Abstract
Purpose
High-dose methotrexate (HD-MTX) is a cornerstone antineoplastic drug in most treatment protocols of pediatric acute lymphoblastic leukemia (ALL). Among the membrane efflux transporters of MTX, the human breast cancer resistant protein is the second member of the G subfamily of ATP-binding cassette (ABC) efflux pump (ABCG2). A single-nucleotide polymorphism (SNP) in ABCG2, the exchange of C to A at position 421, represents 13 % in the Middle Eastern population. We studied the effect of this SNP on the plasma levels of HD-MTX in Egyptian pediatric ALL.
Methods
Two hundred ALL patients were recruited from Children’s Cancer Hospital Egypt-57357, and all were treated according to the St Jude Total XV protocol. Determination of plasma MTX levels was done at 23, 42 and 68 h. Genotyping of C421A of ABCG2 was done by polymerase chain reaction-restriction fragment length polymorphism.
Results
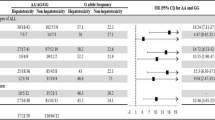
We found 14.5 % of the variant allele of the ABCG2 C421A SNP. The statistical association between ABCG2 421C>A SNP and the cutoff toxic plasma level of 24 h HD-MTX infusion at different time points tested was not statistically significant. There was no statistical significance between steady-state plasma concentration in patients with and without with this SNP.
Conclusion
To date, this is the largest study on Egyptian ALL patients for this SNP. This study shows that there is no effect of ABCG2 421C>A on plasma concentrations of HD-MTX. Replacing candidate gene association studies with genome-wide studies of HD-MTX is now mandatory and is part of our research blueprint.

Similar content being viewed by others
References
Abbott BL (2003) ABCG2 (BCRP) expression in normal and malignant hematopoietic cells. Hematol Oncol 21:115–130
Ackland SP, Schilsky RL (1987) High-dose methotrexate: a critical reappraisal. J Clin Oncol Off J Am Soc Clin Oncol 5:2017–2031
Bailey-Dell KJ, Hassel B, Doyle LA, Ross DD (2001) Promoter characterization and genomic organization of the human breast cancer resistance protein (ATP-binding cassette transporter G2) gene. Biochim Biophys Acta 1520:234–241
Benderra Z, Faussat A-M, Sayada L et al (2004) Breast cancer resistance protein and P-glycoprotein in 149 adult acute myeloid leukemias. Clin Cancer Res Off J Am Assoc Cancer Res 10:7896–7902
Dawson BD, Trapp R (2000) Basic and clinical biostatistics. Lange Medical Books/McGraw Hil, New York
Devita VT, Lawrence TS, Rosenberg SA, DePinto RAWR (2008) Pharmacology of cancer chemotherapy: antimetabolite—methotrexate. In: DeVita VT Jr, Hellman SRS (eds) Principles and practice of oncology, 8th edn. Lippincott Williams & Wilkins, Philadelphia, pp 427–428
Doyle LA, Ross DD (2003) Multidrug resistance mediated by the breast cancer resistance protein BCRP (ABCG2). Oncogene 22:7340–7358
El-Khodary NM, El-Haggar SM, Eid MA, Ebeid EN (2012) Study of the pharmacokinetic and pharmacogenetic contribution to the toxicity of high-dose methotrexate in children with acute lymphoblastic leukemia. Med Oncol (Northwood, London, England) 29:2053–2062
Gervasini G (2009) Polymorphisms in methotrexate pathways: what is clinically relevant, what is not, and what is promising. Curr Drug Metab 10:547–566
Hayashi H, Fujimaki C, Tsuboi S et al (2008) Application of fluorescence polarization immunoassay for determination of methotrexate-polyglutamates in rheumatoid arthritis patients. Tohoku J Exp Med 215:95–101
Hulot J-S, Villard E, Maguy A et al (2005) A mutation in the drug transporter gene ABCC2 associated with impaired methotrexate elimination. Pharmacogenet Genomics 15:277–285
Igarashi S, Manabe A, Ohara A et al (2005) No advantage of dexamethasone over prednisolone for the outcome of standard- and intermediate-risk childhood acute lymphoblastic leukemia in the Tokyo Children’s Cancer Study Group L95-14 protocol. J Clin Oncol Off J Am Soc Clin Oncol 23:6489–6498
Imanishi H, Okamura N, Yagi M et al (2007) Genetic polymorphisms associated with adverse events and elimination of methotrexate in childhood acute lymphoblastic leukemia and malignant lymphoma. J Hum Genet 52:166–171
Keskitalo JE, Zolk O, Fromm MF et al (2009) ABCG2 polymorphism markedly affects the pharmacokinetics of atorvastatin and rosuvastatin. Clin Pharmacol Ther 86:197–203
Lepper ER, Nooter K, Verweij J et al (2005) Mechanisms of resistance to anticancer drugs: the role of the polymorphic ABC transporters ABCB1 and ABCG2. Pharmacogenomics 6:115–138
Lopez-Lopez E, Martin-Guerrero I, Ballesteros J et al (2011) Polymorphisms of the SLCO1B1 gene predict methotrexate-related toxicity in childhood acute lymphoblastic leukemia. Pediatr Blood Cancer 57:612–619
Malaviya AN, Sharma A, Agarwal D et al (2010) Low-dose and high-dose methotrexate are two different drugs in practical terms. Int J Rheum Dis 13:288–293
