Abstract
Objective
δ-Catenin is found to be involved in the progression of several human cancers. However, its expression pattern and biological roles in human ovarian cancers are not clear. In this study, we examined the expression pattern of δ-catenin in 149 ovarian cancer specimens. We also depleted and overexpressed δ-catenin expression in ovarian cancer cell lines and investigated its role in cell proliferation and invasion.
Methods
δ-Catenin expression was analyzed in 149 archived ovarian cancer specimens using immunohistochemistry. siRNA knockdown and plasmid transfection were performed in SKOV3, SW626, and OVCAR3 cell lines. MTT, colony formation assay, soft agar colony assay, and matrigel invasion assay were carried out to assess the role of δ-catenin in cell proliferation and invasion. We also performed cell cycle analysis in δ-catenin depleted and overexpressed cells. In addition, we examined the level of several cell cycle-related molecules using Western blot.
Results
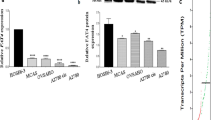
Of the 149 patients in the study, 104 (69.7 %) showed δ-catenin overexpression. δ-catenin overexpression positively correlated with advanced FIGO stage. δ-Catenin depletion in ovarian cancer cell lines inhibited ovarian cancer cell proliferation and invasion. Depletion of δ-catenin also blocked cell cycle progression and downregulated cyclin D1 expression in ovarian cancer cells. Overexpression of δ-catenin enhanced cell proliferation, invasion, and upregulated cyclinD1 expression.
Conclusions
δ-Catenin is overexpressed in ovarian cancers and associated with advanced stage. Our data provide evidence that δ-catenin regulates the ovarian cancer cell proliferation, invasion, and cell cycle. δ-Catenin thus has potential as a therapeutic target.



Similar content being viewed by others
References
Abu-Elneel K, Ochiishi T, Medina M et al (2008) A delta-catenin signaling pathway leading to dendritic protrusions. J Biol Chem 283(47):32781–32791
Arikkath J, Reichardt LF (2008) Cadherins and catenins at synapses: roles in synaptogenesis and synaptic plasticity. Trends Neurosci 31(9):487–494
Bali A, O’Brien PM, Edwards LS et al (2004) Cyclin D1, p53, and p21Waf1/Cip1 expression is predictive of poor clinical outcome in serous epithelial ovarian cancer. Clin Cancer Res 10(15):5168–5177
Darcy KM, Birrer MJ (2010) Translational research in the gynecologic oncology group: evaluation of ovarian cancer markers, profiles, and novel therapies. Gynecol Oncol 117(3):429–439
Despierre E, Lambrechts D, Neven P et al (2010) The molecular genetic basis of ovarian cancer and its roadmap towards a better treatment. Gynecol Oncol 117(2):358–365
Huang FY, Chiu PM, Tam KF et al (2006) Semi-quantitative fluorescent PCR analysis identifies PRKAA1 on chromosome 5 as a potential candidate cancer gene of cervical cancer. Gynecol Oncol 103(1):219–225
Jemal A, Siegel R, Xu J et al (2010) Cancer statistics, 2010. CA Cancer J Clin 60(5):277–300
Kawamura Y, Fan QW, Hayashi H et al (1999) Expression of the mRNA for two isoforms of neural plakophilin-related arm-repeat protein/delta-catenin in rodent neurons and glial cells. Neurosci Lett 277(3):185–188
Keum JS, Kong G, Yang SC et al (1999) Cyclin D1 overexpression is an indicator of poor prognosis in resectable non-small cell lung cancer. Br J Cancer 81(1):127–132
Kim K, Sirota A, Chen YH et al (2002) Dendrite-like process formation and cytoskeletal remodeling regulated by delta-catenin expression. Exp Cell Res 275(2):171–184
