Park C, Kim HJ, Park J, Hwang J, Kim JI, Choi MG, Kim S, Kim KM, Kang MS (2015) Deregulation of immune response genes in patients with Epstein-Barr virus-associated gastric cancer and outcomes. Gastroenterology 148:137–147
Kim HS, Shin SJ, Beom SH, Jung M, Choi YY, Son T (2016) Comprehensive expression profiles of gastric cancer molecular subtypes by immunohistochemistry: implications for individualized therapy. Oncotarget 7:44608–44620
Klintrup K, Mäkinen JM, Kauppila S, Väre PO, Melkko J, Tuominen H, Tuppurainen K, Mäkelä J, Karttunen TJ, Mäkinen MJ (2005) Inflammation and prognosis in colorectal cancer. Eur J Cancer 41:2645–2654
Lu J, Zhou S, Siech M, Seufferlein T, Bachem MG (2014) Pancreatic stellate cells promote hapto-migration of cancer cells through collagen I-mediated signalling pathway. Br J Cancer 110:409–420
Moserle L, Casanovas O (2013) Anti-angiogenesis and metastasis: a tumour and stromal cell alliance. J Intern Med 273:128–137
Mouw JK, Ou G, Weaver VM (2014) Extracellular matrix assembly: a multiscale deconstruction. Nat Rev Mol Cell Biol 15:771–785
Müerköster S, Wegehenkel K, Arlt A, Witt M, Sipos B, Kruse ML, Sebens T, Klöppel G, Kalthoff H, Fölsch UR, Schäfer H (2004) Tumor stroma interactions induce chemoresistance in pancreatic ductal carcinoma cells involving increased secretion and paracrine effects of nitric oxide and interleukin-1beta. Cancer Res 64:1331–1337
Murphy G, Pfeiffer R, Camargo MC, Rabkin CS (2009) Meta-analysis shows that prevalence of Epstein-Barr virus-positive gastric cancer differs based on sex and anatomic location. Gastroenterology 137:824–833
Navab R, Strumpf D, Bandarchi B, Zhu CQ, Pintilie M, Ramnarine VR, Ibrahimov E, Radulovich N, Leung L, Barczyk M, Panchal D, To C, Yun JJ, Der S, Shepherd FA, Jurisica I, Tsao MS (2011) Prognostic gene-expression signature of carcinoma-associated fibroblasts in non–small cell lung cancer. Proc Natl Acad Sci USA 108:7160–7165
Park JH, Richards CH, McMillan DC, Horgan PG, Roxburgh CS (2014) The relationship between tumour stroma percentage, the tumour microenvironment and survival in patients with primary operable colorectal cancer. Ann Oncol 25:644–651
Park JH, McMillan DC, Powell AG, Richards CH, Horgan PG, Edwards J (2015) Evaluation of a tumor microenvironment-based prognostic score in primary operable colorectal cancer. Clin Cancer Res 21:882–888
Provenzano PP, Inman DR, Eliceiri KW, Knittel JG, Yan L, Rueden CT, White JG, Keely PJ (2008) Collagen density promotes mammary tumor initiation and progression. BMC Med 6:11
Romano E, Romero P (2015) The therapeutic promise of disrupting the PD-1/PD-L1 immune checkpoint in cancer: unleashing the CD8 T cell mediated anti-tumor activity results in significant, unprecedented clinical efficacy in various solid tumors. J Immunother. Cancer 3:15
Roxburgh CS, McMillan DC (2012) The role of the in situ local inflammatory response in predicting recurrence and survival in patients with primary operable colorectal cancer. Cancer Treat Rev. 2012:451–466
Shields MA, Dangi-Garimella S, Redig AJ, Munshi HG (2012) Biochemical role of the collagen-rich tumour microenvironment in pancreatic cancer progression. Biochem J 441:541–552
Tosolini M, Kirilovsky A, Mlecnik B, Fredriksen T, Mauger S, Bindea G, Berger A, Bruneval P, Fridman WH, Pagès F, Galon J (2011) Clinical impact of different classes of infiltrating T cytotoxic and helper cells (Th1, th2, treg, th17) in patients with colorectal cancer. Cancer Res 71:1263–1271
Wang K, Ma W, Wang J, Yu L, Zhang X, Wang Z, Tan B, Wang N, Bai B, Yang S, Liu H, Zhu S, Cheng Y (2012) Tumor-stroma ratio is an independent predictor for survival in esophageal squamous cell carcinoma. J Thorac Oncol 7:1457–1461
Wang B, Wu S, Zeng H, Dong W, He W, Chen X, Dong X, Zheng L, Lin T, Huang J (2015) CD103(+)tumor infiltrating lymphocytes predict a favorable prognosis in urothelial cell carcinoma of the bladder. J Urol 194:556–562
Acknowledgements
This work was supported by National Basic Research Program of China (973 Program, No. 2010CB529400).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhou, ZH., Ji, CD., Zhu, J. et al. The prognostic value and pathobiological significance of Glasgow microenvironment score in gastric cancer. J Cancer Res Clin Oncol 143, 883–894 (2017). https://doi.org/10.1007/s00432-017-2346-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-017-2346-1