Maliepaard M, Scheffer GL, Faneyte IF et al (2001) Subcellular localization and distribution of the breast cancer resistance protein transporter in normal human tissues. Cancer Res 61:3458–3464
Meissner K, Heydrich B, Jedlitschky G et al (2006) The ATP-binding cassette transporter ABCG2 (BCRP), a marker for side population stem cells, is expressed in human heart. J Histochem Cytochem Off J Histochem Soc 54:215–221
Metayer C, Milne E, Clavel J et al (2013) The childhood leukemia international consortium. Cancer Epidemiol 37:336–347
Mikkelsen TS, Sparreboom A, Cheng C et al (2011) Shortening infusion time for high-dose methotrexate alters antileukemic effects: a randomized prospective clinical trial. J Clin Oncol Off J Am Soc Clin Oncol 29:1771–1778
Miller SA, Dykes DD, Polesky HF (1988) A simple salting out procedure for extracting DNA from human nucleated cells. Nucleic Acids Res 16:1215
Ni Z, Bikadi Z, Rosenberg MF, Mao Q (2010) Structure and function of the human breast cancer resistance protein (BCRP/ABCG2). Curr Drug Metab 11:603–617
Pauley JL, Panetta JC, Crews KR et al (2013) Between-course targeting of methotrexate exposure using pharmacokinetically guided dosage adjustments. Cancer Chemother Pharmacol 72:369–378
Pession A, Valsecchi MG, Masera G et al (2005) Long-term results of a randomized trial on extended use of high dose L-asparaginase for standard risk childhood acute lymphoblastic leukemia. J Clin Oncol Off J Am Soc Clin Oncol 23:7161–7167
Pui C-H, Evans WE (2006) Treatment of acute lymphoblastic leukemia. N Engl J Med 354:166–178
Pui C-H, Campana D, Pei D et al (2009) Treating childhood acute lymphoblastic leukemia without cranial irradiation. N Engl J Med 360:2730–2741
Pui C-H, Mullighan CG, Evans WE, Relling MV (2012) Pediatric acute lymphoblastic leukemia: where are we going and how do we get there? Blood 120:1165–1174
Rau T, Erney B, Göres R et al (2006) High-dose methotrexate in pediatric acute lymphoblastic leukemia: impact of ABCC2 polymorphisms on plasma concentrations. Clin Pharmacol Ther 80:468–476
Robey RW, Polgar O, Deeken J et al (2007) ABCG2: determining its relevance in clinical drug resistance. Cancer Metastasis Rev 26:39–57
Schmiegelow K (2009) Advances in individual prediction of methotrexate toxicity: a review. Br J Haematol 146:489–503
Silverman LB, Gelber RD, Dalton VK et al (2001) Improved outcome for children with acute lymphoblastic leukemia: results of Dana-Farber Consortium Protocol 91-01. Blood 97:1211–1218
Simon N, Marsot A, Villard E, Choquet S, Khe HX, Zahr N, Lechat P, Leblond V, Hulot JS (2013) Impact of ABCC2 polymorphisms on high-dose methotrexate pharmacokinetics in patients with lymphoid malignancy. Pharmacogenomics J 13(6):507–513
Sissung TM, Baum CE, Kirkland CT et al (2010) Pharmacogenetics of membrane transporters: an update on current approaches. Mol Biotechnol 44:152–167
Stamp LK, O’Donnell JL, Chapman PT et al (2009) Determinants of red blood cell methotrexate polyglutamate concentrations in rheumatoid arthritis patients receiving long-term methotrexate treatment. Arthritis Rheum 60:2248–2256
Tantawy AAG, El-Bostany EA, Adly AAM et al (2010) Methylene tetrahydrofolate reductase gene polymorphism in Egyptian children with acute lymphoblastic leukemia. Blood Coagul Fibrinolysis Int J Haemost Thromb 21:28–34
Vlaming MLH, van Esch A, Pala Z et al (2009) Abcc2 (Mrp2), Abcc3 (Mrp3), and Abcg2 (Bcrp1) are the main determinants for rapid elimination of methotrexate and its toxic metabolite 7-hydroxymethotrexate in vivo. Mol Cancer Ther 8:3350–3359
Wilson CS, Davidson GS, Martin SB et al (2006) Gene expression profiling of adult acute myeloid leukemia identifies novel biologic clusters for risk classification and outcome prediction. Blood 108:685–696
Woodward OM, Köttgen A, Coresh J et al (2009) Identification of a urate transporter, ABCG2, with a common functional polymorphism causing gout. Proc Natl Acad Sci USA 106:10338–10342
Zamber CP, Lamba JK, Yasuda K et al (2003) Natural allelic variants of breast cancer resistance protein (BCRP) and their relationship to BCRP expression in human intestine. Pharmacogenetics 13:19–28
Acknowledgments
Mobinil award from Mobinil company as a support to Children’s Cancer Hospital Egypt-57357 (CCHE-57357) funded this study.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
El Mesallamy, H.O., Rashed, W.M., Hamdy, N.M. et al. High-dose methotrexate in Egyptian pediatric acute lymphoblastic leukemia: the impact of ABCG2 C421A genetic polymorphism on plasma levels, what is next?. J Cancer Res Clin Oncol 140, 1359–1365 (2014). https://doi.org/10.1007/s00432-014-1670-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-014-1670-y