Kim H, Han JR, Park J et al (2008) Delta-catenin-induced dendritic morphogenesis. An essential role of p190RhoGEF interaction through Akt1-mediated phosphorylation. J Biol Chem 283(2):977–987
Knudsen KE, Diehl JA, Haiman CA et al (2006) Cyclin D1: polymorphism, aberrant splicing and cancer risk. Oncogene 25(11):1620–1628
Lu Q, Dobbs LJ, Gregory CW et al (2005) Increased expression of delta-catenin/neural plakophilin-related armadillo protein is associated with the down-regulation and redistribution of E-cadherin and p120ctn in human prostate cancer. Hum Pathol 36(10):1037–1048
Lu Q, Mukhopadhyay NK, Griffin JD et al (2002) Brain armadillo protein delta-catenin interacts with Abl tyrosine kinase and modulates cellular morphogenesis in response to growth factors. J Neurosci Res 67(5):618–624
Lu Q, Paredes M, Medina M et al (1999) Delta-catenin, an adhesive junction-associated protein which promotes cell scattering. J Cell Biol 144(3):519–532
Masamha CP, Benbrook DM (2009) Cyclin D1 degradation is sufficient to induce G1 cell cycle arrest despite constitutive expression of cyclin E2 in ovarian cancer cells. Cancer Res 69(16):6565–6572
Minghao W, Qianze D, Di Z et al (2011) Expression of delta-catenin is associated with progression of human astrocytoma. BMC Cancer 11:514
Ochiishi T, Futai K, Okamoto K et al (2008) Regulation of AMPA receptor trafficking by delta-catenin. Mol Cell Neurosci 39(4):499–507
Permuth-Wey J, Sellers TA (2009) Epidemiology of ovarian cancer. Methods Mol Biol 472:413–437
Ratschiller D, Heighway J, Gugger M et al (2003) Cyclin D1 overexpression in bronchial epithelia of patients with lung cancer is associated with smoking and predicts survival. J Clin Oncol 21(11):2085–2093
Roy PG, Thompson AM (2006) Cyclin D1 and breast cancer. Breast 15(6):718–727
Schwartz DR, Kardia SL, Shedden KA et al (2002) Gene expression in ovarian cancer reflects both morphology and biological behavior, distinguishing clear cell from other poor-prognosis ovarian carcinomas. Cancer Res 62(16):4722–4729
Singer G, Oldt R III, Cohen Y et al (2003) Mutations in BRAF and KRAS characterize the development of low-grade ovarian serous carcinoma. J Natl Cancer Inst 95(6):484–486
Zhang JY, Wang Y, Zhang D et al (2010) Delta-Catenin promotes malignant phenotype of non-small cell lung cancer by non-competitive binding to E-cadherin with p120ctn in cytoplasm. J Pathol 222(1):76–88
Zheng M, Simon R, Mirlacher M et al (2004) TRIO amplification and abundant mRNA expression is associated with invasive tumor growth and rapid tumor cell proliferation in urinary bladder cancer. Am J Pathol 165(1):63–69
Zhou J, Liyanage U, Medina M et al (1997) Presenilin 1 interaction in the brain with a novel member of the Armadillo family. NeuroReport 8(8):2085–2090
Zorn KK, Bonome T, Gangi L et al (2005) Gene expression profiles of serous, endometrioid, and clear cell subtypes of ovarian and endometrial cancer. Clin Cancer Res 11(18):6422–6430
Acknowledgments
This work was supported by grants from the National Natural Science Foundation of China (No. 30973191). We thank Dr. Shun-ichi Nakamura at Kobe University, Japan, for pCMV5-FLAG/δ-catenin plasmid.
Conflict of interest
No potential conflicts of interest were disclosed.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fang, Y., Li, Z., Wang, X. et al. Expression and biological role of δ-catenin in human ovarian cancer. J Cancer Res Clin Oncol 138, 1769–1776 (2012). https://doi.org/10.1007/s00432-012-1257-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-012-1257-4